Abstract
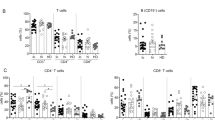
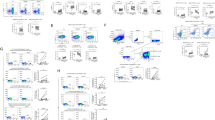
Despite recent advances in the chemotherapy of chronic hepatitis B (CHB), an effective viral suppression after cessation of therapy has not yet been achieved. To investigate whether hepatitis B virus (HBV)-specific T-cell responses are inducible and can contribute to the viral suppression after cessation of the therapy, we conducted a proof-of-concept study with a DNA vaccine comprising of most HBV genes plus genetically engineered interleukin-12 DNA (IL-12N222L) in 12 CHB carriers being treated with lamivudine (LAM). When the ex vivo and/or cultured IFN-γ enzyme-linked immunospot (ELISPOT) assay was performed, the detectable HBV-specific IFN-γ secreting T-cell responses were observed at the end of treatment and during a follow-up. These type 1T-cell responses, particularly CD4+ memory T-cell responses could be maintained for at least 40 weeks after the therapy and correlated with virological responses, but not with alanine aminotransferase elevation. Moreover, DNA vaccination under LAM treatment appeared to be well-tolerated and showed 50% of virological response rate in CHB carriers. Thus, a combination therapy of the DNA vaccine with chemotherapy may be one of new immunotherapeutic methods for the cure of CHB.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Abbreviations
- ALT:
-
alanine aminotransferase
- CHB:
-
chronic hepatitis B
- CTL:
-
cytotoxic T lymphocyte
- ELISPOT:
-
enzyme-linked immunospot
- Env:
-
envelope
- HBeAg:
-
hepatitis B virus e antigen
- HBsAg:
-
hepatitis B virus surface antigen
- HBV:
-
hepatitis B virus
- HBc:
-
hepatitis B virus core
- HCV:
-
hepatitis C virus
- IL-12:
-
interleukin-12
- IFN:
-
interferon
- ISC:
-
IFN-γ secreting cell
- LAM:
-
lamivudine
- PBMC:
-
peripheral blood mononuclear cell
- PCR:
-
polymerase chain reaction
- Pol:
-
polymerase
References
de Jongh FE, Janssen HL, de Man RA, Hop WC, Schalm SW, van Blankenstein M . Survival and prognostic indicators in hepatitis B surface antigen-positive cirrhosis of the liver. Gastroenterology 1992; 103: 1630–1635.
Lok AS, Heathcote EJ, Hoofnagle JH . Management of hepatitis B: 2000 – summary of a workshop. Gastroenterology 2001; 120: 1828–1853.
Chisari FV, Ferrari C . Hepatitis B virus immunopathogenesis. Annu Rev Immunol 1995; 13: 29–60.
Rehermann B, Lau D, Hoofnagle JH, Chisari FV . Cytotoxic T lymphocyte responsiveness after resolution of chronic hepatitis B virus infection. J Clin Invest 1996; 97: 1655–1665.
Rehermann B, Chang KM, McHutchinson J, Kokka R, Houghton M, Rice CM et al. Differential cytotoxic T-lymphocyte responsiveness to the hepatitis B and C viruses in chronically infected patients. J Virol 1996; 70: 7092–7102.
Lai CL, Chien RN, Leung NW, Chang TT, Guan R, Tai DI et al. A one-year trial of lamivudine for chronic hepatitis B. Asia hepatitis lamivudine study group. N Engl J Med 1998; 339: 61–68.
Hadziyannis SJ, Tassopoulos NC, Heathcote EJ, Chang TT, Kitis G, Rizzetto M et al. Adefovir dipivoxil for the treatment of hepatitis B e antigen-negative chronic hepatitis B. N Engl J Med 2003; 348: 800–807.
Boni C, Penna A, Bertoletti A, Lamonaca V, Rapti I, Missale G et al. Transient restoration of anti-viral T cell responses induced by lamivudine therapy in chronic hepatitis B. J Hepatol 2003; 39: 595–605.
Boni C, Penna A, Ogg GS, Bertoletti A, Pilli M, Cavallo C et al. Lamivudine treatment can overcome cytotoxic T-cell hyporesponsiveness in chronic hepatitis B: new perspectives for immune therapy. Hepatology 2001; 33: 963–971.
Rigopoulou EI, Suri D, Chokshi S, Mullerova I, Rice S, Tedder RS et al. Lamivudine plus interleukin-12 combination therapy in chronic hepatitis B: antiviral and immunological activity. Hepatology 2005; 42: 1028–1036.
Mancini M, Hadchouel M, Davis HL, Whalen RG, Tiollais P, Michel ML . DNA-mediated immunization in a transgenic mouse model of the hepatitis B surface antigen chronic carrier state. Proc Natl Acad Sci USA 1996; 93: 12496–12501.
Pancholi P, Lee DH, Liu Q, Tackney C, Taylor P, Perkus M et al. DNA prime/canarypox boost-based immunotherapy of chronic hepatitis B virus infection in a chimpanzee. Hepatology 2001; 33: 448–454.
Mancini-Bourgine M, Fontaine H, Scott-Algara D, Pol S, Brechot C, Michel ML . Induction or expansion of T-cell responses by a hepatitis B DNA vaccine administered to chronic HBV carriers. Hepatology 2004; 40: 874–882.
Maini MK, Boni C, Ogg GS, King AS, Reignat S, Lee CK et al. Direct ex vivo analysis of hepatitis B virus-specific CD8(+) T cells associated with the control of infection. Gastroenterology 1999; 117: 1386–1396.
Ha SJ, Chang J, Song MK, Suh YS, Jin HT, Lee CH et al. Engineering N-glycosylation mutations in IL-12 enhances sustained cytotoxic T lymphocyte responses for DNA immunization. Nat Biotechnol 2002; 20: 381–386.
Ha SJ, Jeon BY, Youn JI, Kim SC, Cho SN, Sung YC . Protective effect of DNA vaccine during chemotherapy on reactivation and reinfection of Mycobacterium tuberculosis. Gene Ther 2005; 12: 634–638.
Prange R, Werr M . DNA-mediated immunization to hepatitis B virus envelope proteins: preS antigen secretion enhances the humoral response. Vaccine 1999; 17: 617–623.
Schirmbeck R, Dikopoulos N, Kwissa M, Leithauser F, Lamberth K, Buus S et al. Breaking tolerance in hepatitis B surface antigen (HBsAg) transgenic mice by vaccination with cross-reactive, natural HBsAg variants. Eur J Immunol 2003; 33: 3342–3352.
Santantonio T, Mazzola M, Iacovazzi T, Miglietta A, Guastadisegni A, Pastore G . Long-term follow-up of patients with anti-HBe/HBV DNA-positive chronic hepatitis B treated for 12 months with lamivudine. J Hepatol 2000; 32: 300–306.
Cavanaugh VJ, Guidotti LG, Chisari FV . Interleukin-12 inhibits hepatitis B virus replication in transgenic mice. J Virol 1997; 71: 3236–3243.
Carreno V, Zeuzem S, Hopf U, Marcellin P, Cooksley WG, Fevery J et al. A phase I/II study of recombinant human interleukin-12 in patients with chronic hepatitis B. J Hepatol 2000; 32: 317–324.
Flanagan KL, Lee EA, Gravenor MB, Reece WH, Urban BC, Doherty T et al. Unique T cell effector functions elicited by Plasmodium falciparum epitopes in malaria-exposed Africans tested by three T cell assays. J Immunol 2001; 167: 4729–4737.
Godkin AJ, Thomas HC, Openshaw PJ . Evolution of epitope-specific memory CD4(+) T cells after clearance of hepatitis C virus. J Immunol 2002; 169: 2210–2214.
Vuola JM, Keating S, Webster DP, Berthoud T, Dunachie S, Gilbert SC et al. Differential immunogenicity of various heterologous prime-boost vaccine regimens using DNA and viral vectors in healthy volunteers. J Immunol 2005; 174: 449–455.
Yoo JK, Cho JH, Lee SW, Sung YC . IL-12 provides proliferation and survival signals to murine CD4+ T cells through phosphatidylinositol 3-kinase/Akt signaling pathway. J Immunol 2002; 169: 3637–3643.
Chang J, Cho JH, Lee SW, Choi SY, Ha SJ, Sung YC . IL-12 priming during in vitro antigenic stimulation changes properties of CD8 T cells and increases generation of effector and memory cells. J Immunol 2004; 172: 2818–2826.
Reece WH, Pinder M, Gothard PK, Milligan P, Bojang K, Doherty T et al. A CD4(+) T-cell immune response to a conserved epitope in the circumsporozoite protein correlates with protection from natural Plasmodium falciparum infection and disease. Nat Med 2004; 10: 406–410.
Wherry EJ, Teichgraber V, Becker TC, Masopust D, Kaech SM, Antia R et al. Lineage relationship and protective immunity of memory CD8 T cell subsets. Nat Immunol 2003; 4: 225–234.
Wang R, Doolan DL, Le TP, Hedstrom RC, Coonan KM, Charoenvit Y et al. Induction of antigen-specific cytotoxic T lymphocytes in humans by a malaria DNA vaccine. Science 1998; 282: 476–480.
Dienstag JL, Schiff ER, Wright TL, Perrillo RP, Hann HW, Goodman Z et al. Lamivudine as initial treatment for chronic hepatitis B in the United States. N Engl J Med 1999; 341: 1256–1263.
Guidotti LG, Ishikawa T, Hobbs MV, Matzke B, Schreiber R, Chisari FV . Intracellular inactivation of the hepatitis B virus by cytotoxic T lymphocytes. Immunity 1996; 4: 25–36.
Guidotti LG, Rochford R, Chung J, Shapiro M, Purcell R, Chisari FV . Viral clearance without destruction of infected cells during acute HBV infection. Science 1999; 284: 825–829.
Maini MK, Boni C, Lee CK, Larrubia JR, Reignat S, Ogg GS et al. The role of virus-specific CD8(+) cells in liver damage and viral control during persistent hepatitis B virus infection. J Exp Med 2000; 191: 1269–1280.
Nowak MA, Bonhoeffer S, Hill AM, Boehme R, Thomas HC, McDade H . Viral dynamics in hepatitis B virus infection. Proc Natl Acad Sci USA 1996; 93: 4398–4402.
Leonard JP, Sherman ML, Fisher GL, Buchanan LJ, Larsen G, Atkins MB et al. Effects of single-dose interleukin-12 exposure on interleukin-12-associated toxicity and interferon-gamma production. Blood 1997; 90: 2541–2548.
Lee H, Lee YH, Huh YS, Moon H, Yun Y . X-gene product antagonizes the p53-mediated inhibition of hepatitis B virus replication through regulation of the pregenomic/core promoter. J Biol Chem 1995; 270: 31405–31412.
Park SH, Yang SH, Lee CG, Youn JW, Chang J, Sung YC . Efficient induction of T helper 1 CD4+ T-cell responses to hepatitis C virus core and E2 by a DNA prime-adenovirus boost. Vaccine 2003; 21: 4555–4564.
Hadziyannis SJ, Vassilopoulos D . Hepatitis B e antigen-negative chronic hepatitis B. Hepatology 2001; 34: 617–624.
Acknowledgements
We thank Dr M Tschaika, E-J Lee, S-Y Eum, and Dr N Opanasyuk for their assistance in processing this study and to Drs J-W Youn and H-T Jin for their revision of the report. This work was supported by grants from National Research Laboratory program of National S&T Program of the Ministry of Science and Technology (2000-N-NL-01-C-202), the Korea Health 21 R&D Project, Ministry of Health & Welfare, Republic of Korea (0405-DB00-0101-0011), the International Cooperation Research Program of the Ministry of Science & Technology (M60401000227-05A0100-22710), POSCO (2000Y013), and Consortium Project (Genexine Co. Ltd, Daewoong Pharm. Co. Ltd, Dong-A Pharm. Co. Ltd, and POSCO).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information accompanies the paper on Gene Therapy website (http://www.nature.com/gt)
Supplementary information
Rights and permissions
About this article
Cite this article
Yang, SH., Lee, CG., Park, SH. et al. Correlation of antiviral T-cell responses with suppression of viral rebound in chronic hepatitis B carriers: a proof-of-concept study. Gene Ther 13, 1110–1117 (2006). https://doi.org/10.1038/sj.gt.3302751
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302751
Keywords
This article is cited by
-
Drugs in Development for Hepatitis B
Drugs (2017)
-
Future Therapy for Hepatitis B Virus: Role of Immunomodulators
Current Hepatology Reports (2016)
-
Therapeutic vaccines in HBV: lessons from HCV
Medical Microbiology and Immunology (2015)
-
Therapeutic vaccines in treating chronic hepatitis B: the end of the beginning or the beginning of the end?
Medical Microbiology and Immunology (2015)
-
Clearance of persistent HPV infection and cervical lesion by therapeutic DNA vaccine in CIN3 patients
Nature Communications (2014)