Abstract
NF1 microdeletion syndrome is a common dominant genomic disorder responsible for around 5% of type I neurofibromatosis cases. The majority of cases are caused by mutations arising within the NF1 gene. NF1 microdeletion carriers present a more severe phenotype than patients with intragenic mutations, including mental retardation, cardiac anomalies and dysmorphic features. Here, we report on two brothers with mental retardation presenting a microduplication of the NF1 microdeletion syndrome region detected by array-CGH analysis. Main phenotypic features are mental deficiency, early onset of baldness (15 years old), dental enamel hypoplasia and minor facial dysmorphism. The breakpoint regions coincide with the repeats, and the recombination hot spots shown to mediate NF1 microdeletion through NAHR. A screening of the patients' familial relatives showed that this microduplication segregates in the family for at least two generations. This result demonstrates that both deletion and duplication of the NF1 region, at cytogenetic band 17q11.2, give rise to viable gametes, even if only NF1 microdeletions have been reported until now. Our study reports seven cases of NF1 microduplication within one family. Similar phenotypic abnormalities were present in most of the individuals, however, two displayed a normal phenotype, suggesting a potential incomplete penetrance of the phenotype associated with NF1 microduplication.
Similar content being viewed by others
Introduction
Neurofibromatosis is the most frequent dominant autosomal disorder, whose clinical symptoms include café au lait spots, axillary/inguinal freckling, multiple dermal neurofibromas, Lisch nodules of the iris and malignant peripheral nerve sheath tumours (MPNSTs). While the majority of cases are due to intragenic mutation, in around 5% of cases,1, 2 this disease is caused by deletion of the NF1 gene and surrounding genes. Patients with an NF1 microdeletion present a more severe phenotype than those having intragenic mutations, including facial dysmorphism, mental retardation,3 early onset of multiple neurofibromas and increased risk of MPNSTs.4
Two types of microdeletions have been reported.5 The larger type I microdeletion (1.4–1.5 Mb) is caused6 by non-allelic homologous recombination (NAHR) during meiosis, which is mediated by the presence of two low-copy repeats (LCRs: NF1REPa and NF1REPc). A detailed analysis7 recently reported two recombination hot spots, PRS1 and PRS2, within these repeats, which are also conserved in a homologous LCR present on chromosome 19. The smaller type II microdeletion (1.2 Mb) is completely embedded within the type I deletion region and involves the JJAZ1 gene and its pseudogene, located downstream and upstream of the NF1 gene, respectively. Such microdeletions occur mainly during mitosis and are responsible for most cases with NF1 microdeletion mosaicism.5
The NF1 microdeletion syndrome is included in a group of clinical entities known as genomic disorders. This group of disorders is defined as genetic conditions that result from a recurrent submicroscopic deletion or duplication and in which the associated clinical phenotype is a direct consequence of an abnormal number of dosage-sensitive genes within the rearranged segment.8
Increasing evidence demonstrates that NAHR between highly homologous flanking repeats plays a central role in recurrent genomic disorders, resulting in either duplication, deletion or both of the intervening sequences.9, 10 Since microdeletions and microduplications arise by the same mechanism, theoretically, both derivatives should exist in the population occurring at a similar frequency. In this paper, we describe the first clinical report of affected individuals with microduplication of the NF1 region. Clinical descriptions of seven NF1 microduplication carriers clearly showed a variable phenotype ranging from normal to abnormal. The key phenotypic features are mild mental retardation, early onset of baldness and dental enamel hypoplasia. Examination of the family tree suggested that this microduplication is segregating within the family for at least two generations. This hypothesis was confirmed using multiple ligation-dependent probe amplification (MLPA) to detect duplication of NF1 gene and other genes located in the NF1 microdeletion locus.
Materials and methods
Patient and family information
Patient III.3
A 37-year-old patient (Figures 1 and 2) was referred for genetic evaluation of a mental deficiency. In childhood, he presented with psychomotor and speech delays, and attended a specialized school for learning disabilities since the age of 7 years. He now works in an ‘occupational workplace’ and lives with his mother. Clinical examination showed normal parameters with height at the tenth percentile and a head circumference at the fifth percentile. Phenotypic features are characterized by an early onset of baldness (around 14–15 years of age) with normal hair pigmentation and texture, mild facial dysmorphism with sparse eyelashes and eyebrows, long midface, malar hypoplasia, nasal deviation, bifid nose tip, flared nares, thin upper lip, dental enamel hypoplasia and large testes (35 ml).
Patient III.4
This 38-year-old patient (Figure 2) is the brother of patient III.3. He was previously evaluated at the age of 28 years. He presented the same evolution and learning disabilities. The phenotypic features were almost identical with early baldness, long midface, malar hypoplasia, flared nares, thin upper lip, dental enamel hypoplasia and large testes (40 ml). Moreover, he had increased lower limb reflexes.
Family of the patients
Some clinical information concerning other family members was provided by the index patient's unaffected sister (III.5) and father (II.6), permitting the construction of a family tree (Figure 2), but only individuals II.6, II.9, III.3, III.4, III.5, III.8 and III.9 could be clinically assessed by clinical geneticists.
The patients belong to a sibship of five (two boys and three girls) all of whom were placed into care, in childhood, due to sociocultural problems within the family. Two of the three sisters (III.5 and III.6) have a normal phenotype and the third (III.7) could not be clinically assessed but was reported to have a normal phenotype. She does, however, have a mentally retarded son for whom we have no other clinical data.
Their father (II.6) also had learning disabilities, baldness since the age of 18 years, sparse eyebrows and dental enamel hypoplasia with easily broken teeth in childhood.
The father (II.6) has three mentally retarded half-sisters (II.10, II.11 and II.15), one of whom has sparse hair, three normal half-brothers (II.12, II.13 and II.14) as well as a mentally retarded half-brother (II.9). Individual II.9 (Figure 1) presents a moderate mental retardation, early onset of baldness, sparse eyebrows, dental enamel hypoplasia, short stature with a height at −4 SD and a head circumference at −2.5 SD. He was operated on for large palpebral fissures as a child. The grandmother (I.4) was also apparently mildly mentally retarded, but no other phenotypic information could be obtained. The father of the index patient also has two other sons (III.8 and III.9) from a second union. One of them (III.8) is 6 years old and has normal psychomotor development without dysmorphism. The second (III.9) is 5 years old. He walked at the age of 15 months but presents speech delay and learning disabilities. He has had seizures since the age of 1 year. His height is at the fifth percentile and his head circumference at the third percentile.
Individual II.4 probably died of meningitis in infancy. Individuals III.10 and III.11 died at 13 and 14 years of age, respectively, without clearly established reasons.
DNA material
DNA was extracted from blood leukocytes using either phenol chloroform extraction or ammonium acetate precipitation. Patient III.3 DNA used for CGH microarray analysis was extracted by the QIAamp DNA Blood Midi Kit (Qiagen, Hilden, Germany). NF1 microduplication status was investigated in other family members by MLPA (see below).
Microarray analysis
CGH microarray analysis was performed using a 44K oligonucleotide microarray according to the manufacturer's recommendations (Agilent, Santa Clara, CA, USA). Briefly, 1 μg of patient and reference DNA (pool of seven male DNA; Promega, Madison, WI, USA) were digested with AluI and RsaI (5 U each) and subsequently labelled with Cyanine-5 dUTP and Cyanine-3 dUTP, respectively, using the BioArray Genomic DNA Labelling System (Enzo, Farmingdale, NY, USA).
Fluorescence in situ hybridization
BAC RP11-142O6 and RP11-290N17, mapping between NF1REPa and NF1REPc, were purified by standard alkaline lysis followed by isopropanol precipitation. One microgram of BAC DNA was labelled by nick translation using Spectrum green and Spectrum red (Vysis, Downers Grove, IL, USA) for 6 h at 15°C. Hybridization was performed using standard procedures and analysed using an Applied imaging analysis system.
Multiple ligation-dependent probe amplification assay
MLPA analysis11 was performed using the MLPA P0122 kit obtained from MRC Holland (Amsterdam, The Netherlands). This kit permits screening of both deletion and duplication of genes within the NF1 microdeletion region. Probes 3, 4, 5, 8, 9, 12, 15, 16, 20, 21, 23 and 25 are located in NF1 exon 1, NF1 exon 23-2, NF1 exon 48, CRLF3, JJAZ1, RNF135, FLJ12735, HCA66, CENTA2, LOC114659, NF1 exon 40 and NF1 exon 12B, respectively. Other probes serve as controls and are spread elsewhere in the genome. Fifty nanograms of DNA were used in the ligation reaction. PCR and electrophoresis on an Applied Biosystems 3100 sequencer (Applied Biosystems, Foster City, CA, USA) were performed according to the manufacturer's recommendations.
Results
CGH microarray analysis of patient III.3
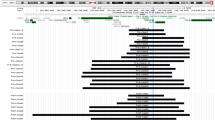
Five copy number variants were observed, two of which correspond to regions reported as being polymorphic (Database of Genomic Variant: http://projects.tcag.ca/variation/). The most striking feature was a duplication of 1.5–1.6 Mb (position 25964192–27475624 on hg18 assembly) on 17q11 with an average log2 ratio of 0.59 consistent with a trisomy. The site of duplication corresponds perfectly to the neurofibromatosis type I microdeletion syndrome region. The genes mapped to this interval, and therefore duplicated in this patient, are listed in Table 1.
Positions of the NF1REPa and NF1REPc repeat regions as defined by Dorschner et al,12 Forbes et al13 and De Raedt et al7 were compared to the breakpoint information obtained from array-CGH. Both repeats are located close to the breakpoint region, suggesting that PRS1 or PRS2 could have mediated the initial recombination breakage (Figure 3).
Alignment of the duplicated region determined by CGH microarray (picture from CGH analytics displaying Agilent 44K CGH array data above) with PRS1 and PRS2 recombination hot spots depicted by arrows with PRS1 and PRS2 labels. Each dot in the above picture corresponds to a log2 ratio value obtained for the microarray probes.
FISH analysis
FISH was performed to determine whether the duplicated region is in tandem or duplicated elsewhere in the genome using RP11-142O6 as a probe. Metaphase analysis showed an enhanced signal at 17q11 on one of the chromosome 17 homologues. Three hybridization signals were visible in interphase nuclei, two of which were located consistently close, consistent with the former hypothesis.
Two colour FISH using BACs located at the extremities of the duplicated region (RP11-142O6 and RP11-290N17) produce a green–red pattern corresponding to the normal chromosome and a green–red–green–red pattern corresponding to the duplicated chromosome (Figure 4). This observation is consistent with the duplication being in a direct orientation.
MLPA survey of a subset of individuals from the family
Ten family members (including the index patient), for whom a DNA sample was available, were investigated by MLPA for copy number changes in the NF1 gene region (Figure 5). NF1 microduplication was observed in six additional individuals: the patient's affected brother (III.4), his apparently normal sister (III.6), their father (II.6), an MR-affected half-uncle (II.9) and the two young half-brothers of the index patient (III.8 and III.9). The other subjects had a normal profile.
MLPA pattern in the NF1 region. Probes are listed along the X-axis as reported in the Salsa MLPA P122 NF1 area probe mix kit from MRC Holland (see Material and method for description). The critical threshold using MLPA is 1.5 for duplication and 0.5 for deletion (dashed lines). Panels 1–4: normal profile observed in control DNA and in unaffected individuals II.13, II.14 and III.5. Panels 5–11: duplicated profile observed in affected individuals II.6, II.9, III.3, III.4, III.9, and unaffected III.6 and III.8.
Discussion
The introduction of array-CGH has revolutionized cytogenetics, allowing investigation of genomic rearrangement at an unprecedented level of resolution unobtainable by karyotyping or FISH.14 This technique has allowed the identification of several new recurrent microduplications and deletions associated with genomic disorders. It is known that these rearrangements are mediated by the presence of flanking repetitive sequences and result from NAHR between them.10 Since this mechanism can produce both duplication and deletion, it would be expected that both reciprocal products would be present in the population at the same frequency. Indeed, this has been shown to be the case, and there are an increasing number of regions where both a deletion and a corresponding duplication disorder have been described. To date, at least 13 such reciprocal recombination disorders have been identified.15 The most well-known example is the duplication, within 17p12, responsible for Charcot–Marie–Tooth syndrome and the corresponding deletion responsible for the hereditary neuropathy with liability to pressure palsies. Other regions include the Smith–Magenis deletion and Potocki–Lupski duplication syndrome at 17p11.2, as well as rearrangements at 7q11.23, 22q11 and 15q11.2.
In this paper, we report seven members of a family with a 1.5-Mb microduplication corresponding to the type I NF1 microdeletion region within 17q11. Given the prevalence of neurofibromatosis (1/2500–1/4000),16 and the fact that 5% arise from microdeletion,1, 2 we can conclude that the incidence of NF1 microdeletion in the population is expected to be around 1/50 000–1/80 000. Since both microdeletion and microduplication are by-products of NAHR, it would be expected that NF1 microduplication would occur at the same frequency. It is therefore surprising that more cases have not been identified despite the large number of cases analysed by this technique. Only one other case with an NF1 microduplication has been reported15 as part of a series of cases analysed by array-CGH. Unfortunately, no clinical information was provided for this individual. One possible explanation for the rarity of such cases is that this rearrangement is unstable and reverts to either a normal copy number or deletion.12 However, our results do not support this hypothesis since we demonstrate that the microduplication, which probably originates from the common mentally retarded grandmother (I.4), segregates within the two half-sibs families and is thus stable over at least two rounds of meiosis. Alternatively, the apparent lower incidence of microduplication could be due to the associated mild phenotype making carrier identification difficult. Indeed of the seven NF1 duplication carriers identified in our study, five clearly display an abnormal phenotype (II.6, II.9, III.3, III.4 and III.9), comprising most of the features reported in the clinical description of the index patient, while two display a normal phenotype (III.6 and III.8). It should, however, be noted that we have only indirect phenotypic evaluation for patient III.6 and that patient III.8 may still be too young to display all features. The apparently normal phenotype of these two individuals does not, however, exclude a causal relationship between NF1 duplication and the abnormal phenotype observed in other family members since incomplete penetrance and variable expressivity have been described for several genomic disorders, including 22q11 deletion17 and duplication18, 19 syndromes.
A last possibility, which cannot be fully ruled out, is that the origin of the phenotype observed in this family is not linked to the NF1 microduplication but has an environmental or other unidentified genetic origin. However, given the concomitant occurrence of the striking phenotypic features reported in this paper and the NF1 microduplication, this option seems unlikely to us. To further determine the pathogenetic consequence of NF1 microduplication, additional individuals need to be identified and clinically evaluated.
The clinical phenotype associated with genomic disorders is thought to result from the aberrant expression of dosage-sensitive genes located within the duplicated or deleted region. The NF1 microdeletion region encompasses 13 genes (Table 1), several of which have been investigated to explain the associated phenotypic characteristics. JJAZ1 and OMG have been proposed as candidate genes with a potential role in mental retardation. The former,5 which is completely deleted or disrupted in type I and type II NF1, is expressed in the brain, and the latter3 encodes a protein described as a potent inhibitor of neurite outgrowth.20, 21 Candidate genes for the observed cardiopathy3 are JJAZ1 and CENTA2, which are expressed in the heart.
The cases described here are trisomic for this region. Although mildly mentally retarded, no cardiopathy was present on clinical evaluation. It may be that altered gene dosage of the proposed candidate genes may also be responsible for the mental impairment observed in these cases. No obvious candidate gene responsible for the patient's other clinical features, such as the early onset of baldness and the dental defects, could be identified. However, the function of many genes in the duplicated interval is either not known or is only partially characterised.
In conclusion, we report for the first time seven patients presenting a microduplication of 17q11, which corresponds to the NF1 type 1 microdeletion. The breakpoints coincide with the NF1REPa and NF1REPc repeats, and duplication is therefore likely to have arisen through NAHR. This microduplication is stable over at least two generations. These patients show moderate mental impairment to borderline normal ability, associated with some recurrent features, such as early onset of baldness (14–15 years of age) and dental enamel hypoplasia.
Despite the absence of reported cases with microduplication, it seems reasonable that further cases will be described and that their identification may explain some unsolved cases of mental retardation. The identification of such individuals will help to establish the genetic and clinical aspects associated with NF1 microduplication.
References
Cnossen MH, van der Est MN, Breuning MH et al: Deletions spanning the neurofibromatosis type 1 gene: implications for genotype–phenotype correlations in neurofibromatosis type 1? Hum Mutat 1997; 9: 458–464.
Kluwe L, Siebert R, Gesk S et al: Screening 500 unselected neurofibromatosis 1 patients for deletions of the NF1 gene. Hum Mutat 2004; 23: 111–116.
Venturin M, Guarnieri P, Natacci F et al: Mental retardation and cardiovascular malformations in NF1 microdeleted patients points to candidate genes in 17q11.2. J Med Genet 2004; 41: 35–41.
De Raedt T, Brems H, Wolkenstein P et al: Elevated risk for MPNST in NF1 microdeletion patients. Am J Hum Genet 2003; 72: 1288–1292.
Kehrer-Sawatzki H, Kluwe L, Sandig C et al: High frequency of mosaicism among patients with neurofibromatosis type 1 (NF1) with microdeletions caused by somatic recombination of the JJAZ1 gene. Am J Hum Genet 2004; 75: 410–423.
Mantripragada KK, Thuresson A-C, Piotrowski A et al: Identification of novel deletion breakpoints bordered by segmental duplications in the NF1 locus using high resolution array-CGH. J Med Genet 2006; 43: 28–38.
De Raedt T, Stephens M, Heyns I et al: Conservation of hotspots for recombination in low-copy repeats associated with the NF1 microdeletion. Nat Genet 2006; 38: 1419–1423.
Lupski JR : Genomic disorders: structural features of the genome can lead to DNA rearrangement and human disease traits. Trends Genet 1998; 14: 417–422.
Shaw CJ, Lupski JR : Implication of human genome architecture for rearrangement-based disorders: the genomic basis of disease. Hum Mol Genet 2004; 13: R57–R64.
Lupski JR, Stankiewicz P : Genomic disorders: molecular mechanisms for rearrangements and conveyed phenotypes. PloS Genet 2005; 1: e49.
Schouten JP, McElgunn CJ, Waaijer R, Zwijnenburg D, Diepvens F, Pals G : Relative quantification of 40 nucleic acid sequences by multiplex ligation-dependent probe amplification. Nucleic Acids Res 2002; 30: e57.
Dorschner MO, Sybert VP, Weaver M, Pletcher BA, Stephens K : NF1 microdeletion breakpoints are clustered at flanking repetitive sequences. Hum Mol Genet 2000; 9: 35–46.
Forbes SH, Dorschner MO, Le R, Stephens K : Genomic context of paralogous recombination hotspots mediating recurrent NF1 region microdeletion. Genes Chromosomes Cancer 2004; 41: 12–25.
Shaffer LG, Bejjani BA : Medical applications of array CGH and the transformation of clinical cytogenetics. Cytogenet Genome Res 2006; 115: 303–309.
Lu X, Shaw CA, Patel A et al: Clinical implementation of chromosomal microarray analysis: summary of 2513 postnatal cases. PloS ONE 2007; 2: e327.
Huson SM, Compston DA, Clark P, Harper PS : A genetic study of von Recklinghausen neurofibromatosis in south east Wales. I. Prevalence, fitness, mutation rate, and effect of parental transmission on severity. J Med Genet 1989; 26: 704–711.
Robin NH, Shprintzen RJ : Defining the clinical spectrum of deletion 22q11.2. J Pediatr 2005; 147: 90–96.
Ensenauer RE, Adeyinka A, Flynn HC et al: Microduplication 22q11.2 an emerging syndrome: clinical, cytogenetic, and molecular analysis of thirteen patients. Am J Hum Genet 2003; 73: 1027–1040.
Yobb TM, Somerville MJ, Willatt L et al: Microduplication and triplication of 22q11.2 a highly variable syndrome. Am J Hum Genet 2005; 76: 865–876.
Wang KC, Koprivica V, Kim JA et al: Oligodendocyte-myelin glycoprotein is a Nogo receptor ligand that inhibits neurite outgrowth. Nature 2002; 417: 941–944.
Venturin M, Moncini S, Villa V et al: Mutations and novel polymorphisms in coding regions and UTRs of CDK5R1 and OMG genes in patients with non-syndromic mental retardation. Neurogenetics 2006; 7: 59–66.
Acknowledgements
We acknowledge M D'Amico for technical assistance in MLPA analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grisart, B., Rack, K., Vidrequin, S. et al. NF1 microduplication first clinical report: association with mild mental retardation, early onset of baldness and dental enamel hypoplasia?. Eur J Hum Genet 16, 305–311 (2008). https://doi.org/10.1038/sj.ejhg.5201978
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejhg.5201978
Keywords
This article is cited by
-
Clinical findings and genetic analysis of patients with copy number variants involving 17p13.3 using a single nucleotide polymorphism array: a single-center experience
BMC Medical Genomics (2022)
-
Emerging genotype–phenotype relationships in patients with large NF1 deletions
Human Genetics (2017)
-
Severe intellectual disability and autistic features associated with microduplication 2q23.1
European Journal of Human Genetics (2012)
-
Copy-number variations associated with neuropsychiatric conditions
Nature (2008)