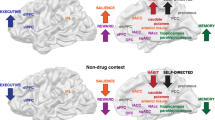
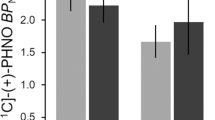
Abstract
The involvement of dopamine in drug reinforcement is well recognized but its role in drug addiction is much less clear. Imaging studies have shown that the reinforcing effects of drugs of abuse in humans are contingent upon large and fast increases in dopamine that mimic but exceed in the intensity and duration those induced by dopamine cell firing to environmental events. In addition, imaging studies have also documented a role of dopamine in motivation, which appears to be encoded both by fast as well as smooth DA increases. Since dopamine cells fire in response to salient stimuli, the supraphysiological activation by drugs is likely to be experienced as highly salient (driving attention, arousal conditioned learning and motivation) and may also reset the thresholds required for environmental events to activate dopamine cells. Indeed, imaging studies have shown that in drug-addicted subjects, dopamine function is markedly disrupted (decreases in dopamine release and in dopamine D2 receptors in striatum) and this is associated with reduced activity of the orbitofrontal cortex (neuroanatomical region involved with salience attribution and motivation and implicated in compulsive behaviors) and the cingulate gyrus (neuroanatomical region involved with inhibitory control and attention and implicated in impulsivity). However, when addicted subjects are exposed to drug-related stimuli, these hypoactive regions become hyperactive in proportion to the expressed desire for the drug. We postulate that decreased dopamine function in addicted subjects results in decreased sensitivity to nondrug-related stimuli (including natural reinforcers) and disrupts frontal inhibition, both of which contribute to compulsive drug intake and impaired inhibitory control. These findings suggest new strategies for pharmacological and behavioral treatments, which focus on enhancing DA function and restoring brain circuits disrupted by chronic drug use to help motivate the addicted subject in activities that provide alternative sources of reinforcement, counteract conditioned responses, enhance their ability to control their drive to take drugs and interfere with their compulsive administration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Koob GF, Bloom FE . Cellular molecular mechanism of drug dependence. Science 1988; 242: 715–723.
Di Chiara G, Imperato A . Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc Nat Acad Sci USA 1988; 85: 5274–5278.
Schultz W, Trembal L, Hollerman JR . Reward processing in primate orbitofrontal cortex and basal ganglia. Cereb Cortex 2000; 10: 272–284.
Zink CF, Pagnoni G, Martin ME, Dhamala M, Berns GS . Human striatal response to salient nonrewarding stimuli. J Neurosci 2003; 23: 8092–8097.
McClure SM, Daw ND, Montague PR . A computational substrate for incentive salience. Trends Neurosci 2003; 25: 423–428.
Fenu S, Di Chiara G . Facilitation of conditioned taste aversion learning by systemic amphetamine: role of nucleus accumbens shell dopamine D1 receptors. Eur J Neurosci 2003; 18: 2025–2030.
Ritz MC, Lamb RJ, Goldberg SR, Kuhar MJ . Cocaine receptors on dopamine transporters are related to self-administration of cocaine. Science 1987; 237: 1219–1223.
Parran TV, Jasinski DR . Intravenous methylphenidate abuse: prototype for prescription drug abuse. Arch Intern Med 1991; 151: 781–783.
Fischman MW, Foltin RW . Utility of subjective-effects measurements in assessing abuse liability of drugs in humans. Br J Addict 1991; 86: 1563–1570.
Volkow ND, Ding YS, Fowler JS, Wang GJ, Logan J, Gatley JS et al. Is methylphenidate like cocaine? Studies on their pharmacokinetics and distribution in human brain. Arch Gen Psychiatry 1995; 52: 456–463.
Madras BK, Fahey MA, Bergman J, Canfield DR, Spealman RD . Effects of cocaine and related drugs in non-human primates. I [3H]cocaine binding sites in caudate-putamen. J Pharmacol Exp Ther 1989; 251: 131–141.
Volkow ND, Wang G-J, Fischman MW, Foltin RW, Fowler JS, Vitkun S et al. Relationship between subjective effects of cocaine and dopamine transporter occupancy. Nature 1997b; 386: 827–830.
Volkow ND, Wang G-J, Fowler JS, Gatley SJ, Logan J, Ding Y-S et al. Blockade of striatal dopamine transporters by intravenous methylphenidate is not sufficient to induce self-reports of ‘High’. J Pharmacol Exp Ther 1999a; 288: 14–20.
Volkow ND, Wang GJ, Fowler JS, Fischman M, Foltin R, Gatley SJ et al. Methylphenidate and cocaine have a similar in vivo potency to block dopamine transporters in the human brain. Life Sci 1999b; 65: 7–12.
Egilmez Y, Jung ME, Lane JD, Emmett-Oglesby MW . Dopamine release during cocaine self administration: effect of SCH23390. Brain Res 1995; 701: 142–150.
Volkow ND, Wang GJ, Fowler JS, Logan J, Schlyer D, Hitzemann R et al. Imaging endogenous dopamine competition with raclopride in the human brain. Synapse 1994; 16: 255–262.
Volkow ND, Fowler JS, Gatley SJ, Dewey SL, Wang GJ, Logan J et al. Comparable changes in synaptic dopamine induced by methylphenidate and by cocaine in the baboon brain. Synapse 1999; 31: 59–66.
Volkow ND, Wang G-J, Fowler JS, Logan J, Gatley SJ, Wong C et al. Reinforcing effects of psychostimulants in humans are associated with increases in brain dopamine and occupancy of D2 receptors. J Pharmacol Exp Ther 1999; 291: 409–415.
Schlaepfer TE, Pearlson GD, Wong DF, Marenco S, Dannals RF . PET study of competition between intravenous cocaine and raclopride at dopamine receptors in human subjects. Am J Psychiatry 1997; 154: 1209–1213.
Drevets WC, Gautier C, Price JC, Kupfer DJ, Kinahan PE, Grace AA et al. Amphetamine-induced dopamine release in human ventral striatum correlates with euphoria. Biol Psychiatry 2001; 49: 81–96.
Laruelle M, Abi-Dargham A, van Dyck CH, Rosenblatt W, Zea-Ponce Y, Zoghbi SS et al. SPECT imaging of striatal dopamine release after amphetamine challenge. J Nucl Med 1995; 36: 1182–1190.
Pecina S, Cagniard B, Berridge KC, Aldridge JW, Zhuang X . Hyperdopaminergic mutant mice have higher ‘wanting’ but not ‘liking’ for sweet rewards. J Neurosci 2003; 23: 9395–9402.
Bassareo V, De Luca MA, Di Chiara G . Differential expression of motivational stimulus properties by dopamine in nucleus accumbens shell versus core and prefrontal cortex. J Neurosci 2002; 22: 4709–4719.
Seeman P, Madras BK . Anti-hyperactivity medication: methylphenidate and amphetamine. Mol Psychiatry 1998; 3: 386–396.
Volkow ND, Wang G, Fowler JS, Logan J, Gerasimov M, Maynard L et al. Therapeutic doses of oral methylphenidate significantly increase extracellular dopamine in the human brain. J Neurosci 2001a; 21: RC121.
Bell K, Duffy P, Kalivas PW . Context-specific enhancement of glutamate transmission by cocaine. Neuropsychopharmacology 2000; 23: 335–344.
Robinson TE, Berridge KC . The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res Rev 1993; 18: 247–291.
Mitchell SH, Laurent CL, de Wit H . Interaction of expectancy and the pharmacological effects of D-amphetamine: subjective effects and self-administration. Psychopharmacology (Berl) 1996; 125: 371–378.
Kirk JM, Doty P, De Wit H . Effects of expectancies on subjective responses to oral delta 9-tetrahydrocannabinol. Pharmacol Biochem Behav 1998; 59: 287–293.
Duvauchelle CL, Ikegami A, Asami S, Robens J, Kressin K, Castaneda E . Effects of cocaine context on NAcc dopamine and behavioral activity after repeated intravenous cocaine administration. Brain Res 2000; 862: 49–58.
Hemby SE, Co C, Koves TR, Smith JE, Dworkin SI . Differences in extracellular dopamine concentrations in the nucleus accumbens during response-dependent and response-independent cocaine administration in the rat. Psychopharmacology (Berl) 1997; 133: 7–16.
Sokoloff L . Relation between physiological function and energy metabolism in the central nervous system. J Neurochem 1977; 29: 13–26.
Graham J, Porrino LJ . Neuroanatomical substrates of cocaine self-administration. In: Hammer R (ed) Neurobiology of Cocaine. CRC Press: Boca Raton, FL, 1995 pp 3–14.
Knapp CM, Printseva B, Cottam N, Kornetsky C . Effects of cue exposure on brain glucose utilization 8 days after repeated cocaine administration. Brain Res 2002; 950: 119–126.
Volkow ND, Wang GJ, Ma Y, Fowler JS, Zhu W, Maynard L et al. Expectation enhances the regional brain metabolic and the reinforcing effects of stimulants in cocaine abusers. J Neurosci 2003; 23: 11461–11468.
Volkow ND, Wang GJ, Fowler JS, Logan J, Jayne M, Franceschi D et al. ‘Nonhedonic’ Food motivation in humans involves dopamine in the dorsal striatum and methylphenidate amplifies this effect. Synapse 2002a; 44: 175–180.
Volkow ND, Wang GJ, Fowler JS, Telang F, Maynard L, Logan J et al. Evidence that methylphenidate enhances the saliency of a mathematical task by increasing dopamine in the human brain. Am J Psychiatry, (in press).
Volkow ND, Wang G-J, Fowler JS, Logan J, Gatley SJ, Hitzemann R et al. Decreased striatal dopaminergic responsivity in detoxified cocaine abusers. Nature 1997c; 386: 830–833.
Grant S, London ED, Newlin DB, Villemagne VL, Liu X, Contoreggi C et al. Activation of memory circuits during cue-elicited cocaine craving. Proc Natl Acad Sci USA 1996; 93: 12040–12045.
Childress AR, Mozley PD, McElgin W, Fitzgerald J, Reivich M, O'Brien CP . Limbic activation during cue-induced cocaine craving. Am J Psychiatry 1999; 156: 11–18.
Volkow ND, Fowler JS . Addiction, a disease of compulsion and drive: involvement of the orbitofrontal cortex. Cereb Cortex 2000; 10: 318–325.
White NM . Addictive drugs as reinforcers: multiple partial actions on memory systems. Addiction 1996; 91: 921–949.
Cornish JL, Kalivas PW . Glutamate transmission in the nucleus accumbens mediates relapse in cocaine addiction. J Neurosci 2000; 20: RC89.
Volkow ND, Wang G-J, Fowler JS, Logan J, Franceschi D, Maynard L et al. Relationship between blockade of dopamine transporters by oral methylphenidate and the increases in extracellular dopamine: therapeutic implications. Synapse 2002b; 43: 181–187.
Garavan H, Pankiewicz J, Bloom A, Cho JK, Sperry L, Ross TJ et al. Cue-induced cocaine craving: neuroanatomical specificity for drug users and drug stimuli. Am J Psychiatry 2000; 157: 1789–1798.
Martin-Soelch C, Chevalley AF, Kunig G, Missimer J, Magyar S, Mino A et al. Changes in reward-induced brain activation in opiate addicts. Eur J Neurosci 2001; 14: 1360–1368.
Gurevich EV, Joyce JN . Distribution of dopamine D3 receptor expressing neurons in the human forebrain: comparison with D2 receptor expressing neurons. Neuropsychopharmacology 1999; 20: 60–80.
Malison RT, Best SE, van Dyck CH, McCance EF, Wallace EA, Laruelle M et al. Elevated striatal dopamine transporters during acute cocaine abstinence as measured by [123I] beta-CIT SPECT. Am J Psychiatry 1998; 155: 832–834.
Laine TP, Ahonen A, Torniainen P, Heikkila J, Pyhtinen J, Rasanen P et al. Dopamine transporters increase in human brain after alcohol withdrawal. Mol Psychiatry 1999; 4: 189–191.
McCann UD, Wong DF, Yokoi F, Villemagne V, Dannals RF, Ricaurte GA . Reduced striatal dopamine transporter density in abstinent methamphetamine and methcathinone users: evidence from positron emission tomography studies with [11C]WIN-35,428. J Neurosci 1998; 18: 8417–8422.
Volkow ND, Chang L, Wang GJ, Fowler JS, Leonido-Yee M, Franceschi D et al. Association of dopamine transporter reduction with psychomotor impairment in methamphetamine abusers. Am J Psychiatry 2001; 158: 377–382.
Sekine Y, Iyo M, Ouchi Y, Matsunaga T, Tsukada H, Okada H et al. Methamphetamine-related psychiatric symptoms and reduced brain dopamine transporters studied with PET. Am J Psychiatry 2001; 158: 1206–1214.
Volkow ND, Chang L, Wang GJ, Fowler JS, Franceschi D, Sedler M et al. Loss of dopamine transporters in methamphetamine abusers recovers with protracted abstinence. J Neurosci 2001; 21: 9414–9418.
Crabb JC, Phillips TJ . Genetics of alcohol and other abused drugs. Drug Alcohol Depend 1998; 51: 61–71.
Maldonado R, Saiardi A, Valverde O, Samad TA, Roques BP, Borrelli E . Absence of opiate rewarding effects in mice lacking dopamine D2 receptors. Nature 1997; 388: 586–589.
De Wit H, Wise RA . Blockade of cocaine reinforcement in rats with the dopamine receptor blocker pimozide, but not with the noradrenergic blockers phentolamine or phenoxybenzamine. Can J Psychol 1977; 31: 195–203.
Dagher A, Bleicher C, Aston JA, Gunn RN, Clarke PB, Cumming P . Reduced dopamine D1 receptor binding in the ventral striatum of cigarette smokers. Synapse 2001; 42: 48–53.
Volkow ND, Fowler JS, Wang GJ, Hitzemann R, Logan J, Schlyer DJ et al. Decreased dopamine D2 receptor availability is associated with reduced frontal metabolism in cocaine abusers. Synapse 1993; 14: 169–177.
Volkow ND, Chang L, Wang GJ, Fowler JS, Ding YS, Sedler M et al. Low levels of brain dopamine D(2) receptors in methamphetamine abusers: association with metabolism in the orbitofrontal cortex. Am J Psychiatry 2001; 158: 2015–2021.
Volkow ND, Fowler JS, Wolf AP, Hitzemann R, Dewey S, Bendriem B et al. Changes in brain glucose metabolism in cocaine dependence and withdrawal. Am J Psychiatry 1991; 148: 621–626.
Volkow ND, Wang G-J, Fowler JS, Hitzemann R, Angrist B, Gatley SJ et al. Association of methylphenidate-induced craving with changes in right striato-orbitofrontal metabolism in cocaine abusers: implications in addiction. Am J Psychiatry 1999; 156: 19–26.
Wang G-J, Volkow ND, Fowler JS, Cervany P, Hitzemann RJ, Pappas N et al. Regional brain metabolic activation during craving elicited by recall of previous drug experiences. Life Sci 1999; 64: 775–784.
Franklin TR, Acton PD, Maldjian JA, Gray JD, Croft JR, Dackis CA et al. Decreased gray matter concentration in the insular, orbitofrontal, cingulate, and temporal cortices of cocaine patients. Biol Psychiatry 2002; 51: 134–142.
Goldstein RZ, Volkow ND . Drug addiction and its underlying neurobiological basis: neuroimaging evidence for the involvement of the frontal cortex. Am J Psychiatry 2002; 159: 1642–1652.
Tremblay L, Schultz W . Relative reward preference in primate orbitofrontal cortex. Nature 1999; 398: 704–708.
Tucker DM, Luu P, Pribram KH . Social and emotional self-regulation. Ann NY Acad Sci 1995; 769: 213–239.
Rolls ET . The orbitofrontal cortex and reward. Cereb Cortex 2000; 10: 284–294.
Isaac WL, Nonneman AJ, Neisewander J, Landers T, Bardo MT . Prefrontal cortex lesions differentially disrupt cocaine-reinforced conditioned place preference but not conditioned taste aversion. Behav Neurosci 1989; 103: 345–355.
Bokura H, Yamaguchi S, Kobayashi S . Electrophysiological correlates for response inhibition in a Go/NoGo task. Clin Neurophysiol 2001; 112: 2224–2232.
Insel TR . Towards a neuroanatomy of obsessive–compulsive disorder. Arch Gen Psychiatry 1992; 49: 739–744.
Piazza PV, Rouge-Pont F, Deminiere JM, Kharoubi M, Le Moal M, Simon H . Dopaminergic activity is reduced in the prefrontal cortex and increased in the nucleus accumbens of rats predisposed to develop amphetamine self administration. Brain Res 1991; 567: 169–174.
Thanos PK, Volkow ND, Freimuth P, Umegaki H, Ikari H, Roth G et al. Overexpression of dopamine D2 receptors reduces alcohol self-administration. J Neurochem 2001; 78: 1094–1103.
Volkow ND, Wang G-J, Fowler JS, Logan J, Gatley SJ, Gifford A et al. Prediction of reinforcing responses to psychostimulants in humans by brain dopamine D2 receptor levels. Am J Psychiatry 1999; 156: 1440–1443.
Volkow ND, Wang GJ, Fowler JS, Thanos PP, Logan J, Gatley SJ et al. Brain DA D2 receptors predict reinforcing effects of stimulants in humans: replication study. Synapse 2002; 46: 79–82.
Papp M, Klimek V, Willner P . Parallel changes in dopamine D2 receptor binding in limbic forebrain associated with chronic mild stress-induced anhedonia and its reversal by imipramine. Psychopharmacology 1994; 115: 441–446.
Morgan D, Grant KA, Gage HD, Mach RH, Kaplan JR, Prioleau O et al. Social dominance in monkeys: dopamine D2 receptors and cocaine self-administration. Nat Neurosci 2002; 5: 169–174.
Volkow ND, Fowler JS, Wang GJ . The addicted human brain: insights from imaging studies. J Clin Invest 2003; 111: 1444–1451.
Kreek MJ, LaForge KS, Butelman E . Pharmacotherapy of addictions. Nat Rev Drug Discov 2002; 1: 710–726.
Grabowski J, Roache JD, Schmitz JM, Rhoades H, Creson D, Korszun A . Replacement medication for cocaine dependence: methylphenidate. J Clin Psychopharmacol 1997; 17: 485–488.
Shearer J, Wodak A, van Beek I, Mattick RP, Lewis J . Pilot randomized double blind placebo-controlled study of dexamphetamine for cocaine dependence. Addiction 2003; 98: 1137–1141.
Gatley SJ, Volkow ND, Gifford AN, Ding YS, Logan J, Wang GJ . Model for estimating dopamine transporter occupancy and subsequent increases in synaptic dopamine using positron emission tomography and carbon-11-labeled cocaine. Biochem Pharmacol 1997; 53: 43–52.
Ranaldi R, Pocock D, Zereik R, Wise RA . Dopamine fluctuations in the nucleus accumbens during maintenance, extinction, and reinstatement of intravenous D-amphetamine self-administration. J Neurosci 1999; 19: 4102–4109.
De Wit H, Wise RA . Blockade of cocaine reinforcement in rats with the dopamine receptor blocker pimozide, but not with the noradrenergic blockers phentolamine or phenoxybenzamine. Can J Psychol 1977; 31: 195–203.
Brauer LH, Goudie AJ, de Wit H . Dopamine ligands and the stimulus effects of amphetamine: animal models versus human laboratory data. Psychopharmacology (Berl) 1997; 130: 2–13.
LeDuc PA, Mittleman G . Schizophrenia and psychostimulant abuse: a review and re-analysis of clinical evidence. Psychopharmacology (Berl) 1995; 121: 407–427.
Grunder G, Carlsson A, Wong DF . Mechanism of new antipsychotic medications: occupancy is not just antagonism. Arch Gen Psychiatry 2003; 60: 974–977.
van Harten PN, van Trier JC, Horwitz EH, Matroos GE, Hoek HW . Cocaine as a risk factor for neuroleptic-induced acute dystonia. J Clin Psychiatry 1998; 59: 128–130.
Evans SM, Walsh SL, Levin FR, Foltin RW, Fischman MW, Bigelow GE . Effect of flupenthixol on subjective and cardiovascular responses to intravenous cocaine in humans. Drug Alcohol Depend 2001; 64: 271–283.
Grabowski J, Rhoades H, Silverman P, Schmitz JM, Stotts A, Creson D et al. Risperidone for the treatment of cocaine dependence: randomized, double-blind trial. J Clin Psychopharmacol 2000; 20: 305–310.
Gerasimov MR, Ashby Jr CR, Gardner EL, Mills MJ, Brodie JD, Dewey SL . Gamma-vinyl GABA inhibits methamphetamine, heroin, or ethanol-induced increases in nucleus accumbens dopamine. Synapse 1999; 34: 11–19.
Fadda P, Scherma M, Fresu A, Collu M, Fratta W . Baclofen antagonizes nicotine-, cocaine-, and morphine-induced dopamine release in the nucleus accumbens of rat. Synapse 2003; 50: 1–6.
Dewey SL, Morgan AE, Ashby Jr CR, Horan B, Kushner SA, Logan J et al. A novel strategy for the treatment of cocaine addiction. Synapse 1998; 30: 119–129.
Di Ciano P, Everitt BJ . The GABA(B) receptor agonist baclofen attenuates cocaine- and heroin-seeking behavior by rats. Neuropsychopharmacology 2003; 28: 510–518.
Brodie JD, Figueroa E, Dewey SL . Treating cocaine addiction: from preclinical to clinical trial experience with gamma-vinyl GABA. Synapse 2003; 50: 261–265.
Gonzalez G, Sevarino K, Sofuoglu M, Poling J, Oliveto A, Gonsai K et al. Tiagabine increases cocaine-free urines in cocaine-dependent methadone-treated patients: results of a randomized pilot study. Addiction 2003; 98: 1625–1632.
Johnson BA, Ait-Daoud N, Bowden CL, DiClemente CC, Roache JD, Lawson K et al. Oral topiramate for treatment of alcohol dependence: a randomized controlled trial. Lancet 2003; 361: 1677–1685.
O'Brien CP, Volpicelli LA, Volpicelli JR . Naltrexone in the treatment of alcoholism: a clinical review. Alcohol 1996; 13: 35–39.
Wewers ME, Dhatt R, Tejwani GA . Naltrexone administration affects ad libitum smoking behavior. Psychopharmacology (Berl) 1998; 140: 185–190.
Julian MD, Martin AB, Cuellar B, Rodriguez De Fonseca F, Navarro M et al. Neuroanatomical relationship between type 1 cannabinoid receptors and dopaminergic systems in the rat basal ganglia. Neuroscience 2003; 119: 309–318.
Wallmichrath I, Szabo B . Cannabinoids inhibit striatonigral GABAergic neurotransmission in the mouse. Neuroscience 2002; 113: 671–682.
De Vries TJ, Shaham Y, Homberg JR, Crombag H, Schuurman K, Dieben J et al. A cannabinoid mechanism in relapse to cocaine seeking. Nat Med 2001; 7: 1151–1154.
Kosten TR, George TP, Kosten TA . The potential of dopamine agonists in drug addiction. Expert Opin Invest Drugs 2002; 11: 491–499.
Lamensdorf I, Youdim MB, Finberg JP . Effect of long-term treatment with selective monoamine oxidase A and B inhibitors on dopamine release from rat striatum in vivo. J Neurochem 1996; 67: 1532–1539.
Schiffer WK, Azmoodeh M, Gerasimov M, Volkow ND, Fowler JS, Dewey SL . Selegiline potentiates cocaine-induced increases in rodent nucleus accumbens dopamine. Synapse 2003; 48: 35–38.
George TP, Vessicchio JC, Termine A, Jatlow PI, Kosten TR, O'Malley SS . A preliminary placebo-controlled trial of selegiline hydrochloride for smoking cessation. Biol Psychiatry 2003; 53: 136–143.
Miranda MI, LaLumiere RT, Buen TV, Bermudez-Rattoni F, McGaugh JL . Blockade of noradrenergic receptors in the basolateral amygdala impairs taste memory. Eur J Neurosci 2003; 18: 2605–2610.
Kampman KM, Volpicelli JR, Mulvaney F, Alterman AI, Cornish J, Gariti P et al. Effectiveness of propranolol for cocaine dependence treatment may depend on cocaine withdrawal symptom severity. Drug Alcohol Depend 2001; 63: 69–78.
Bailey DJ, Tetzlaff JE, Cook JM, He X, Helmstetter FJ . Effects of hippocampal injections of a novel ligand selective for the alpha 5 beta 2 gamma 2 subunits of the GABA/benzodiazepine receptor on Pavlovian conditioning. Neurobiol Learn Mem 2002; 78: 1–10.
Franklin TR, Druhan JP . Involvement of the nucleus accumbens and medial prefrontal cortex in the expression of conditioned hyperactivity to a cocaine-associated environment in rats. Neuropsychopharmacology 2000; 23: 633–644.
Hotsenpiller G, Wolf ME . Baclofen attenuates conditioned locomotion to cues associated with cocaine administration and stabilizes extracellular glutamate levels in rat nucleus accumbens. Neuroscience 2003; 118: 123–134.
Kujala T, Karma K, Ceponiene R, Belitz S, Turkkila P, Tervaniemi M et al. Plastic neural changes and reading improvement caused by audiovisual training in reading-impaired children. Proc Natl Acad Sci USA 2001; 98: 10509–10514.
Papanicolaou AC, Simos PG, Breier JI, Fletcher JM, Foorman BR, Francis D et al. Brain mechanisms for reading in children with and without dyslexia: a review of studies of normal development and plasticity. Dev Neuropsychol 2003; 24: 593–612.
Hayes EA, Warrier CM, Nicol TG, Zecker SG, Kraus N . Neural plasticity following auditory training in children with learning problems. Clin Neurophysiol 2003; 114: 673–684.
Fraser C, Power M, Hamdy S, Rothwell J, Hobday D, Hollander I et al. Driving plasticity in human adult motor cortex is associated with improved motor function after brain injury. Neuron 2002; 34: 831–840.
Acknowledgements
This research was supported in part by the US Department of Energy (Office of Biological and Environmental Research), the National Institute on Drug Abuse (Grants No. DA 06891, DA 09490 and DA 06278) and the National Institute on Alcohol Abuse and Alcoholism (AA 09481) and the Office of National Drug Control Policy (ONDCP).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Volkow, N., Fowler, J., Wang, GJ. et al. Dopamine in drug abuse and addiction: results from imaging studies and treatment implications. Mol Psychiatry 9, 557–569 (2004). https://doi.org/10.1038/sj.mp.4001507
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.mp.4001507
Keywords
This article is cited by
-
The novel psychoactive substance 25E-NBOMe induces reward-related behaviors via dopamine D1 receptor signaling in male rodents
Archives of Pharmacal Research (2024)
-
Sex-specific inflammatory and white matter effects of prenatal opioid exposure: a pilot study
Pediatric Research (2023)
-
Hypodopaminergic state of the nigrostriatal pathway drives compulsive alcohol use
Molecular Psychiatry (2023)
-
Neuroimaging and immunological features of neurocognitive function related to substance use in people with HIV
Journal of NeuroVirology (2023)
-
Conjugated polymer nanoparticles and their nanohybrids as smart photoluminescent and photoresponsive material for biosensing, imaging, and theranostics
Microchimica Acta (2022)