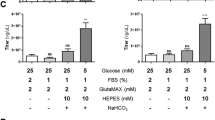
Abstract
Last-generation adenovirus vectors, also called helper-dependent or gutless adenovirus, are very attractive for gene therapy because the associated in vivo immune response is highly reduced compared to first- and second-generation adenovirus vectors, while maintaining high transduction efficiency and tropism. Nowadays, gutless adenovirus is administered in different organs, such as the liver, muscle or the central nervous system achieving high-level and long-term transgene expression in rodents and primates. However, as devoid of all viral coding regions, gutless vectors require viral proteins supplied in trans by a helper virus. To remove contamination by a helper virus from the final preparation, different systems based on the excision of the helper-packaging signal have been generated. Among them, Cre-loxP system is mostly used, although contamination levels still are 0.1–1% too high to be used in clinical trials. Recently developed strategies to avoid/reduce helper contamination were reviewed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Horwitz MS . Adenoviridae and their replication. Virology 1990; 2: 1679–1720.
Danthinne X, Imperiale MJ . Production of first generation adenovirus vectors: a review. Gene Therapy 2000; 7: 1707–1714.
Graham FL, Smiley J, Russell WC, Nairn R . Characteristics of a human cell line transformed by DNA from human adenovirus type 5. J Gen Virol 1977; 36: 59–74.
Fallaux FJ et al. Characterization of 911: a new helper cell line for the titration and propagation of early region 1-deleted adenoviral vectors. Hum Gene Ther 1996; 7: 215–222.
Schiedner G, Hertel S, Kochanek S . Efficient transformation of primary human amniocytes by E1 functions of Ad5: generation of new cell lines for adenoviral vector production. Hum Gene Ther 2000; 11: 2105–2116.
Fallaux FJ et al. New helper cells and matched early region 1-deleted adenovirus vectors prevent generation of replication-competent adenoviruses. Hum Gene Ther 1998; 9: 1909–1917.
Yang Y et al. Cellular immunity to viral antigens limits E1-deleted adenoviruses for gene therapy. Proc Natl Acad Sci USA 1994; 91: 4407–4411.
Amalfitano A et al. Production and characterization of improved adenovirus vectors with the E1, E2b, and E3 genes deleted. J Virol 1998; 72: 926–933.
Armentano D et al. Effect of the E4 region on the persistence of transgene expression from adenovirus vectors. J Virol 1997; 71: 2408–2416.
Alemany R et al. Complementation of helper-dependent adenoviral vectors: size effects and titer fluctuations. J Virol Methods 1997; 68: 147–159.
Bett AJ, Prevec L, Graham FL . Packaging capacity and stability of human adenovirus type 5 vectors. J Virol 1993; 67: 5911–5921.
Ghosh-Choudhury G, Graham FL . Stable transfer of a mouse dihydrofolate reductase gene into a deficient cell line using human adenovirus vector. Biochem Biophys Res Commun 1987; 147: 964–973.
Mitani K, Graham FL, Caskey CT, Kochanek S . Rescue, propagation, and partial purification of a helper virus-dependent adenovirus vector. Proc Natl Acad Sci USA 1995; 92: 3854–3858.
Parks RJ, Graham FL . A helper-dependent system for adenovirus vector production helps define a lower limit for efficient DNA packaging. J Virol 1997; 71: 3293–3298.
Parks R, Evelegh C, Graham F . Use of helper-dependent adenoviral vectors of alternative serotypes permits repeat vector administration. Gene Therapy 1999; 6: 1565–1573.
Schiedner G et al. Variables affecting in vivo performance of high-capacity adenovirus vectors. J Virol 2002; 76: 1600–1609.
Sandig V et al. Optimization of the helper-dependent adenovirus system for production and potency in vivo. Proc Natl Acad Sci USA 2000; 97: 1002–1007.
Palmer D, Ng P . Improved system for helper-dependent adenoviral vector production. Mol Ther 2003; 8: 846–852.
Sato M, Suzuki S, Kubo S, Mitani K . Replication and packaging of helper-dependent adenoviral vectors. Gene Therapy 2002; 9: 472–476.
Soudais C, Skander N, Kremer EJ . Long-term in vivo transduction of neurons throughout the rat CNS using novel helper-dependent CAV-2 vectors. FASEB J 2004; 18: 391–393.
Parks RJ et al. A helper-dependent adenovirus vector system: removal of helper virus by Cre-mediated excision of the viral packaging signal. Proc Natl Acad Sci USA 1996; 93: 13565–13570.
Silver DP, Livingston DM . Self-excising retroviral vectors encoding the Cre recombinase overcome Cre-mediated cellular toxicity. Mol Cell 2001; 8: 233–243.
Zang Y, Schneider R . Adenovirus inhibition of cellular protein synthesis and the specific translation of late viral mRNA. Semin Virol 1993; 4: 233–243.
Ng PG, Graham FL . Helper-dependent adenoviral vectors for gene therapy. In: Templeton NS (ed). Gene and Cell Therapy. Marcel Dekker Inc: New York, 2004 pp 53–70.
Som T, Armstrong KA, Volkert FC, Broach JR . Autoregulation of 2 micron circle gene expression provides a model for maintenance of stable plasmid copy levels. Cell 1988; 52: 27–37.
Ng P et al. Development of a FLP/frt system for generating helper-dependent adenoviral vectors. Mol Ther 2001; 3: 809–815.
Groth AC, Olivares EC, Thyagarajan B, Calos MP . A phage integrase directs efficient site-specific integration in human cells. Proc Natl Acad Sci USA 2000; 97: 5995–6000.
Sargent KL et al. Development of a size-restricted pIX-deleted helper virus for amplification of helper-dependent adenovirus vectors. Gene Therapy 2004; 11: 504–511.
Kubo S, Saeki Y, Chiocca EA, Mitani K . An HSV amplicon-based helper system for helper-dependent adenoviral vectors. Biochem Biophys Res Commun 2003; 307: 826–830.
Cheshenko N, Krougliak N, Eisensmith RC, Krougliak VA . A novel system for the production of fully deleted adenovirus vectors that does not require helper adenovirus. Gene Therapy 2001; 8: 846–854.
Graham FL . Growth of 293 cells in suspension culture. J Gen Virol 1987; 68 (Pt 3): 937–940.
Sakhuja K et al. Optimization of the generation and propagation of gutless adenoviral vectors. Hum Gene Ther 2003; 14: 243–254.
Schagen FH, Ossevoort M, Toes RE, Hoeben RC . Immune responses against adenoviral vectors and their transgene products: a review of strategies for evasion. Crit Rev Oncol Hematol 2004; 50: 51–70.
Worgall S, Wolff G, Falck-Pedersen E, Crystal RG . Innate immune mechanisms dominate elimination of adenoviral vectors following in vivo administration. Hum Gene Ther 1997; 8: 37–44.
Liu Q et al. The role of capsid-endothelial interactions in the innate immune response to adenovirus vectors. Hum Gene Ther 2003; 14: 627–643.
Schnell MA et al. Activation of innate immunity in nonhuman primates following intraportal administration of adenoviral vectors. Mol Ther 2001; 3: 708–722.
Molinier-Frenkel V et al. Immune response to recombinant adenovirus in humans: capsid components from viral input are targets for vector-specific cytotoxic T lymphocytes. J Virol 2000; 74: 7678–7682.
Paul WE, Seder RA . Lymphocyte responses and cytokines. Cell 1994; 76: 241–251.
Kuzmin AI, Finegold MJ, Eisensmith RC . Macrophage depletion increases the safety, efficacy and persistence of adenovirus-mediated gene transfer in vivo. Gene Therapy 1997; 4: 309–316.
Wolff G et al. Enhancement of in vivo adenovirus-mediated gene transfer and expression by prior depletion of tissue macrophages in the target organ. J Virol 1997; 71: 624–629.
Dai Y et al. Cellular and humoral immune responses to adenoviral vectors containing factor IX gene: tolerization of factor IX and vector antigens allows for long-term expression. Proc Natl Acad Sci USA 1995; 92: 1401–1405.
Fang B et al. Gene therapy for hemophilia B: host immunosuppression prolongs the therapeutic effect of adenovirus-mediated factor IX expression. Hum Gene Ther 1995; 6: 1039–1044.
Kaplan JM, Smith AE . Transient immunosuppression with deoxyspergualin improves longevity of transgene expression and ability to readminister adenoviral vector to the mouse lung. Hum Gene Ther 1997; 8: 1095–1104.
Kuriyama S et al. Immunomodulation with FK506 around the time of intravenous re-administration of an adenoviral vector facilitates gene transfer into primed rat liver. Int J Cancer 2000; 85: 839–844.
Otake K, Ennist DL, Harrod K, Trapnell BC . Nonspecific inflammation inhibits adenovirus-mediated pulmonary gene transfer and expression independent of specific acquired immune responses. Hum Gene Ther 1998; 9: 2207–2222.
Zuckerman JB et al. A phase I study of adenovirus-mediated transfer of the human cystic fibrosis transmembrane conductance regulator gene to a lung segment of individuals with cystic fibrosis. Hum Gene Ther 1999; 10: 2973–2985.
Poller W et al. Stabilization of transgene expression by incorporation of E3 region genes into an adenoviral factor IX vector and by transient anti-CD4 treatment of the host. Gene Therapy 1996; 3: 521–530.
Sawchuk SJ et al. Anti-T cell receptor monoclonal antibody prolongs transgene expression following adenovirus-mediated in vivo gene transfer to mouse synovium. Hum Gene Ther 1996; 7: 499–506.
Kay MA et al. Long-term hepatic adenovirus-mediated gene expression in mice following CTLA4Ig administration. Nat Genet 1995; 11: 191–197.
Kay MA et al. Transient immunomodulation with anti-CD40 ligand antibody and CTLA4Ig enhances persistence and secondary adenovirus-mediated gene transfer into mouse liver. Proc Natl Acad Sci USA 1997; 94: 4686–4691.
Stein CS, Pemberton JL, van Rooijen N, Davidson BL . Effects of macrophage depletion and anti-CD40 ligand on transgene expression and redosing with recombinant adenovirus. Gene Therapy 1998; 5: 431–439.
Wilson CB et al. Transient inhibition of CD28 and CD40 ligand interactions prolongs adenovirus-mediated transgene expression in the lung and facilitates expression after secondary vector administration. J Virol 1998; 72: 7542–7550.
DeMatteo RP et al. Long-lasting adenovirus transgene expression in mice through neonatal intrathymic tolerance induction without the use of immunosuppression. J Virol 1997; 71: 5330–5335.
Ilan Y et al. Oral tolerization to adenoviral proteins permits repeated adenovirus-mediated gene therapy in rats with pre-existing immunity to adenoviruses. Hepatology 1998; 27: 1368–1376.
Mastrangeli A et al. ‘Sero-switch’ adenovirus-mediated in vivo gene transfer: circumvention of anti-adenovirus humoral immune defenses against repeat adenovirus vector administration by changing the adenovirus serotype. Hum Gene Ther 1996; 7: 79–87.
Morral N et al. Administration of helper-dependent adenoviral vectors and sequential delivery of different vector serotype for long-term liver-directed gene transfer in baboons. Proc Natl Acad Sci USA 1999; 96: 12816–12821.
Fitzgerald JC et al. A simian replication-defective adenoviral recombinant vaccine to HIV-1 gag. J Immunol 2003; 170: 1416–1422.
Kremer EJ, Boutin S, Chillon M, Danos O . Canine adenovirus vectors: an alternative for adenovirus-mediated gene transfer. J Virol 2000; 74: 505–512.
Chillon M, Lee JH, Fasbender A, Welsh MJ . Adenovirus complexed with polyethylene glycol and cationic lipid is shielded from neutralizing antibodies in vitro. Gene Therapy 1998; 5: 995–1002.
Croyle MA, Chirmule N, Zhang Y, Wilson JM . ‘Stealth’ adenoviruses blunt cell-mediated and humoral immune responses against the virus and allow for significant gene expression upon readministration in the lung. J Virol 2001; 75: 4792–4801.
Croyle MA, Chirmule N, Zhang Y, Wilson JM . PEGylation of E1-deleted adenovirus vectors allows significant gene expression on readministration to liver. Hum Gene Ther 2002; 13: 1887–1900.
Lochmuller H et al. Immunosuppression by FK506 markedly prolongs expression of adenovirus-delivered transgene in skeletal muscles of adult dystrophic [mdx] mice. Biochem Biophys Res Commun 1995; 213: 569–574.
Vilquin JT et al. FK506 immunosuppression to control the immune reactions triggered by first-generation adenovirus-mediated gene transfer. Hum Gene Ther 1995; 6: 1391–1401.
Chen HH et al. DNA from both high-capacity and first-generation adenoviral vectors remains intact in skeletal muscle. Hum Gene Ther 1999; 10: 365–373.
Maione D et al. Prolonged expression and effective readministration of erythropoietin delivered with a fully deleted adenoviral vector. Hum Gene Ther 2000; 11: 859–868.
Morsy MA et al. An adenoviral vector deleted for all viral coding sequences results in enhanced safety and extended expression of a leptin transgene. Proc Natl Acad Sci USA 1998; 95: 7866–7871.
O'Neal WK et al. Toxicity associated with repeated administration of first-generation adenovirus vectors does not occur with a helper-dependent vector. Mol Med 2000; 6: 179–195.
Schiedner G et al. Genomic DNA transfer with a high-capacity adenovirus vector results in improved in vivo gene expression and decreased toxicity. Nat Genet 1998; 18: 180–183.
Roth MD et al. Helper-dependent adenoviral vectors efficiently express transgenes in human dendritic cells but still stimulate antiviral immune responses. J Immunol 2002; 169: 4651–4656.
Muruve DA et al. Helper-dependent adenovirus vectors elicit intact innate but attenuated adaptive host immune responses in vivo. J Virol 2004; 78: 5966–5972.
De Geest BR, Van Linthout SA, Collen D . Humoral immune response in mice against a circulating antigen induced by adenoviral transfer is strictly dependent on expression in antigen-presenting cells. Blood 2003; 101: 2551–2556.
Brown BD et al. Factors influencing therapeutic efficacy and the host immune response to helper-dependent adenoviral gene therapy in hemophilia A mice. J Thromb Haemost 2004; 2: 111–118.
Kremer EJ . CAR chasing: canine adenovirus vectors-all bite and no bark? J Gene Med 2004; 6 (Suppl 1): S139–S151.
Thomas CE et al. Peripheral infection with adenovirus causes unexpected long-term brain inflammation in animals injected intracranially with first-generation, but not with high-capacity, adenovirus vectors: toward realistic long-term neurological gene therapy for chronic diseases. Proc Natl Acad Sci USA 2000; 97: 7482–7487.
Thomas CE et al. Pre-existing antiadenoviral immunity is not a barrier to efficient and stable transduction of the brain, mediated by novel high-capacity adenovirus vectors. Hum Gene Ther 2001; 12: 839–846.
Brunetti-Pierri N et al. Acute toxicity after high-dose systemic injection of helper-dependent adenoviral vectors into nonhuman primates. Hum Gene Ther 2004; 15: 35–46.
Croyle MA et al. PEGylated helper-dependent adenoviral vectors: highly efficient vectors with an enhanced safety profile. Gene Therapy 2005; 12: 579–587.
Mok H, Palmer DJ, Ng P, Barry MA . Evaluation of polyethylene glycol modification of first-generation and helper-dependent adenoviral vectors to reduce innate immune responses. Mol Ther 2005; 11: 66–79.
Harui A et al. Vaccination with helper-dependent adenovirus enhances the generation of transgene-specific CTL. Gene Therapy 2004; 11: 1617–1626.
Chen HH et al. Persistence in muscle of an adenoviral vector that lacks all viral genes. Proc Natl Acad Sci USA 1997; 94: 1645–1650.
Kim IA et al. Potential of adenoviral p53 gene therapy and irradiation for the treatment of malignant gliomas. Int J Oncol 2001; 19: 1041–1047.
Morral N et al. High doses of a helper-dependent adenoviral vector yield supraphysiological levels of alpha1-antitrypsin with negligible toxicity. Hum Gene Ther 1998; 9: 2709–2716.
Ehrhardt A et al. A gene-deleted adenoviral vector results in phenotypic correction of canine hemophilia B without liver toxicity or thrombocytopenia. Blood 2003; 102: 2403–2411.
Reddy PS et al. Sustained human factor VIII expression in hemophilia A mice following systemic delivery of a gutless adenoviral vector. Mol Ther 2002; 5: 63–73.
Belalcazar LM et al. Long-term stable expression of human apolipoprotein A–I mediated by helper-dependent adenovirus gene transfer inhibits atherosclerosis progression and remodels atherosclerotic plaques in a mouse model of familial hypercholesterolemia. Circulation 2003; 107: 2726–2732.
Oka K et al. Long-term stable correction of low-density lipoprotein receptor-deficient mice with a helper-dependent adenoviral vector expressing the very low-density lipoprotein receptor. Circulation 2001; 103: 1274–1281.
Pastore L et al. Helper-dependent adenoviral vector-mediated long-term expression of human apolipoprotein A–I reduces atherosclerosis in apo E-deficient mice. Gene 2004; 327: 153–160.
Mian A et al. Long-term correction of ornithine transcarbamylase deficiency by WPRE-mediated overexpression using a helper-dependent adenovirus. Mol Ther 2004; 10: 492–499.
Kojima H et al. NeuroD-betacellulin gene therapy induces islet neogenesis in the liver and reverses diabetes in mice. Nat Med 2003; 9: 596–603.
Aurisicchio L et al. Liver-specific alpha 2 interferon gene expression results in protection from induced hepatitis. J Virol 2000; 74: 4816–4823.
Fiedler M et al. Helper-dependent adenoviral vector-mediated delivery of woodchuck-specific genes for alpha interferon (IFN-alpha) and IFN-gamma: IFN-alpha but not IFN-gamma reduces woodchuck hepatitis virus replication in chronic infection in vivo. J Virol 2004; 78: 10111–10121.
Pastore L et al. Use of a liver-specific promoter reduces immune response to the transgene in adenoviral vectors. Hum Gene Ther 1999; 10: 1773–1781.
Kass-Eisler A et al. Circumventing the immune response to adenovirus-mediated gene therapy. Gene Therapy 1996; 3: 154–162.
Mack CA et al. Circumvention of anti-adenovirus neutralizing immunity by administration of an adenoviral vector of an alternate serotype. Hum Gene Ther 1997; 8: 99–109.
Roy S, Shirley PS, McClelland A, Kaleko M . Circumvention of immunity to the adenovirus major coat protein hexon. J Virol 1998; 72: 6875–6879.
Smith CA, Woodruff LS, Rooney C, Kitchingman GR . Extensive cross-reactivity of adenovirus-specific cytotoxic T cells. Hum Gene Ther 1998; 9: 1419–1427.
Burcin MM et al. Adenovirus-mediated regulable target gene expression in vivo. Proc Natl Acad Sci USA 1999; 96: 355–360.
Salucci V et al. Tight control of gene expression by a helper-dependent adenovirus vector carrying the rtTA2(s)-M2 tetracycline transactivator and repressor system. Gene Therapy 2002; 9: 1415–1421.
Zerby D et al. In vivo ligand-inducible regulation of gene expression in a gutless adenoviral vector system. Hum Gene Ther 2003; 14: 749–761.
Ehrhardt A, Xu H, Kay MA . Episomal persistence of recombinant adenoviral vector genomes during the cell cycle in vivo. J Virol 2003; 77: 7689–7695.
Bramson JL et al. Helper-dependent adenoviral vectors containing modified fiber for improved transduction of developing and mature muscle cells. Hum Gene Ther 2004; 15: 179–188.
Biermann V et al. Targeting of high-capacity adenoviral vectors. Hum Gene Ther 2001; 12: 1757–1769.
Bulfield G, Siller WG, Wight PA, Moore KJ . X chromosome-linked muscular dystrophy (mdx) in the mouse. Proc Natl Acad Sci USA 1984; 81: 1189–1192.
Dudley RW et al. Sustained improvement of muscle function one year after full-length dystrophin gene transfer into mdx mice by a gutted helper-dependent adenoviral vector. Hum Gene Ther 2004; 15: 145–156.
Gilbert R et al. Prolonged dystrophin expression and functional correction of mdx mouse muscle following gene transfer with a helper-dependent (gutted) adenovirus-encoding murine dystrophin. Hum Mol Genet 2003; 12: 1287–1299.
Bilbao R et al. Comparison of high-capacity and first-generation adenoviral vector gene delivery to murine muscle in utero. Gene Therapy 2005; 12: 39–47.
DelloRusso C et al. Functional correction of adult mdx mouse muscle using gutted adenoviral vectors expressing full-length dystrophin. Proc Natl Acad Sci USA 2002; 99: 12979–12984.
Zou L, Zhou H, Pastore L, Yang K . Prolonged transgene expression mediated by a helper-dependent adenoviral vector (hdAd) in the central nervous system. Mol Ther 2000; 2: 105–113.
Thomas CE et al. Acute direct adenoviral vector cytotoxicity and chronic, but not acute, inflammatory responses correlate with decreased vector-mediated transgene expression in the brain. Mol Ther 2001; 3: 36–46.
Semkova I et al. Autologous transplantation of genetically modified iris pigment epithelial cells: a promising concept for the treatment of age-related macular degeneration and other disorders of the eye. Proc Natl Acad Sci USA 2002; 99: 13090–13095.
Oshima Y et al. Intraocular gutless adenoviral-vectored VEGF stimulates anterior segment but not retinal neovascularization. J Cell Physiol 2004; 199: 399–411.
Fleury S et al. Helper-dependent adenovirus vectors devoid of all viral genes cause less myocardial inflammation compared with first-generation adenovirus vectors. Basic Res Cardiol 2004; 99: 247–256.
Tuettenberg A et al. Early adenoviral gene expression mediates immunosuppression by transduced dendritic cell (DC): implications for immunotherapy using genetically modified DC. J Immunol 2004; 172: 1524–1530.
Wen S, Graf S, Massey PG, Dichek DA . Improved vascular gene transfer with a helper-dependent adenoviral vector. Circulation 2004; 110: 1484–1491.
Koehler DR et al. Protection of Cftr knockout mice from acute lung infection by a helper-dependent adenoviral vector expressing Cftr in airway epithelia. Proc Natl Acad Sci USA 2003; 100: 15364–15369.
Goncalves MA et al. Transfer of the full-length dystrophin-coding sequence into muscle cells by a dual high-capacity hybrid viral vector with site-specific integration ability. J Virol 2005; 79: 3146–3162.
Yant SR et al. Transposition from a gutless adeno-transposon vector stabilizes transgene expression in vivo. Nat Biotechnol 2002; 20: 999–1005.
Kreppel F, Kochanek S . Long-term transgene expression in proliferating cells mediated by episomally maintained high-capacity adenovirus vectors. J Virol 2004; 78: 9–22.
Acknowledgements
We would like to acknowledge Dr Mercè Monfar for critically reading the manuscript. Our work is supported by MCYT-SAF2003-03256, Marató TV3-2002-031632 and Instituto de Salud Carlos III (C03/08). AB has a contract from the Ramon y Cajal Program (Ministerio Educación y Ciencia, Spain), and RA is a recipient of an FI-Generalitat fellowship.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alba, R., Bosch, A. & Chillon, M. Gutless adenovirus: last-generation adenovirus for gene therapy. Gene Ther 12 (Suppl 1), S18–S27 (2005). https://doi.org/10.1038/sj.gt.3302612
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302612
Keywords
This article is cited by
-
CRISPR somatic genome engineering and cancer modeling in the mouse pancreas and liver
Nature Protocols (2022)
-
Intranasal HD-Ad vaccine protects the upper and lower respiratory tracts of hACE2 mice against SARS-CoV-2
Cell & Bioscience (2021)
-
Potential of helper-dependent Adenoviral vectors in CRISPR-cas9-mediated lung gene therapy
Cell & Bioscience (2021)
-
Recombinant adenovirus carrying a core neutralizing epitope of porcine epidemic diarrhea virus and heat-labile enterotoxin B of Escherichia coli as a mucosal vaccine
Archives of Virology (2020)
-
Unravelling Receptor and RGD Motif Dependence of Retargeted Adenoviral Vectors using Advanced Tumor Model Systems
Scientific Reports (2019)