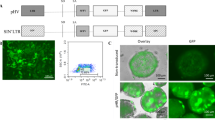
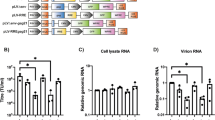
Abstract
Human immunodeficiency virus (HIV)-based vectors are being increasingly used in vitro for gene transfer and in vivo for gene therapy. The proportion of integrated retroviral vectors that are silenced or remain transcriptionally active, and the stability of gene expression in the latter remains poorly explored. To study this, T cells were infected with an HIV-1-based vector construct containing a long terminal repeat-driven reporter gene. Only a small percentage of detectable integrated vector expressed gene product. In clones derived from cells with transcriptionally active vector, gene expression was remarkably stable with more than 80% continuing to express for greater than 18 months. Failure to continue expressing the vector was associated with epigenetic changes. Our data suggest that there are two forms of vector silencing: one occurring immediately after integration affecting the majority of the vectors, and one occurring in the much longer term affecting a small minority of vectors which had previously established expression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Dropulic B, June CH . Gene-based immunotherapy for human immunodeficiency virus infection and acquired immunodeficiency syndrome. Hum Gene Ther 2006; 17: 577–588.
Chun TW, Davey Jr RT, Engel D, Lane HC, Fauci AS . Re-emergence of HIV after stopping therapy. Nature 1999; 401: 874–875.
Davey Jr RT, Bhat N, Yoder C, Chun TW, Metcalf JA, Dewar R et al. HIV-1 and T cell dynamics after interruption of highly active antiretroviral therapy (HAART) in patients with a history of sustained viral suppression. Proc Natl Acad Sci USA 1999; 96: 15109–15114.
Siliciano JD, Kajdas J, Finzi D, Quinn TC, Chadwick K, Margolick JB et al. Long-term follow-up studies confirm the stability of the latent reservoir for HIV-1 in resting CD4+ T cells. Nat Med 2003; 9: 727–728.
Finzi D, Blankson J, Siliciano JD, Margolick JB, Chadwick K, Pierson T et al. Latent infection of CD4+ T cells provides a mechanism for lifelong persistence of HIV-1, even in patients on effective combination therapy. Nat Med 1999; 5: 512–517.
Spina CA, Guatelli JC, Richman DD . Establishment of a stable, inducible form of human immunodeficiency virus type 1 DNA in quiescent CD4 lymphocytes in vitro. J Virol 1995; 69: 2977–2988.
Ruff CT, Ray SC, Kwon P, Zinn R, Pendleton A, Hutton N et al. Persistence of wild-type virus and lack of temporal structure in the latent reservoir for human immunodeficiency virus type 1 in pediatric patients with extensive antiretroviral exposure. J Virol 2002; 76: 9481–9492.
Parera M, Ibanez A, Clotet B, Martinez MA . Lack of evidence for protease evolution in HIV-1-infected patients after 2 years of successful highly active antiretroviral therapy. J Infect Dis 2004; 189: 1444–1451.
Patterson BK, Till M, Otto P, Goolsby C, Furtado MR, McBride LJ et al. Detection of HIV-1 DNA and messenger RNA in individual cells by PCR-driven in situ hybridization and flow cytometry. Science 1993; 260: 976–979.
Embretson J, Zupancic M, Beneke J, Till M, Wolinsky S, Ribas JL et al. Analysis of human immunodeficiency virus-infected tissues by amplification and in situ hybridization reveals latent and permissive infections at single-cell resolution. Proc Natl Acad Sci USA 1993; 90: 357–361.
Embretson J, Zupancic M, Ribas JL, Burke A, Racz P, Tenner-Racz K et al. Massive covert infection of helper T lymphocytes and macrophages by HIV during the incubation period of AIDS. Nature 1993; 362: 359–362.
Peng H, Callison D, Li P, Burrell C . Long-term protection against HIV-1 infection conferred by tat or rev antisense RNA was affected by the design of the retroviral vector. Virology 1996; 220: 377–389.
O'Rourke JP, Newbound GC, Kohn DB, Olsen JC, Bunnell BA . Comparison of gene transfer efficiencies and gene expression levels achieved with equine infectious anemia virus- and human immunodeficiency virus type 1-derived lentivirus vectors. J Virol 2002; 76: 1510–1515.
Bahner I, Kearns K, Hao QL, Smogorzewska EM, Kohn DB . Transduction of human CD34+ hematopoietic progenitor cells by a retroviral vector expressing an RRE decoy inhibits human immunodeficiency virus type 1 replication in myelomonocytic cells produced in long-term culture. J Virol 1996; 70: 4352–4360.
Case SS, Price MA, Jordan CT, Yu XJ, Wang L, Bauer G et al. Stable transduction of quiescent CD34(+)CD38(−) human hematopoietic cells by HIV-1-based lentiviral vectors. Proc Natl Acad Sci USA 1999; 96: 2988–2993.
Luther-Wyrsch A, Costello E, Thali M, Buetti E, Nissen C, Surbek D et al. Stable transduction with lentiviral vectors and amplification of immature hematopoietic progenitors from cord blood of preterm human fetuses. Hum Gene Ther 2001; 12: 377–389.
Lorincz MC, Schubeler D, Goeke SC, Walters M, Groudine M, Martin DI . Dynamic analysis of proviral induction and de novo methylation: implications for a histone deacetylase-independent, methylation density-dependent mechanism of transcriptional repression. Mol Cell Biol 2000; 20: 842–850.
Kuriyama S, Sakamoto T, Kikukawa M, Nakatani T, Toyokawa Y, Tsujinoue H et al. Expression of a retrovirally transduced gene under control of an internal housekeeping gene promoter does not persist due to methylation and is restored partially by 5-azacytidine treatment. Gene Therapy 1998; 5: 1299–1305.
McInerney JM, Nawrocki JR, Lowrey CH . Long-term silencing of retroviral vectors is resistant to reversal by trichostatin A and 5-azacytidine. Gene Therapy 2000; 7: 653–663.
Zentilin L, Qin G, Tafuro S, Dinauer MC, Baum C, Giacca M . Variegation of retroviral vector gene expression in myeloid cells. Gene Therapy 2000; 7: 153–166.
Quivy V, Adam E, Collette Y, Demonte D, Chariot A, Vanhulle C et al. Synergistic activation of human immunodeficiency virus type 1 promoter activity by NF-kappaB and inhibitors of deacetylases: potential perspectives for the development of therapeutic strategies. J Virol 2002; 76: 11091–11103.
Bednarik DP, Cook JA, Pitha PM . Inactivation of the HIV LTR by DNA CpG methylation: evidence for a role in latency. EMBO J 1990; 9: 1157–1164.
Bednarik DP, Mosca JD, Raj NB . Methylation as a modulator of expression of human immunodeficiency virus. J Virol 1987; 61: 1253–1257.
Koiwa T, Hamano-Usami A, Ishida T, Okayama A, Yamaguchi K, Kamihira S et al. 5′-Long terminal repeat-selective CpG methylation of latent human T-cell leukemia virus type 1 provirus in vitro and in vivo. J Virol 2002; 76: 9389–9397.
Parolin C, Dorfman T, Palu G, Gottlinger H, Sodroski J . Analysis in human immunodeficiency virus type 1 vectors of cis-acting sequences that affect gene transfer into human lymphocytes. J Virol 1994; 68: 3888–3895.
Malim MH, Hauber J, Le SY, Maizel JV, Cullen BR . The HIV-1 rev trans-activator acts through a structured target sequence to activate nuclear export of unspliced viral mRNA. Nature 1989; 338: 254–257.
D'Agostino DM, Felber BK, Harrison JE, Pavlakis GN . The Rev protein of human immunodeficiency virus type 1 promotes polysomal association and translation of gag/pol and vpu/env mRNAs. Mol Cell Biol 1992; 12: 1375–1386.
Anson DS, Fuller M . Rational development of a HIV-1 gene therapy vector. J Gene Med 2003; 5: 829–838.
Richardson JH, Child LA, Lever AM . Packaging of human immunodeficiency virus type 1 RNA requires cis-acting sequences outside the 5′ leader region. J Virol 1993; 67: 3997–4005.
Li XD, Moore B, Cloyd MW . Gradual shutdown of virus production resulting in latency is the norm during the chronic phase of human immunodeficiency virus replication and differential rates and mechanisms of shutdown are determined by viral sequences. Virology 1996; 225: 196–212.
Aiken C . Pseudotyping human immunodeficiency virus type 1 (HIV-1) by the glycoprotein of vesicular stomatitis virus targets HIV-1 entry to an endocytic pathway and suppresses both the requirement for Nef and the sensitivity to cyclosporin A. J Virol 1997; 71: 5871–5877.
Ciuffi A, Bleiber G, Munoz M, Martinez R, Loeuillet C, Rehr M et al. Entry and transcription as key determinants of differences in CD4 T-cell permissiveness to human immunodeficiency virus type 1 infection. J Virol 2004; 78: 10747–10754.
Grainger DJ, Lever AM . Blockade of chemokine-induced signalling inhibits CCR5-dependent HIV infection in vitro without blocking gp120/CCR5 interaction. Retrovirology 2005; 2: 23.
Weinberger LS, Burnett JC, Toettcher JE, Arkin AP, Schaffer DV . Stochastic gene expression in a lentiviral positive-feedback loop: HIV-1 Tat fluctuations drive phenotypic diversity. Cell 2005; 122: 169–182.
Nash KL, Lever AM . Green fluorescent protein: green cells do not always indicate gene expression. Gene Therapy 2004; 11: 882–883.
Westervelt P, Henkel T, Trowbridge DB, Orenstein J, Heuser J, Gendelman HE et al. Dual regulation of silent and productive infection in monocytes by distinct human immunodeficiency virus type 1 determinants. J Virol 1992; 66: 3925–3931.
Lusic M, Marcello A, Cereseto A, Giacca M . Regulation of HIV-1 gene expression by histone acetylation and factor recruitment at the LTR promoter. EMBO J 2003; 22: 6550–6561.
Kiernan RE, Vanhulle C, Schiltz L, Adam E, Xiao H, Maudoux F et al. HIV-1 tat transcriptional activity is regulated by acetylation. EMBO J 1999; 18: 6106–6118.
Yu SF, von Ruden T, Kantoff PW, Garber C, Seiberg M, Ruther U et al. Self-inactivating retroviral vectors designed for transfer of whole genes into mammalian cells. Proc Natl Acad Sci USA 1986; 83: 3194–3198.
Logan AC, Nightingale SJ, Haas DL, Cho GJ, Pepper KA, Kohn DB . Factors influencing the titer and infectivity of lentiviral vectors. Hum Gene Ther 2004; 15: 976–988.
Lee YH, Peng CA . Enhanced retroviral gene delivery in ultrasonic standing wave fields. Gene Therapy 2005; 12: 625–633.
Sadelain M . Insertional oncogenesis in gene therapy: how much of a risk? Gene Therapy 2004; 11: 569–573.
Fehse B, Kustikova OS, Bubenheim M, Baum C . Pois(s)on – it's a question of dose. Gene Therapy 2004; 11: 879–881.
Jordan A, Defechereux P, Verdin E . The site of HIV-1 integration in the human genome determines basal transcriptional activity and response to Tat transactivation. EMBO J 2001; 20: 1726–1738.
Mitchell RS, Beitzel BF, Schroder AR, Shinn P, Chen H, Berry CC et al. Retroviral DNA integration: ASLV, HIV, and MLV show distinct target site preferences. PLoS Biol 2004; 2: E234.
Van Lint C . Role of chromatin in HIV-1 transcriptional regulation. Adv Pharmacol 2000; 48: 121–160.
Lehrman G, Hogue IB, Palmer S, Jennings C, Spina CA, Wiegand A et al. Depletion of latent HIV-1 infection in vivo: a proof-of-concept study. Lancet 2005; 366: 549–555.
Gaudet F, Hodgson JG, Eden A, Jackson-Grusby L, Dausman J, Gray JW et al. Induction of tumors in mice by genomic hypomethylation. Science 2003; 300: 489–492.
Seligson DB, Horvath S, Shi T, Yu H, Tze S, Grunstein M et al. Global histone modification patterns predict risk of prostate cancer recurrence. Nature 2005; 435: 1262–1266.
Gluzman Y . SV40-transformed simian cells support the replication of early SV40 mutants. Cell 1981; 23: 175–182.
Schneider U, Schwenk HU, Bornkamm G . Characterization of EBV-genome negative ‘null’ and ‘T’ cell lines derived from children with acute lymphoblastic leukemia and leukemic transformed non-Hodgkin lymphoma. Int J Cancer 1977; 19: 621–626.
Mok HP, Lever A . A method to estimate the efficiency of gene expression from an integrated retroviral vector. Retrovirology 2006; 3: 51.
Reeves MB, MacAry PA, Lehner PJ, Sissons JG, Sinclair JH . Latency, chromatin remodeling, and reactivation of human cytomegalovirus in the dendritic cells of healthy carriers. Proc Natl Acad Sci USA 2005; 102: 4140–4145.
Acknowledgements
We thank Derek Mann, Jelena Mann and Matthew Reeves for helpful advice in ChIP assays, Sarah Noton for advice on CAT assay, and Frederic Bushman, John Sinclair and Jane Greatorex for helpful discussions. This work was supported by the Overseas Research Scholarship, the Cambridge Commonwealth Trust, the Elmore fund and the MRC.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mok, H., Javed, S. & Lever, A. Stable gene expression occurs from a minority of integrated HIV-1-based vectors: transcriptional silencing is present in the majority. Gene Ther 14, 741–751 (2007). https://doi.org/10.1038/sj.gt.3302923
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302923
Keywords
This article is cited by
-
The CpG-sites of the CBX3 ubiquitous chromatin opening element are critical structural determinants for the anti-silencing function
Scientific Reports (2017)
-
Detailed comparison of retroviral vectors and promoter configurations for stable and high transgene expression in human induced pluripotent stem cells
Gene Therapy (2017)
-
A Ubiquitous Chromatin Opening Element (UCOE) Confers Resistance to DNA Methylation–mediated Silencing of Lentiviral Vectors
Molecular Therapy (2010)
-
Efficient entry inhibition of human and nonhuman primate immunodeficiency virus by cell surface-expressed gp41-derived peptides
Gene Therapy (2008)
-
Efficient In Vivo Targeting of Epidermal Stem Cells by Early Gestational Intraamniotic Injection of Lentiviral Vector Driven by the Keratin 5 Promoter
Molecular Therapy (2008)