Abstract
Data sources
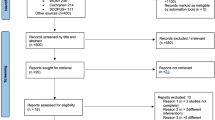
Ovid, Medline, PubMed, Embase and Dentistry and Oral Sciences Source. Databases were searched from January 2006 to March 2016 and restricted to English, manual searches were also carried out in the relevant major journals.
Study selection
Two reviewers independently selected studies. Human prospective and retrospective observational studies with a minimum sample size of ten implants and involving at least one surgical regenerative treatment method for peri-implantitis were considered. Excluded studies included animal and in vitro studies, patients with uncontrolled systemic disease that put the implant at risk and ceramic or coated implants.
Data extraction and synthesis
Data abstraction and quality appraisal were carried out by two independent reviewers. The Cochrane Collaboration tool for assessing risk of bias in randomised trials was used.1 Meta-analysis of similar studies was conducted and the main outcome measures were; changes in radiologic bone level, probing depth and bleeding on probing change.
Results
Eighteen studies were included, eight prospective clinical studies, seven case series and three randomised clinical trials (RCTs). A total of 528 patients with 713 implants were treated. Two studies were at low risk of bias, one moderate, and three high. The remainder were classified as unclear.
Conclusions
Within the limits of this systematic review, surgical regenerative treatment is a predictable option in managing peri-implantitis and improving clinical parameters of peri-implant tissues. There is no fundamental advantage of membrane use for bone graft coverage or submergence of the healing site on the final outcome of peri-implant defect regeneration. Due to the limited number of randomised clinical trials, at the time there is a lack of scientific evidence in the literature regarding the superiority of the regenerative versus non-regenerative surgical treatment.
Similar content being viewed by others
Commentary
Implant treatment has a well-established body of evidence supporting its long-term success and efficacy. Following in the shadow of this success however we now have the problem of peri-implantitis, characterised by inflammation and degeneration of the hard and soft tissues surrounding the implant and eventually leading to its loss from the jaw bone. Various techniques have been advocated to treat this infection taking its origins from periodontal treatment such as non-surgical, surgical and regenerative procedures. The purpose this study was to systematically review the literature on the surgical regenerative treatment of the peri-implantitis and to determine an effective therapeutic predictable option for its clinical management.
In this excellent review Daugela et al. addressed the majority of weaknesses in the evidence base such as short follow-up time, small sample sizes and marked variability in study design leading to high heterogeneity. The absence of control groups was also mentioned, as was a lack of high quality studies, with only two out of the 18 fulfilling the Cochrane Collaboration Risk of Bias Tool criteria. The author concluded that, even though regenerative treatment is a predictable treatment option, there was no advantage in membrane usage or submergence of the healing site in regards to the outcome, including a lack of evidence for the superiority of regenerative versus non-regenerative treatments..
To get the maximum value out of Daugela et al's review, it needs to be paired up with a parallel publication created in conjunction with the lead author's team. This paper by Ramanauskaite was titled 'Surgical non-regenerative treatments for peri-implantitis: a systematic review'.2 The author concluded:
'Surgical non-regenerative treatment of peri-implantitis was found to be effective to reduce the soft tissue inflammation and decrease probing depth. More randomised controlled clinical trials are needed to assess the efficacy of surgical non-regenerative therapy of peri-implantitis.'
The author did not state how effective non-regenerative treatment was even though the primary papers could have been included in a meta-analysis. Without knowing the effectiveness of the standard treatment, in this case, non-regenerative treatment, it becomes difficult to judge the true effectiveness of the regenerative treatment with its increased complexity, morbidity and expense.
To answer this question of efficacy in non-regenerative treatment we have used the mean pocket depth reduction metric, as both papers use this as a primary outcome measure to create a control group. Initially it was necessary to calculate the mean difference at 12 months in pocket depth for the non-regenerative studies, some of the figures have had to be intuited as one paper had a shorter study duration (six months) and missing standard deviations. Additionally, in the other studies we pooled the data where there were no statistical differences between test and control groups. The data were then transformed into a forest plot (Fig. 1).
From this systematic review, weighted post treatment mean pocket reduction was 1.86mm.
The second part of the analysis was to utilise the new mean difference in probing depth as the control for the probing depth reduction in the regenerative studies. As with the first meta-analysis this has been transformed into a forest plot to interpret the results (Fig. 2).
The results of the second meta-analysis confirm that there is a statistical difference between the non-regenerative and regenerative groups. The important item to note is the mean probing depth reduction between the two groups is less than 1mm, and a standard pocket measuring probe is divided into 1mm increments, giving it a maximum accuracy of 0.5mm, suggesting the clinical significance is weak.
An additional point to which the Daugela et al. study alludes is that the mean pre-treatment probing depth for the non-regenerative studies was 5.4mm and for the regenerative studies 6.7mm, creating more variability when trying to find a control for these studies. In effect, the deeper the pocket the more likely the treatment will default to a complex procedure.
Regarding radiographic bone level measurement this could possibly be viewed as a surrogate outcome. A surrogate being a measure of effect of a specific treatment that may correlate with a real clinical endpoint.3,4 As the author mentions, we cannot be certain that the bone seen in the radiograph is truly regenerated bone or just an image of the bone graft and connective tissue that takes up to ten years to resorb.
In conclusion both regenerative and non-regenerative therapies have been shown to reduce pocket depths, but there is only weak evidence as to which treatment is best since the clinical significance is only just measurable. We need to go beyond the conclusion that more long-term randomised control studies are required and follow the advice of John Ioannidis in his paper 'How to make more published research true'.5 Instead of a large number of lead authors trying to investigate the same area in slightly different ways, the profession needs to adopt a collaborative approach and replication culture based on pre-registration, open data sharing and standardisation. This is particularly important in the case of dental research where individual study-centre sample sizes are by their nature small. The lead for this research should be co-ordinated by the international specialist groups such as the European Association of Osseointegration so clinicians can reference high quality research results in the future.
References
Higgins J P, Altman D G, Gøtzsche P C, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ 2011; 343: 889–893.
Ramanauskaite A, Daugela P, Faria de Almeida R, Saulacic N . Surgical Non-Regenerative Treatments for Peri-Implantitis: a Systematic Review. J Oral Maxillofac Res 2016; 7: e14.
Yudkin JS, Lipska KJ, Montori VM . The idolatry of the surrogate. BMJ 2011; 343: d7995.
Heneghan C, Goldacre B, Mahtani KR . Why clinical trial outcomes fail to translate into benefits for patients. Trials 2017; 18: 122.
Ioannidis JP . How to make,more published research true. PLoS Med 2014; 11: e1001747.
Author information
Authors and Affiliations
Additional information
Address for correspondence: Povilas Daugela, Department of Oral and Maxillofacial Surgery, Lithuanian University of Health Sciences, Eiveniu str. 2, LT-50009, Kaunas, Lithuania. E-mail: p.daugela@gmail.com
Abstracted from Daugela P, Cicciù M, Saulacic N. Surgical Regenerative Treatments for Peri-Implantitis: Meta-analysis of Recent Findings in a Systematic Literature Review. J Oral Maxillofac Res 2016; 7: e15. eCollection 2016 Jul-Sep. Review. PubMed PMID: 27833740; PubMed Central PMCID: PMC5100640.
Rights and permissions
About this article
Cite this article
Steven-Howe, M., Richards, D. Surgical regenerative treatment of peri-implantitis. Evid Based Dent 18, 79–81 (2017). https://doi.org/10.1038/sj.ebd.6401256
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ebd.6401256
This article is cited by
-
A modified surgical approach for hard and soft tissue reconstruction of severe periimplantitis defects: laser-assisted periimplant defect regeneration (LAPIDER)
International Journal of Implant Dentistry (2020)