Abstract
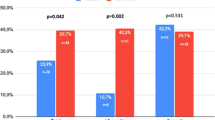
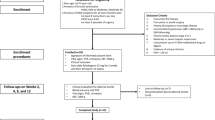
The efficacy and safety of tadalafil, dosed once a day for the treatment of erectile dysfunction, was assessed in a randomized, double-blind, placebo-controlled, parallel-design study at 15 US centers. Following a 4-week treatment-free run-in period, patients (⩾18 years of age) were randomly assigned to 24 weeks treatment with tadalafil 2.5 mg, tadalafil 5 mg or placebo. Primary efficacy endpoints were change at 24 weeks in International Index of Erectile Function Erectile Function (EF) Domain score and mean per-patient percentage ‘yes’ responses to Sexual Encounter Profile diary questions 2 and 3. Tadalafil significantly improved erectile function compared with placebo for all three co-primary efficacy endpoints. Few patients discontinued because of adverse events (2.1%, placebo; 6.3%, tadalafil 2.5 mg; 4.1%, tadalafil 5 mg). Common treatment-emergent adverse events (⩾5%) were nasopharyngitis, influenza, viral gastroenteritis and back pain. Tadalafil 2.5 mg and 5 mg, dosed once a day for 24 weeks, was well tolerated and significantly improved erectile function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
National Institutes of Health Consensus Statement on Impotence, December, 1992. JAMA 1993; 270: 83–90.
Goldstein I, Lue TF, Padma-Nathan H, Rosen RC, Steers WD, Wicker PA . Oral sildenafil in the treatment of erectile dysfunction. Sildenafil Study Group. N Engl J Med 1998; 338: 1397–1404 (Erratum in N Engl J Med 1998; 339: 59).
Porst H, Rosen R, Padma-Nathan H, Goldstein I, Giuliano F, Ulbrich E et al. The efficacy and tolerability of vardenafil, a new, oral, selective phosphodiesterase type 5 inhibitor, in patients with erectile dysfunction: the first at-home clinical trial. Int J Impot Res 2001; 13: 192–199.
Padma-Nathan H, McMurray JG, Pullman WE, Whitaker JS, Saoud JB, Ferguson KM et al. On-demand IC351 (Cialis) enhances erectile function in patients with erectile dysfunction. Int J Impot Res 2001; 13: 2–9.
Hatzimouratidis K, Hatzichristou DG . A comparative review of the options for treatment of erectile dysfunction, which treatment for which patient? Drugs 2005; 65: 1621–1650.
Porst H, Padma-Nathan H, Giuliano F, Anglin G, Varanese L, Rosen R . Efficacy of tadalafil for the treatment of erectile dysfunction at 24 and 36 h after dosing: a randomized controlled trial. Urology 2003; 62: 121–125.
Young JM, Feldman RA, Auerbach SM, Kaufman JM, Garcia CS, Shen W et al. Tadalafil improved erectile function at twenty-four and thirty-six hours after dosing in men with erectile dysfunction: US trial. J Androl 2005; 26: 310–318.
Forgue ST, Patterson BE, Bedding AW, Payne CD, Phillips DL, Wrishko RE et al. Tadalafil pharmacokinetics in healthy subjects. Br J Clin Pharmacol 2005; 61: 280–288.
McMahon C . Efficacy and safety of daily tadalafil in men with erectile dysfunction previously unresponsive to on-demand tadalafil. J Sex Med 2004; 1: 292–300.
McMahon C . Comparison of efficacy, safety, and tolerability of on-demand tadalafil and daily dosed tadalafil for the treatment of erectile dysfunction. J Sex Med 2005; 2: 415–427.
Porst H, Giuliano F, Glina S, Ralph D, Casabé AR, Elion-Mboussa A et al. Evaluation of the efficacy and safety of daily dosing of tadalafil 5 mg and 10 mg in the treatment of erectile dysfunction: results of a multicenter, randomized, double-blind, placebo-controlled trial. Eur Urol 2006 (in press).
Rosen RC, Riley A, Wagner G, Osterloh IH, Kirkpatrick J, Mishra A . The International Index of Erectile Function (IIEF): a multidimensional scale for the assessment of erectile dysfunction. Urology 1997; 49: 822–830.
Swindle RW, Cameron AE, Lockhart DC, Rosen RC . The psychological and interpersonal relationship scales: assessing psychological and relationship outcomes associated with erectile dysfunction and its treatment. Arch Sex Behav 2004; 33: 19–30.
Brock GB, McMahon CG, Chen KK, Costigan T, Shen W, Watkins VS et al. Efficacy and safety of tadalafil in the treatment of erectile dysfunction: results of integrated analyses. J Urol 2002; 168: 1332–1336 (Erratum in J Urol 2005; 173: 664).
Rosen R, Broderick G, Shabsigh R, Swindle R, Lockhart D, Cameron A . Sensitivity of the psychological and interpersonal relationship scales to oral therapies for erectile dysfunction. J Sex Med 2005; 2: 461–468.
Dean J, Hackett G, Vincenzo G, Pirozzi Farina F, Rosen RC, Zhao Y et al. Psychosocial outcomes and drug attributes affecting treatment choice in men receiving sildenafil citrate and tadalafil for the treatment of erectile dysfunction in an open label, randomized, multicenter, crossover study. J Sex Med 2006; 3: 650–661.
Swindle R, Cameron A, Rosen R . A 15-item short form of the psychological and interpersonal relationship scales. Int J Impot Res 2006; 18: 82–88.
Vernet D, Magee TR, Qian A, Nolazco G, Rajfer J, Gonzalez-Cadavid NF . Phosphodiesterase type 5 is not upregulated by tadalafil in cultures of human penile cells. J Sex Med 2006; 3: 84–95.
Ferrini MG, Valente EG, Rajfer J, Gonzolez-Cadavid NF . Long-term treatment with high doses of sildenafil does not up-regulate the levels of phosphodiesterase 5 (PDE5) in the rat penis. J Urol 2004; 171 (Suppl 4): 424.
Musicki B, Champion HC, Becker RE, Kramer MF, Liu T, Sezen SF et al. In vivo analysis of chronic phosphodiesterase-5 inhibition with sildenafil in penile erectile tissues: no tachyphylaxis effect. J Urol 2005; 174: 1493–1496.
Lin G, Xin ZC, Lue TF, Lin CS . Up and down-regulation of phosphodiesterase-5 as related to tachyphylaxis and priapism. J Urol 2003; 170: S15–S18.
McMahon CG, Carson CC, Fischer CJ, Wang WC, Florio VA, Bradley JD . Tolerance to the therapeutic effect of tadalafil does not occur during 6 months of treatment: a randomized, double-blind, placebo-controlled study in men with erectile dysfunction. J Sex Med 2006; 3: 504–511.
Carson CC, Rajfer J, Eardley I, Carrier S, Denne JS, Walker DJ et al. The efficacy and safety of tadalafil: an update. BJU Int 2004; 93: 1276–1281.
Acknowledgements
We acknowledge the additional study investigators: Stephen Auerbach, MD; Bradley Davis, MD; Eugene Dula, MD; Robert Feldman, MD; Darrell N. Fiske, MD; Michael Gambla, MD; Joel Kaufman, MD; Ira Klimberg, MD; Gary Ruoff, MD; David R Talley, MD; and Jay Young, MD. We thank Kate Loughney, PhD, ICOS Corporation for help in preparation of the manuscript and Steven D Watts, MS, Eli Lilly and Company for statistical analyses.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclosure
Study (study code H6D-MC-LVFP) funded by Lilly ICOS LLC.
Conflict of Interest Statements
Dr J Rajfer
Grants, Advisory Board, and Consultant for Lilly ICOS LLC, Pfizer, Bayer, Glaxo SmithKline.
Dr PJ Aliotta
Principal Investigator, Speaker, and Consultant for Lilly ICOS LLC, Pfizer, Glaxo SmithKline, Watson Pharmaceuticals, Novartis, Schering Plough; Principal Investigator and Consultant for Alza Pharmaceuticals, Speaker and Consultant for Boehringer Ingelheim and Ortho McNeil Pharmaceuticals.
Dr CP Steidle
Speaker and consultant for Lilly ICOS LLC, Speaker and Investigator for Pfizer, Bayer GSK.
Dr WP Fitch III
Speaker, Grant Support, Consultant for Lilly ICOS LLC, Speaker and Grant Support for Glaxo SmithKline, Grant Support from Johnson and Johnson.
Dr Y Zhao
Employee, Eli Lilly and Company.
Dr A Yu
Employee, ICOS Corporation.
Rights and permissions
About this article
Cite this article
Rajfer, J., Aliotta, P., Steidle, C. et al. Tadalafil dosed once a day in men with erectile dysfunction: a randomized, double-blind, placebo-controlled study in the US. Int J Impot Res 19, 95–103 (2007). https://doi.org/10.1038/sj.ijir.3901496
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijir.3901496
Keywords
This article is cited by
-
Efficacy and safety of combination of tadalafil and aspirin versus tadalafil or aspirin alone in patients with vascular erectile dysfunction: a comparative randomized prospective study
International Urology and Nephrology (2019)
-
Comparison of the efficacy and safety of 5-mg once-daily versus 5-mg alternate-day tadalafil in men with erectile dysfunction and lower urinary tract symptoms
International Journal of Impotence Research (2015)
-
Konservative Therapie der erektilen Dysfunktion
Der Urologe (2015)
-
Psychosocial outcomes after initial treatment of erectile dysfunction with tadalafil once daily, tadalafil on demand or sildenafil citrate on demand: results from a randomized, open-label study
International Journal of Impotence Research (2014)
-
Retrospective analysis of the efficacy and safety of once-daily tadalafil in patient subgroups: men with mild vs moderate ED and aged <50 vs ⩾50 years
International Journal of Impotence Research (2013)