Abstract
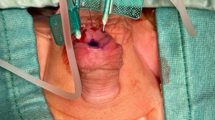
To examine the hypothesis that the glans penis acts protectively, absorbing forces, during coitus. Five potent patients (mean age 46.8±9.7 y), who had indication for surgical excision of the glans for penile carcinoma were included in the present study. Intraoperatively, intracavernosal pressure (ICP) was adjusted by saline infusion and maintained by a pressure feedback infusion pump to a pressure value of 70 mmHg. Using a dynamometer, an external compressive force of 0.5 kg was applied at the glans penis and the changes in ICP were monitored. Measurements were repeated after surgical excision of the glans. Significant ICP changes were noticed in all patients after excision of the glans. Mean preoperative ICP was 161±11.5 mmHg, while after glansectomy it reached 206.6±13 mmHg. ΔICP was 45.8±10.57 mmHg. Two of the patients' partners reported pain during intercourse postoperatively, possibly due to the impact of the force applied by the rigid corpora cavernosa on the anterior vaginal wall without any absorption by the glans. The glans penis restricts the increase in ICP during sexual intercourse, playing a protective role for both the corpora cavernosa and the female genitalia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Lue TF, Tanagho EA . Physiology of erection and pharmacological management of impotence. J Urol 1987; 137: 829–836.
Lue TF . Physiology of penile erection and pathophysiology of erectile dysfunction and priapism. In: Walsh PC, Retik AB, Vaughan ED, Wein AJ (eds). Campell's Urology. Saunders: Philadelphia, 1998, pp 1157–1179.
Lue TF, Tanagho EA . Hemodynamics of erection. In: Tanagho EA, Lue TF, McClure RD (eds). Contemporary Management of Impotence and Infertility. Williams & Wilkins: Baltimore, 1988, pp 28–38.
Hatzichristou DG et al. Glansectomy: an alternative surgical treatment for Buschke–Lowenstein tumors of the penis. Urology 2001; 57: 966–969.
Pescatori ES, Hatzichristou DG, Namburi S, Goldstein I . A positive intracavernous injection test implies normal veno-occlusive but not necessarily normal arterial function: a hemodynamic study. J Urol 1994; 151: 1209–1216.
Goldstein AM, Padma-Nathan H . The microarchitecture of the intracavernosal smooth muscle and the cavernosal fibrous skeleton. J Urol 1990; 144: 1144–1146.
Hatzichristou DG et al. In vivo assessment of trabecular smooth muscle tone, its application in pharmaco-cavernosometry and analysis of intracavernous pressure determinants. J Urol 1995; 153: 1126–1135.
Udelson D et al. Engineering analysis of penile hemodynamic and structural–dynamic relationships: Part I—clinical implications of penile tissue mechanical properties. Int J Impot Res 1998; 10: 15–24.
Nitahara KS, Lue TF . Microscopic anatomy of the penis. In: Carson C, Kirby R, Goldstein I (eds). Textbook of Erectile Dysfunction. Isis Medical Media: Oxford, 1999, pp 31–41.
Udelson D et al. Engineering analysis of penile hemodynamic and structural–dynamic relationships: Part III—clinical considerations of penile hemodynamic and rigidity erectile responses. Int J Impot Res 1998; 10: 89–99.
Berman JR, Goldstein I . Female sexual dysfunction. Urol Clin North Am 2001; 28: 405–416.
Levin RJ . Sex and the human female reproductive tract—what really happens during and after coitus. Int J Impot Res 1998; 10 (Suppl 1): S14–S21.
Karacan I, Moore C, Sahmay S . Measurement of pressure necessary for vaginal penetration. Sleep Res 1985; 14: 269A.
Hardy SB, Whitten PL . Patterning of sexual activity. In: Smus BB, Cheney DL, Seyfarth RM, Wrangham RW, Struhsaker TT (eds). Primate Societies. Chicago University Press: Chicago, 1987, pp 370–384.
Penson DF et al. The hemodynamic pathophysiology of impotence following blunt trauma to the erect penis. J Urol 1992; 148: 1171–1180.
Bitsch M, Kromann-Andersen B, Schou J, Sjontoft E . The elasticity and the tensile strength of tunica albuginea of the corpora cavernosa. J Urol 1990; 143: 642–645.
Hoch Z . Vaginal erotic sensitivity by sexological examination. Acta Obstet Gynecol Scand 1986; 65: 767–773.
Fox CA . Some aspects and implications of coital physiology. J Sex Marital Ther 1976; 2: 205–213.
Kermanidis T . Strength of Materials, Vol. II. Singular Publications: Patras, 1995.
Shigley JE, Mischke CR . Mechanical Engineering Design. McGraw-Hill: New York, 1989.
Abramowitz M, Stegun IA . Handbook of Mathematical Functions with Formulas, Graphs and Mathematical Tables. Dover Publications: New York, 1996.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
The mechanical behaviour of the examined structure in this paper (penis with and without the glans) can be initially modelled as shown in Figure 2.
Both cases (I, II) can be treated as columns subjected to an axial compressive static load, which in the specific application had a value of 0.5 kp.
The observation was made such that the pressures needed to render the two structures (I, II) capable to withstand the load P without buckling are: σI=206±13 mmHg and σII=161±11.5 mmHg, respectively.
This means that stresses σcrI=−σI and σcrII=−σII are developed in each section of the structures, respectively.
Considering buckling of a column, having Young's modulus E, length L, moment of inertia I and section A, and loaded with a compressive axial load, the critical load of components I, II can be expressed according to the following equations:19,20


where V(x) is the deflection function.
Since

Hence,

From equations (1) and (3) we get

Splitting the integral on the left side of the inequality leads to

Considering that A2=A1, I2=I1, E2=E1, inequality (5) can be reduced to

The function V(x) is of the type: V(x)=C, sin (πx/L), where C=constant.
Then

According to Abramowitz-Stegun21 (relationship 4.3.139, p. 78), for α=0, n=2, b=π/L1, and z=x:

From equations (6) and (7) we can write

Similarly, we can write

Applying relationship (4.3.138) from Abramowitz and Stegun:21

Hence,

From equations (8) and (9) we get

For a circular section

From equations (10) and (11)

Inequality (12) is valid because d1, d3>0, d1<d3 → d12<d32 and E1<E3, given that the construction with glans is the initial penis construction, reinforced with the glans, thus leading to a higher value of Young's modulus.
Rights and permissions
About this article
Cite this article
Hatzichristou, D., Tzortzis, V., Hatzimouratidis, K. et al. Protective role of the glans penis during coitus. Int J Impot Res 15, 337–342 (2003). https://doi.org/10.1038/sj.ijir.3901039
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijir.3901039