Abstract
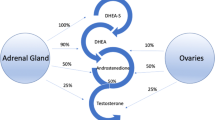
Female sexual dysfunction is a complex problem with multiple overlapping etiologies. Androgens play an important role in healthy female sexual function, especially in stimulating sexual interest and in maintaining desire. There are a multitude of reasons why women can have low androgen levels with the most common reasons being age, oophorectomy and the use of oral estrogens. Symptoms of androgen insufficiency include absent or greatly diminished sexual motivation and/or desire, that is, libido, persistent unexplainable fatigue or lack of energy, and a lack of sense of well being. Although there is no androgen preparation that has been specifically approved by the FDA for the treatment of Women's Sexual Interest/Desire Disorder or for the treatment of androgen insufficiency in women, androgen therapy has been used off-label to treat low libido and sexual dysfunction in women for over 40 y. Most clinical trials in postmenopausal women with loss of libido have demonstrated that the addition of testosterone to estrogen significantly improved multiple facets of sexual functioning including libido and sexual desire, arousal, frequency and satisfaction. In controlled clinical trials of up to 2 y duration of testosterone therapy, women receiving androgen therapy tolerated androgen administration well and demonstrated no serious side effects. The results of these trials suggest that testosterone therapy in the low-dose regimens is efficacious for the treatment of Women's Sexual Interest and Desire Disorder in postmenopausal women who are adequately estrogenized. Based on the evidence of current studies, it is reasonable to consider testosterone therapy for a symptomatic androgen-deficient woman with Women's Sexual Interest and Desire Disorder.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Laumann EO, Paik A, Rosen RC . Sexual dysfunction in the United States: prevalence and predictors. JAMA 1999; 281: 537–544.
Basson R et al. Report of the international consensus development conference on female sexual dysfunction: definitions and classifications. J Urol 2000; 163: 888–893.
Basson R . Female sexual response: the role of drugs in the management of sexual dysfunction. Obstet Gynecol 2001; 98: 350–353.
Basson R et al. Definitions of women's sexual dysfunction reconsidered: advocating expansion and revision. J Psychosom Obstet Gynecol 2003; 24: 221–229.
Ogawa S et al. Survival of reproductive behaviors in estrogen receptor beta gene-deficient male and female mice. Proc Natl Acad Sci USA 1999; 96: 12887–12892.
Cone RD, Low MJ, Elmquist JK, Cameron JL . Neuroendocrinology. In: Larsen PR, Kroneneberg HM, Melmed S, Polonsky KS (eds). Williams Textbook of Endocrinology. Elsevier: New York, 2003 p 134.
Bixo M, Backstrom T, Winblad B, Andersson A . Estradiol and testosterone in specific regions of the human female brain in different endocrine states. J Steroid Biochem Mol Biol 1995; 55: 297–303.
Davis SR, Tran J . Testosterone influences libido and well being in women. Trends Endocrinol Metab 2001; 12: 33–37.
Perksy H et al. Plasma testosterone levels and the sexual behavior of couples. Arch Sex Behav 1978; 7: 157–173.
Morris NM, Udry JR, Khan-Dawood F, Dawood MY . Marital sexual frequency and midcycle female testosterone. Arch Sex Behav 1987; 16: 27–37.
Van Goozen SHM et al. Psychoendocrinological assessment of the menstrual cycle: the relationship between hormones, sexuality, and mood. Arch Sex Behav 1997; 26: 359–382.
Tuiten A et al. Discrepancies between genital responses and subjective sexual function during testosterone substitution in women with hypothalamic amenorrhea. Psychosom Med 1996; 58: 234–241.
Nathorst-Boos J, von Schoultz B . Psychological reactions and sexual life after hysterectomy with and without oophorectomy. Gynecol Obstet Invest 1992; 34: 97–101.
Berman JR et al. Correlation of androgen receptors, aromatase, and 5-alpha reductase in the human vagina with menopausal status. Fertil Steril 2003; 79: 925–931.
Hodgins MB, Spike RC, Mackie RM, MacLean AB . An immunohistochemical study of androgen, oestrogen and progesterone receptors in the vulva and vagina. Br J Obstet Gynaecol 1998; 105: 216–222.
Clayton AH . Sexual function and dysfunction in women. Psychiat Clin N Am 2003; 26: 673–682.
Sarrel PM . Sexuality and menopause. Obstet Gynecol 1990; 75: 26S–30S.
Zumoff B, Strain GW, Miller LK, Rosner W . Twenty-four-hour mean plasma testosterone concentration declines with age in normal premenopausal women. J Clin Endocrinol Metab 1995; 80: 1429–1430.
Lasley BL et al. The relationship of circulating DHEA, testosterone, and estradiol to stages of the menopausal transition and ethnicity. J Clin Endocrinol Metab 2002; 87: 3760–3767.
Burger HG et al. A prospective longitudinal study of serum testosterone, DHEA-S, and SHBG levels through the menopause transition. J Clin Endocrinol Metab 2000; 85: 2832–2838.
Judd HL, Lucas WE, Yen SC . Effects of oophorectomy on circulating testosterone and androsteinedione levels in patients with endometrial cancer. Am J Obstet Gynecol 1974; 118: 793–798.
Hughes CL, Wall LL, Creasman WT . Reproductive hormone levels in gynecologic oncology patients undergoing surgical castration after spontaneous menopause. Gynecol Oncol 1991; 40: 42–45.
Laughlin GA, Barrett-Conner E, Kritz-Silverstein D, Von Muhlen D . Hysterectomy, oophorectomy, and endogenous sex hormone levels in older women: the Rancho Bernardo Study. J Clin Endocrinol Metab 2000; 85: 645–651.
Simon JA . Estrogen replacement therapy and the endogenous androgen milieu. Fertil Steril 2002; 77: s77–s82.
Vehkavaara S et al. Differential effects of oral and transdermal estrogen replacement therapy on endothelial function in postmenopausal women. Circulation 2000; 102: 2687–2693.
Braunstein GD . Androgen insufficiency in women: a summary of critical issues. Fertil Steril 2002; 77: S94–S99.
Bachmann G et al. Female androgen insufficiency: the Princeton consensus statement on definition, classification, and assessment. Fertil Steril 2002; 77: 660–665.
Miller KK et al. Measurement of free testosterone in normal women and women with androgen deficiency: a comparison of methods. J Clin Endocrinol Metab 2004; 89: 525–533.
Sarrel P, Dobay B, Wiita B . Estrogen and estrogen–androgen therapy in postmenopausal women with dissatisfied with estrogen-only therapy. J Reprod Med 1998; 43: 847–856.
Greenblatt RB et al. Evaluation of an androgen, estrogen, estrogen–androgen combination, and a placebo in the treatment of menopause. J Clin Endocrinol Metab 1950; 10: 1547–1558.
Simon JA et al. Transdermal testosterone patch improves sexual activity and desire in sexual activity and desire in surgically menopausal women. Obstet Gynecol 2004; 103 (s): 64s.
Goldsmith CL et al. Esterified estrogens and methyltestosterone: effects on sexual interest and hormone profiles. Obstet Gynecol 2004; 103 (suppl): 63S.
Goldstat R et al. Transdermal testosterone therapy improves well-being, mood, and sexual function in premenopausal women. Menopause 2003; 10: 390–398.
Braunstein GD et al. Testosterone patch for the treatment of low sexual desire in surgically menopausal women. Abstr Menopause 2003; 10: 587.
Lobo RA et al. Comparative effects of oral esterified estrogens with and without methyltestosterone on endocrine profiles and dimensions of sexual function in postmenopausal women with hypoactive desire disorder. Fertil Steril 2003; 79: 1341–1352.
Davis S et al. Efficacy and safety of testosterone patches for the treatment of low sexual desire in surgically menopausal women. Fertil Steril 2003; 30s: 76.
Floter A, Nathorst-Boos K, Carlstrom K, Schoultz B . Addition of testosterone to estrogen replacement in oophorectomized women: effects on sexuality and well-being. Climacteric 2002; 5: 357–365.
Shifren JL et al. Transdermal testosterone treatment in women with impaired sexual function after oophorectomy. New England Journal of Medicine 2000; 343: 682–688.
Davis SR, McCloud P, Strauss BJG, Burger H . Testosterone enhances estradiol's effects on postmenopausal bone density and sexuality. Maturitas 1995; 21: 227–236.
Myers LS et al. Effects of estrogen, androgen, and progestin on sexual psychophysiology and behavior in postmenopausal women. J Clin Endocrinol Metab 1990; 70: 1124–1131.
Burger H, Hailes J, Nelson J, Menelaus M . Effects of combined implants of estradiol and testosterone on libido in postmenopausal women. Br Med J 1987; 294: 936–937.
Sherwin BB, Gelfand MM . Role of androgen in the maintenance of sexual functioning in oophorectomized women. Psychosom Med 1987; 49: 397–409.
Sherwin BB, Gelfand M, Brender W . Androgen enhances sexual motivation in females: a prospective, crossover study of sex steroid administration in the surgical menopause. Psychosom Med 1985; 47: 339–351.
Sherwin BB, Gelfand MM . Differential symptom response to parental estrogen and/or androgen administration in the surgical menopause. Am J Obstet Gynecol 1985; 151: 153–160.
Burger H et al. The management of persistent menopausal symptoms with oestradiol–testosterone implants: clinical, lipid and hormonal results. Maturitas 1984; 6: 351–358.
Dow MG, Hart DM . Hormonal treatments of sexually unresponsiveness in postmenopausal women: a comparative study. Br J Obstet Gynecol 1983; 90: 361–366.
Geist SH . Androgen therapy in gynecology. JAMA 1940; 117: 2207–2213.
Braunstein GD, Cameron DR . Postmenopausal androgen therapy. Female Patient 2004; 29: 1.
Barrett-Connor E et al. A two-year, double blind comparison of estrogen–androgen and conjugated estrogens in surgical menopausal women. Effects on bone mineral density, symptoms and lipid profiles. J Reprod Med 1999; 44: 1012–1020.
Raisz LG et al. Comparison of effects of estrogen alone and estrogen plus androgen on biochemical markers of bone formation and resorption in postmenopausal women. J Clin Endocrinol Metab 1996; 81: 37–43.
Basaria S, Dobs AS . Safety and adverse effects of androgens: how to counsel patients. Mayo Clinic Proc 2004; 79: S25–S32.
Worboys S et al. Evidence that parenteral testosterone therapy may improve endothelium-dependent and -independent vasodilation in postmenopausal women already receiving estrogen. J Clin Endocrinol Metab 2001; 86: 158–161.
Key T, Appleby P, Barnes I, Reeves G . Endogenous hormones and breast cancer collaborative group. Endogenous sex hormones and breast cancer in postmenopausal women: reanalysis of nine prospective studies. J Natl Cancer Inst 2002; 94: 606–616.
Labrie F et al. Endocrine and intracrine sources of androgens in women: inhibition of breast cancer and other roles of androgens and their precursors DHEA. Endocr Rev 2003; 24: 152–182.
Somboonporn W, Davis SR . Testosterone effects on breast: implications for testosterone therapy for women. Endocr Rev 2004; 25: 374–388.
Dimitrakakis C et al. A physiological role for testosterone in limiting estrogenic stimulation of the breast. Menopause 2003; 10: 292–298.
Dimitrakakis C, Jones RA, Liu A, Bondy CA . Breast cancer incidence in postmenopausal women using testosterone in addition to usual hormone therapy. Menopause 2004; 11: 531–535.
Tuckerman EM, Okon MA, Li TC, Laird SM . Do androgens have a direct inhibitory effect on endometrial function? An in vitro study. Fertil Steril 2000; 74: 771–779.
Miller N, Bedard YC, Cooter NB, Shaul DL . Histological changes in the genital tract in transsexual women following androgen therapy. Histopathology 1986; 10: 661–669.
Alexander JL et al. The effects of postmenopausal hormone therapies on female sexual functioning: a review of double-blind, randomized controlled trials. Menopause 2004; 11: 740–765.
Cameron DR, Braunstein GD . Androgen replacement therapy in women. Fertil Steril 2004; 82: 273–289.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bolour, S., Braunstein, G. Testosterone therapy in women: a review. Int J Impot Res 17, 399–408 (2005). https://doi.org/10.1038/sj.ijir.3901334
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijir.3901334
Keywords
This article is cited by
-
Safety and influence of a novel extract of fenugreek on healthy young women: a randomized, double-blinded, placebo-controlled study
Clinical Phytoscience (2021)
-
Improving the sexual activity and reproduction of female zebrafish with high testosterone levels
Scientific Reports (2021)
-
Intrinsic links among sex, emotion, and reproduction
Cellular and Molecular Life Sciences (2018)
-
Elevated free triiodothyronine may lead to female sexual dysfunction in Chinese urban women: A hospital-based survey
Scientific Reports (2017)
-
When and When Not To Use Testosterone for Palliation in Cancer Care
Current Oncology Reports (2014)