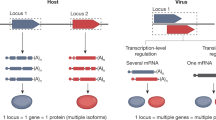
Abstract
Viral vectors are currently the best tools for gene delivery in a therapeutic setting, especially for in vivo use. Alphaviruses, a family of positive singlestranded RNA viruses, have been engineered to allow the formation of a highly efficient replicon. Using these replicons, it is possible to generate recombinant particles. Parental viruses and recombinant vectors share certain pathways while interacting with their target cells. In this review, we describe the consecutive events leading to transduction, and transgene expression, in view of the cellular factors that affect each individual step. Classical virology will benefit from the knowledge accumulated studying vectors, and such work will shed light on crosstalk between intruding viruses and their hosts. Ultimately, these data should help the design of vectors adapted to specific target cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Everts B, Poel HG . Replication-selective oncolytic viruses in the treatment of cancer. Cancer Gene Ther 2004; 8: 8.
Fielding AK . Measles as a potential oncolytic virus. Rev Med Virol 2004; 16: 16.
Larin SS, Georgiev GP, Kiselev SL . Gene transfer approaches in cancer immunotherapy. Gene Therapy 2004; 11 (Suppl 1): S18–S25.
Leitner WW et al. Enhancement of tumor-specific immune response with plasmid DNA replicon vectors. Cancer Res 2000; 60: 51–55.
Smyth JW, Fleeton MN, Sheahan BJ, Atkins GJ . Treatment of rapidly growing K-BALB and CT26 mouse tumours using Semliki Forest virus and its derived vector. Gene Therapy 2004; 16: 16.
Zhang J, Frolov I, Russell SJ . Gene therapy for malignant glioma using Sindbis vectors expressing a fusogenic membrane glycoprotein. J Gene Med 2004; 6: 1082–1091.
Morley S et al. The dl1520 virus is found preferentially in tumor tissue after direct intratumoral injection in oral carcinoma. Clin Cancer Res 2004; 10: 4357–4362.
Shah AC, Benos D, Gillespie GY, Markert JM . Oncolytic viruses: clinical applications as vectors for the treatment of malignant gliomas. J Neurooncol 2003; 65: 203–226.
Rheme C, Ehrengruber MU, Grandgirard D . Alphaviral cytotoxicity and its implication in vector development. Exp Physiol 2004; 12: 12.
Lundstrom K . Biology and application of alphaviruses in gene therapy. Gene Therapy 2005; 12 (suppl 1): S92–S97.
Boorsma M et al. Alphavirus cDNA-based expression vectors: effects of RNA transcription and nuclear export. Biotechnol Bioeng 2003; 81: 553–562.
Ehrengruber MU . Alphaviral gene transfer in neurobiology. Brain Res Bull 2002; 59: 13–22.
Tseng JC et al. In vivo antitumor activity of Sindbis viral vectors. J Natl Cancer Inst 2002; 94: 1790–1802.
Tseng JC et al. Systemic tumor targeting and killing by Sindbis viral vectors. Nat Biotechnol 2004; 22: 70–77. Epub 2003 Nov 2030.
Tseng JC et al. Using Sindbis viral vectors for specific detection and suppression of advanced ovarian cancer in animal models. Cancer Res 2004; 64: 6684–6692.
Dorange F et al. Vesicular stomatitis virus glycoprotein: a transducing coat for SFV-based RNA vectors. J Gene Med 2004; 6: 1014–1022.
Goff SP . Retrovirus restriction factors. Mol Cell 2004; 16: 849–859.
Higgs S, Powers AM, Olson KE . Alphavirus expression systems: applications to mosquito vector studies. Parasitol Today 1993; 9: 444–452.
Liljestrom P . Alphavirus expression systems. Curr Opin Biotechnol 1994; 5: 495–500.
Lundstrom K, Abenavoli A, Malgaroli A, Ehrengruber MU . Novel Semliki Forest virus vectors with reduced cytotoxicity and temperature sensitivity for long-term enhancement of transgene expression. Mol Ther 2003; 7: 202–209.
Polo JM et al. Stable alphavirus packaging cell lines for Sindbis virus and Semliki Forest virus-derived vectors. Proc Natl Acad Sci USA 1999; 96: 4598–4603.
Schlesinger S . Alphavirus vectors: development and potential therapeutic applications. Expert Opin Biol Ther 2001; 1: 177–191.
Smerdou C, Liljestrom P . Two-helper RNA system for production of recombinant Semliki Forest virus particles. J Virol 1999; 73: 1092–1098.
Rolls MM, Haglund K, Rose JK . Expression of additional genes in a vector derived from a minimal RNA virus. Virology 1996; 218: 406–411.
Rolls MM, Webster P, Balba NH, Rose JK . Novel infectious particles generated by expression of the vesicular stomatitis virus glycoprotein from a self-replicating RNA. Cell 1994; 79: 497–506.
Martinez-Salas E, Fernandez-Miragall O . Picornavirus IRES: structure function relationship. Curr Pharm Des 2004; 10: 3757–3767.
Sieczkarski SB, Whittaker GR . Viral entry. Curr Top Microbiol Immunol 2005; 285: 1–23.
Pages JC, Bru T . Toolbox for retrovectorologists. J Gene Med 2004; 6 (Suppl 1): S67–S82.
Bachrach E et al. Efficient cell infection by Moloney murine leukemia virus-derived particles requires minimal amounts of envelope glycoprotein. J Virol 2000; 74: 8480–8486.
Bachrach E et al. Effects of virion surface gp120 density on infection by HIV-1 and viral production by infected cells. Virology 2005; 332: 418–429.
Arudchandran R et al. Polyethylene glycol-mediated infection of non-permissive mammalian cells with Semliki Forest virus: application to signal transduction studies. J Immunol Methods 1999; 222: 197–208.
Strauss JH, Strauss EG . The alphaviruses: gene expression, replication, and evolution. Microbiol Rev 1994; 58: 491–562.
Ohno K et al. Cell-specific targeting of Sindbis virus vectors displaying IgG-binding domains of protein A. Nat Biotechnol 1997; 15: 763–767.
Perri S et al. An alphavirus replicon particle chimera derived from venezuelan equine encephalitis and sindbis viruses is a potent gene-based vaccine delivery vector. J Virol 2003; 77: 10394–10403.
Sandrin V, Russell SJ, Cosset FL . Targeting retroviral and lentiviral vectors. Curr Top Microbiol Immunol 2003; 281: 137–178.
Lebedeva I, Fujita K, Nihrane A, Silver J . Infectious particles derived from Semliki Forest virus vectors encoding murine leukemia virus envelopes. J Virol 1997; 71: 7061–7067.
Pelkmans L, Helenius A . Insider information: what viruses tell us about endocytosis. Curr Opin Cell Biol 2003; 15: 414–422.
Vashishtha M et al. A single point mutation controls the cholesterol dependence of Semliki Forest virus entry and exit. J Cell Biol 1998; 140: 91–99.
Waarts BL, Bittman R, Wilschut J . Sphingolipid and cholesterol dependence of alphavirus membrane fusion. Lack of correlation with lipid raft formation in target liposomes. J Biol Chem 2002; 277: 38141–38147.
Lu YE, Cassese T, Kielian M . The cholesterol requirement for sindbis virus entry and exit and characterization of a spike protein region involved in cholesterol dependence. J Virol 1999; 73: 4272–4278.
Hunziker W, Whitney JA, Mellman I . Brefeldin A and the endocytic pathway. Possible implications for membrane traffic and sorting. FEBS Lett 1992; 307: 93–96.
Kujala P et al. Biogenesis of the Semliki Forest virus RNA replication complex. J Virol 2001; 75: 3873–3884.
Kaariainen L, Ahola T . Functions of alphavirus nonstructural proteins in RNA replication. Prog Nucleic Acid Res Mol Biol 2002; 71: 187–222.
Laakkonen P, Ahola T, Kaariainen L . The effects of palmitoylation on membrane association of Semliki forest virus RNA capping enzyme. J Biol Chem 1996; 271: 28567–28571.
Katze MG, He Y, Gale Jr M . Viruses and interferon: a fight for supremacy. Nat Rev Immunol 2002; 2: 675–687.
Sarkar SN, Sen GC . Novel functions of proteins encoded by viral stress-inducible genes. Pharmacol Ther 2004; 103: 245–259.
Malmgaard L . Induction and regulation of IFNs during viral infections. J Interferon Cytokine Res 2004; 24: 439–454.
Sudhakar A et al. Phosphorylation of serine 51 in initiation factor 2 alpha (eIF2 alpha) promotes complex formation between eIF2 alpha(P) and eIF2B and causes inhibition in the guanine nucleotide exchange activity of eIF2B. Biochemistry 2000; 39: 12929–12938.
Kaufman RJ . Regulation of mRNA translation by protein folding in the endoplasmic reticulum. Trends Biochem Sci 2004; 29: 152–158.
Gorchakov R et al. PKR-dependent and -independent mechanisms are involved in translational shutoff during Sindbis virus infection. J Virol 2004; 78: 8455–8467.
Landis H et al. Human MxA protein confers resistance to Semliki Forest virus and inhibits the amplification of a Semliki Forest virus-based replicon in the absence of viral structural proteins. J Virol 1998; 72: 1516–1522.
Terenzi F et al. The antiviral enzymes PKR and RNase L suppress gene expression from viral and non-viral based vectors. Nucleic Acids Res 1999; 27: 4369–4375.
Matsumoto M, Funami K, Oshiumi H, Seya T . Toll-like receptor 3: a link between toll-like receptor, interferon and viruses. Microbiol Immunol 2004; 48: 147–154.
Edelmann KH et al. Does Toll-like receptor 3 play a biological role in virus infections? Virology 2004; 322: 231–238.
Gao G, Guo X, Goff SP . Inhibition of retroviral RNA production by ZAP, a CCCH-type zinc finger protein. Science 2002; 297: 1703–1706.
Guo X et al. The zinc finger antiviral protein directly binds to specific viral mRNAs through the CCCH zinc finger motifs. J Virol 2004; 78: 12781–12787.
Liu L, Chen G, Ji X, Gao G . ZAP is a CRM1-dependent nucleocytoplasmic shuttling protein. Biochem Biophys Res Commun 2004; 321: 517–523.
Li ML, Stollar V . Alphaviruses and apoptosis. Int Rev Immunol 2004; 23: 7–24.
Fazakerley JK, Boyd A, Mikkola ML, Kaariainen L . A single amino acid change in the nuclear localization sequence of the nsP2 protein affects the neurovirulence of Semliki Forest virus. J Virol 2002; 76: 392–396.
Glasgow GM et al. The Semliki Forest virus vector induces p53-independent apoptosis. J Gen Virol 1998; 79: 2405–2410.
Perri S et al. Replicon vectors derived from Sindbis virus and Semliki forest virus that establish persistent replication in host cells. J Virol 2000; 74: 9802–9807.
Jeromin A et al. A modified Sindbis vector for prolonged gene expression in neurons. J Neurophysiol 2003; 90: 2741–2745.
Choi K et al. Identification of cellular proteins enhancing activities of internal ribosomal entry sites by competition with oligodeoxynucleotides. Nucleic Acids Res 2004; 32: 1308–1317, Print 2004.
Thoma C et al. Enhancement of IRES-mediated translation of the c-myc and BiP mRNAs by the poly(A) tail is independent of intact eIF4G and PABP. Mol Cell 2004; 15: 925–935.
Bayer TS, Smolke CD . Programmable ligand-controlled riboregulators of eukaryotic gene expression. Nat Biotechnol 2005; 23: 337–343.
Moumen A et al. Evidence for a mechanism of recombination during reverse transcription dependent on the structure of the acceptor RNA. J Biol Chem 2003; 278: 15973–15982.
Negroni M, Buc H . Mechanisms of retroviral recombination. Annu Rev Genet 2001; 35: 275–302.
Fayzulin R et al. Sindbis virus with a tricomponent genome. J Virol 2005; 79: 637–643.
Acknowledgements
We thank GJ Towers for careful reading of the manuscript and the AFM and VLM for their financial support.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Piver, E., Collin, C., Diatta, A. et al. Cellular factors influencing Semliki Forest Virus vector biology. Gene Ther 12 (Suppl 1), S111–S117 (2005). https://doi.org/10.1038/sj.gt.3302625
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302625
Keywords
This article is cited by
-
Corpus Callosum: a Favorable Target for rSFV-Mediated Gene Transfer to Rat Brain with Broad and Efficient Expression
Journal of Molecular Neuroscience (2010)
-
Analysis of human immunodeficiency virus type 1 vector cis- and trans-acting elements production by means of Semliki Forest virus
Gene Therapy (2009)