Abstract
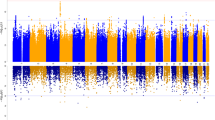
Obsessive-compulsive disorder (OCD) is the tenth most disabling medical condition worldwide. Twin and family studies implicate a genetic etiology for this disorder, although specific genes have yet to be identified. Here, we present the first large-scale model-free linkage analysis of both extended and nuclear families using both ‘broad’ (definite and probable diagnoses) and ‘narrow’ (definite only) definitions of OCD. We conducted a genome-scan analysis of 219 families collected as part of the OCD Collaborative Genetics Study. Suggestive linkage signals were revealed by multipoint analysis on chromosomes 3q27–28 (P=0.0003), 6q (P=0.003), 7p (P=0.001), 1q (P=0.003), and 15q (P=0.006). Using the ‘broad’ OCD definition, we observed the strongest evidence for linkage on chromosome 3q27-28. The maximum overall Kong and Cox LODall score (2.67) occurred at D3S1262 and D3S2398, and simulation based P-values for these two signals were 0.0003 and 0.0004, respectively, although for both signals, the simulation-based genome-wide significance levels were 0.055. Covariate-linkage analyses implicated a possible role of gene(s) on chromosome 1 in increasing the risk for an earlier onset form of OCD. We are currently pursuing fine mapping in the five regions giving suggestive signals, with a particular focus on 3q27–28. Given probable etiologic heterogeneity in OCD, mapping gene(s) involved in the disorder may be enhanced by replication studies, large-scale family-based linkage studies, and the application of novel statistical methods.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Samuels J, Nestadt G . Int Rev Psychiatry 1997; 9: 61–71.
Murray CJ, Lopez AD . The Global Burden of Disease. Harvard University Press, Cambridge, MA, 1996.
Carey G, Gottesman II . Twin and family studies of anxiety, phobic, and obsessive disorders. In: Klein DF, Rabkin JG (eds). Anxiety: New research and changing concepts. Raven Press: NY, 1981, pp 117–136.
Pauls DL, Alsobrook JP, Goodman W, Rasmussen S, Leckman JF . Am J Psychiatry 1995; 152: 76–84.
Nestadt G, Samuels J, Riddle M, Bienvenu III OJ, Liang KY, LaBuda M et al. Arch Gen Psychiatry 2000a; 57: 358–363.
Lenane MC, Swedo SE, Leonard H, Pauls DL, Sceery W, Rapoport JL . J Am Acad Child Adolesc Psychiatry 1990; 29: 407–412.
Riddle MA, Scahill L, King R, Hardin MT, Towbin KE, Ort SI et al. J Am Acad Child Adolesc Psychiatry 1990; 29: 766–772.
Hanna GL, Veenstra-VanderWeele J, Cox NJ, Boehnke M, Himle JA, Curtis GC et al. Am J Med Genet 2002; 114B: 541–552.
Nestadt G, Lan T, Samuels J, Riddle M, Bienvenu III OJ, Liang KY et al. Am J Hum Genet 2000b; 67: 1611–1616.
Nicolini H, Hanna G, Baxter Jr L, Schwartz J, Weissbacker K, Spence MA . Ursus Med 1991; 1: 25–28.
Cavallini MC, Bertelli S, Chiapparino D, Riboldi S, Bellodi L . Am J Med Genet 2000; 96: 384–391.
Samuels JF, Riddle MA, Greenberg BD, Fyer AJ, McCracken JT, Rauch SL et al. Am J Med Genet 2006; 141: 201–207.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th edn, American Psychiatric Association: Washington, DC, 1994.
Mannuzza S, Fyer AJ, Klein DF, Endicott J . J Psychiatr Res 1986; 20: 317–325.
Goodman WK, Price LH, Rasmussen SA, Mazure C, Fleischmann RL, Hill CL et al. Arch Gen Psychiatry 1989; 46: 1006–1011.
Spitzer RL, Williams JBW, Gibbon M, First MB . Arch Gen Psychiatry 1992; 49: 624–629.
Pfohl B, Blum N, Zimmerman M, Stangl D . Structured interview for DSM-IIIR personality SIDR-R. University of Iowa College of Medicine: Iowa City, IA, 1989.
Mannuzza S, Fyer AJ, Endicott J, Klein DF . Family Informant Schedule and Criteria. Anxiety Family Study Unit: New York, NY, New York State Psychiatric Institute: NY, 1985.
Young A . GAS Manual. Oxford University: Oxford, 1998, Version 2.3.
Broman KW, Weber JL . Am J Hum Genet 1998; 63: 1563–1564.
O'Connell JR, Weeks DE . Am J Hum Genet 1998; 63: 259–266.
Douglas JA, Skol AD, Boehnke M . Am J Hum Genet 2002; 70: 487–495.
Abecasis GR, Cherny SS, Cookson WO, Cardon LR . Nat Genet 2002; 30: 97–101.
Kong A, Cox NJ . Am J Hum Genet 1997; 61: 1179–1188.
Sengul H, Weeks DE, Feingold E . Am J Hum Genet 2001; 69: 179–190.
Davis S, Weeks DE . Am J Hum Genet 1997; 61: 1431–1444.
Olson JM . Am J Hum Genet 1999; 65: 1760–1769.
Kruglyak L, Daly M . Am J Hum Genet 1998; 62: 994–997.
Kruglyak L, Daly M . Am J Hum Genet 1998; 74: 20–39.
Olson JM, Goddard KA, Dudek DM . Am J Hum Genet 2001; 69: 895–899.
SAGE. Department of Epidemiology and Biostatistics, Rammelkamp Center for Education and Research, MetroHealth Campus. Case Western Reserve University: Cleveland, 2005.
Meira-Lima I, Shavitt RG, Miguita K, Ikenaga E, Miguel EC, Vallada H . Genes Brain Behav 2004; 3: 75–79.
Niesler B, Frank B, Kapeller J, Rappold GA . Gene 2003; 22: 101–111.
Samuels J, Bienvenu III OJ, Riddle MA, Cullen BAM, Grados MA, Liang K-Y et al. Behav Res Therapy 2002; 40: 517–528.
Miguel EC, Leckman JF, Rauch S, Rosário-Campos MC, Hounie AG, Mercadante MT et al. Mol Psychiatry 2005; 10: 258–275.
Lander E, Kruglyak L . Nat Genet 1995; 11: 241–247.
Acknowledgements
Genotyping services at CIDR was funded through the National Institutes of Health via The Johns Hopkins University, Contract Number N01-HG-65403. We are grateful to the numerous individuals and families who generously donated their time and resources, and to the Obsessive Compulsive Foundation. We thank David Houseman, MD, Kathleen Merikangas, PhD, Ann Pulver, PhD, and Alec Wilson, PhD, for consultation; and clinicians and coordinators at each site: Providence (Maria Mancebo, PhD, Richard Marsland, RN, Shirley Yen, PhD); New York (Renee Goodwin, PhD, Joshua Lipsitz, PhD); Baltimore (Laura Eisen, BS; Karan Lamb, PsyD, Tracey Lichner, PhD, Yung-mei Leong, PhD; Krista Vermillion, BA, Ying Wang, MS); Boston (Dan Geller, MD, Anne Chosak, PhD, Michelle Wedig, BS, Evelyn Stewart, MD, Michael Jenike, MD, Beth Gershuny, PhD, Sabine Wilhelm, PhD); Bethesda (Lucy Justement, Diane Kazuba, V Holland LaSalle-Ricci, Theresa B DeGuzman); Los Angeles (R Lindsey Bergman, PhD, Susanna Chang, PhD, Audra Langley, PhD, Amanda Pearlman, BA). This study is supported by NIMH R01 MH50214 to GN, K23-MH64543 to OJB, and 7K23MH066284 to MAG from the National Institute of Mental Health, and NIH, NCRR, OPD-GCRC RR 00052 from the National Institutes of Health. YYS is, in part, supported by the Department of Epidemiology, Johns Hopkins Bloomberg School of Public Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shugart, Y., Samuels, J., Willour, V. et al. Genomewide linkage scan for obsessive-compulsive disorder: evidence for susceptibility loci on chromosomes 3q, 7p, 1q, 15q, and 6q. Mol Psychiatry 11, 763–770 (2006). https://doi.org/10.1038/sj.mp.4001847
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.mp.4001847
Keywords
This article is cited by
-
Revealing the complex genetic architecture of obsessive–compulsive disorder using meta-analysis
Molecular Psychiatry (2018)
-
Glutamate-system defects behind psychiatric manifestations in a familial hemiplegic migraine type 2 disease-mutation mouse model
Scientific Reports (2016)
-
Common variants of HTR3 genes are associated with obsessive-compulsive disorder and its phenotypic expression
Scientific Reports (2016)
-
Whole-genome association analysis of treatment response in obsessive-compulsive disorder
Molecular Psychiatry (2016)