Abstract
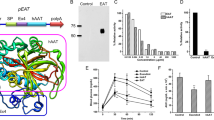
Insulin gene therapy in clinical medicine is currently hampered by the inability to regulate insulin secretion in a physiological manner, the inefficiency with which the gene is delivered, and the short duration of gene expression. To address these issues, we injected the liver of streptozotocin-induced diabetic rats with hemagglutinating virus of Japan-envelope (HVJ-E) vectors containing Epstein–Barr virus (EBV) plasmids encoding the genes for insulin and the GLUT 2 transporter. Efficient delivery of the genes was achieved with the HVJ-E vector, and the use of the EBV replicon vector led to prolonged hepatic gene expression. Blood glucose levels were normalized for at least 3 weeks as a result of the gene therapy. Cotransfection of GLUT 2 with insulin permitted the diabetic rats to regulate their blood glucose levels upon exogenous glucose loading in a physiologically appropriate manner and improved postprandial glucose levels. Moreover, cotransfection with insulin and GLUT 2 genes led to in vitro glucose-stimulated insulin secretion that involved the closure of KATP channels. The present study represents a new way to efficiently deliver insulin gene in vivo that is regulated by ambient glucose level with prolonged gene expression. This may provide a basis to overcome limitations of insulin gene therapy in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Miller AD . Human gene therapy comes of age. Nature 1992; 357: 455–460.
Thompson L . At age 2, gene therapy enters a growth phase. Science 1992; 258: 744–746.
Hollingsworth SJ, Barker SG . Gene therapy: into the future of surgery. Lancet 1999; 353 (Suppl): SI19–SI20.
Yoon JW, Jun HS . Recent advances in insulin gene therapy for type 1 diabetes. Trends Mol Med 2002; 8: 62–68.
Yechoor V, Chan L . Gene therapy progress and prospects: gene therapy for diabetes mellitus. Gene Therapy 2004; 12: 101–107.
Lee HC, Kim SJ, Kim KS, Shin HC, Yoon JW . Remission in models of type 1 diabetes by gene therapy using a single-chain insulin analogue. Nature 2000; 408: 483–488.
Chen R, Meseck ML, Woo SL . Auto-regulated hepatic insulin gene expression in type 1 diabetic rats. Mol Ther 2001; 3: 584–590.
Mitanchez D, Chen R, Massias JF, Porteu A, Mignon A, Bertagna X et al. Regulated expression of mature human insulin in the liver of transgenic mice. FEBS Lett 1998; 421: 285–289.
Liu GJ, Simpson AM, Swan MA, Tao C, Tuch BE, Crawford RM et al. ATP-sensitive potassium channels induced in liver cells after transfection with insulin cDNA and the GLUT 2 transporter regulate glucose-stimulated insulin secretion. FASEB J 2003; 17: 1682–1684.
Min KA, Oh ST, Yoon KH, Kim CK, Lee SK . Prolonged gene expression in primary porcine pancreatic cells using an Epstein–Barr virus-based episomal vector. Biochem Biophys Res Commun 2003; 305: 108–115.
Saeki Y, Wataya-Kaneda M, Tanaka K, Kaneda Y . Sustained transgene expression in vitro and in vivo using an Epstein–Barr virus replicon vector system combined with HVJ liposomes. Gene Therapy 1998; 5: 1031–1037.
Tsujie M, Isaka Y, Nakamura H, Kaneda Y, Imai E, Hori M . Prolonged transgene expression in glomeruli using an EBV replicon vector system combined with HVJ liposomes. Kidney Int 2001; 59: 1390–1396.
Lupton S, Levine AJ . Mapping genetic elements of Epstein–Barr virus that facilitate extrachromosomal persistence of Epstein–Barr virus-derived plasmids in human cells. Mol Cell Biol 1985; 5: 2533–2542.
Yates JL, Warren N, Sugden B . Stable replication of plasmids derived from Epstein–Barr virus in various mammalian cells. Nature 1985; 313: 812–815.
Wu H, Kapoor P, Frappier L . Separation of the DNA replication, segregation, transcriptional activation functions of Epstein–Barr nuclear antigen 1. J Virol 2002; 76: 2480–2490.
Ilan Y, Saito H, Thummala NR, Chowdhury NR . Adenovirus-mediated gene therapy of liver diseases. Semin Liver Dis 1999; 19: 49–59.
Muruve DA, Barnes MJ, Stillman IE, Libermann TA . Adenoviral gene therapy leads to rapid induction of multiple chemokines and acute neutrophil-dependent hepatic injury in vivo. Hum Gene Ther 1999; 10: 965–976.
Park YM, Woo S, Lee GT, Ko JY, Lee Y, Zhao ZS et al. Safety and efficacy of adeno-associated viral vector-mediated insulin gene transfer via portal vein to the livers of streptozotocin-induced diabetic Sprague–Dawley rats. J Gene Med 2005; 7: 621–629.
Kay MA, Glorioso JC, Naldini L . Viral vectors for gene therapy: the art of turning infectious agents into vehicles of therapeutics. Nat Med 2001; 7: 33–40.
Russell DW, Kay MA . Adeno-associated virus vectors and haematology. Blood 1999; 94: 864–874.
Mulligan RC . The basic science of gene therapy. Science 1993; 260: 926–932.
Dzau VJ, Mann MJ, Morishita R, Kaneda Y . Fusigenic viral liposome for gene therapy in cardiovascular diseases. Proc Natl Acad Sci USA 1996; 93: 11421–11425.
Ahn JD, Morishita R, Kaneda Y, Kim HS, Chang YC, Lee KU et al. Novel E2F decoy oligodeoxynucleotides inhibit in vitro vascular smooth muscle cell proliferation and in vivo neointimal hyperplasia. Gene Therapy 2002; 9: 1682–1692.
Ahn JD, Morishita R, Kaneda Y, Kim HJ, Kim YD, Lee HJ et al. Transcription factor decoy for AP-1 reduces mesangial cell proliferation and extracellular matrix production in vitro and in vivo. Gene Therapy 2004; 11: 916–923.
Morishita R, Gibbons GH, Kaneda Y, Ogihara T, Dzau VJ . Systemic administration of HVJ viral coat-liposome complex containing human insulin vector decreases glucose level in diabetic mouse: a model of gene therapy. Biochem Biophys Res Commun 2000; 273: 666–674.
Kaneda Y, Nakajima T, Nishikawa T, Yamamoto S, Ikegami H, Suzuki N et al. Hemagglutinating virus of Japan (HVJ) envelope vector as a versatile gene delivery system. Mol Ther 2002; 6: 219–226.
Burcelin R, Dolci W, Thorens B . Glucose sensing by the hepatoportal sensor is GLUT2-dependent: in vivo analysis in GLUT2-null mice. Diabetes 2000; 49: 1643–1648.
Malhi H, Irani AN, Rajvanshi P, Suadicani SO, Spray DC, McDoland TV et al. KATP channels regulate mitogenically induced proliferation in primary rat hepatocytes and human liver cell lines. Implications for liver growth control and potential therapeutic targeting. J Biol Chem 2000; 275: 26050–26057.
Henderson RM, Graf J, Boyer JL . Inward-rectifying potassium channels in rat hepatocytes. Am J Physiol 1989; 171: G1028–G1035.
Nishikawa M, Huang L . Nonviral vectors in the new millennium: delivery barriers in gene transfer. Hum Gene Ther 2001; 12: 861–870.
Kaneda Y, Iwai K, Uchida T . Introduction and expression of the human insulin gene in adult rat liver. J Biol Chem 1989; 264: 12126–12129.
Calos MP . The potential of extrachromosomal replicating vectors for gene therapy. Trends Genet 1996; 12: 463–466.
Yasutomi K, Itokawa Y, Asada H, Kishida T, Cui FD, Ohashi S et al. Intravascular insulin gene delivery as potential therapeutic intervention in diabetes mellitus. Biochem Biophys Res Commun 2003; 310: 897–903.
Chen ZY, Yant SR, He CY, Meuse L, Shen S . Linear DNAs concatemerize in vivo and result in sustained transgene expression in mouse liver. Mol Ther 2001; 3: 403–410.
Chen ZY, He CY, Ehrhardt A, Kay MA . Minicircle DNA vectors devoid of bacterial DNA result in persistent and high-level transgene expression in vivo. Mol Ther 2003; 8: 495–500.
Fregien N, Davidson N . Activating elements in the promoter region of the chicken beta-actin gene. Gene 1986; 48: 1–11.
Acknowledgements
This work was supported by the Grant No. RTI04-01-01 and RTI04-03-02 from the Regional Technology Innovation Program of the Ministry of Commerce, Industry and Energy (MOCIE). We thank Ishihara Sangyo Kaisha, Ltd (Osaka, Japan) for donating HVJ-E vector.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, Y., Park, KG., Morishita, R. et al. Liver-directed gene therapy of diabetic rats using an HVJ-E vector containing EBV plasmids expressing insulin and GLUT 2 transporter. Gene Ther 13, 216–224 (2006). https://doi.org/10.1038/sj.gt.3302644
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302644
Keywords
This article is cited by
-
Preparation and Evaluation of Gene-transfected Cultured Skin as a Novel Drug Delivery System for Severely Burned Skin
Pharmaceutical Research (2007)