Abstract
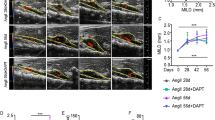
The pathophysiology of abdominal aortic aneurysms (AAA) is considered to be complicated. As matrix degradation contributes to the progression of AAA, the destruction and degradation of elastin fibers caused by an increase in matrix metalloproteinases (MMPs) plays a pivotal role in the development of AAA. Although ets, an essential transcription factor for angiogenesis, regulates MMPs, the role of ets in the development of AAA has not yet been clarified. Thus, we evaluated the role of ets in a rat AAA model using a decoy strategy. Transfection of ODN into AAA was performed by transient aortic perfusion of elastase and by wrapping the AAA in a delivery sheet containing decoy ODN. The inhibitory effect of ets decoy ODN on ets binding activity was confirmed by gel mobility shift assay. MMPs expression was decreased in the aorta transfected with ets decoy ODN as compared to scrambled decoy ODN. Also, ultrasound study demonstrated that elastase-induced aneurismal dilation was significantly suppressed by transfection of ets decoy ODN at 4 weeks after treatment as compared to scrambled decoy ODN. Moreover, the destruction of elastin fibers was inhibited in the aorta transfected with ets decoy ODN, accompanied by a reduction of MMPs expression. An inhibitory effect of decoy ODN on MMP expression was confirmed by ex vivo experiments showing that transfection of decoy ODN into an organ culture of human aorta resulted in significant inhibition of the secretion of both MMP-1 and MMP-9. Here, we demonstrated that ets may play a pivotal role in the progression of AAA through the activation of MMPs in a rat model. Ets might be a potential target to develop pharmacotherapy/gene therapy to treat AAA through the inhibition of MMPs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Wasylyk B, Hahn SL, Giovane A . The Ets family of transcription factors. Eur J Biochem 1993; 15: 7–18.
Ho IC et al. Sequence-specific binding of human Ets-1 to the T cell receptor alpha gene enhancer. Science 1990; 250: 814–818.
Rabault B, Ghysdael J . Calcium-induced phosphorylation of ETS1 inhibits its specific DNA binding activity. J Biol Chem 1994; 269: 28143–28151.
Bhat NK et al. Reciprocal expression of human ETS1 and ETS2 genes during T-cell activation: regulatory role for the protooncogene ETS1. Proc Natl Acad Sci USA 1990; 87: 3723–3727.
Yamamoto H et al. Expression of ets-related transcriptional factor E1AF is associated with tumor progression and over-expression of matrilysin in human gastric cancer. Carcinogenesis 2004; 25: 325–332; (Epub 2003 Nov 06).
Nakamura Y et al. Ets-1 regulates TNF-alpha-induced matrix metalloproteinase-9 and tenascin expression in primary bronchial fibroblasts. J Immunol 2004; 172: 1945–1952.
Vandenbunder B et al. Does the transcription factor c-ets1 take part in the regulation of angiogenesis and tumor invasion? Folia Biol Praha 1994; 40: 301–313.
Nerlov C, Rorth P, Blasi F, Johnsen M . Essential AP-1 and PEA3 binding elements in the human urokinase enhancer display cell type-specific activity. Oncogene 1991; 6: 1583–1592.
Gum R et al. Stimulation of 92-kDa gelatinase B promoter activity by ras is mitogen-activated protein kinase kinase 1-independent and requires multiple transcription factor binding sites including closely spaced PEA3/ets and AP-1 sequences. J Biol Chem 1996; 271: 10672–10680.
Watabe T et al. The Ets-1 and Ets-2 transcription factors activate the promoters for invasion-associated urokinase and collagenase genes in response to epidermal growth factor. Int J Cancer 1998; 77: 128–137.
Saraff K, Babamusta F, Cassis LA, Daugherty A . Aortic dissection precedes formation of aneurysms and atherosclerosis in angiotensin II-infused, apolipoprotein E-deficient mice. Arterioscler Thromb Vasc Biol 2003; 23: 1621–1626.
Liao S et al. Suppression of experimental abdominal aortic aneurysms in the rat by treatment with angiotensin-converting enzyme inhibitors. J Vasc Surg. 2001; 33: 1057–1064.
Newman KM et al. Cellular localization of matrix metalloproteinases in the abdominal aortic aneurysm wall. J Vasc Surg 1994; 20: 814–820.
Thompson RW et al. Production and localization of 92-kilodalton gelatinase in abdominal aortic aneurysms. An elastolytic metalloproteinase expressed by aneurysm-infiltrating macrophages. J Clin Invest 1995; 96: 318–326.
Petrinec D et al. Doxycycline inhibition of aneurysmal degeneration in an elastase-induced rat model of abdominal aortic aneurysm: preservation of aortic elastin associated with suppressed production of 92 kD gelatinase. J Vasc Surg 1996; 23: 336–346.
Knox JB, Sukhova GK, Whittemore AD, Libby P . Evidence for altered balance between matrix metalloproteinases and their inhibitors in human aortic diseases. Circulation 1997; 95: 205–212.
Tamarina NA, McMillan WD, Shively VP, Pearce WH . Expression of matrix metalloproteinases and their inhibitors in aneurysms and normal aorta. Surgery 1997; 122: 264–272.
McMillan WD et al. Size matters: the relationship between MMP-9 expression and aortic diameter. Circulation 1997; 96: 2228–2232.
Treharne GD et al. Marimastat inhibits elastin degradation and matrix metalloproteinase 2 activity in a model of aneurysm disease. Br J Surg 1999; 86: 1053–1058.
Moore G et al. Suppression of experimental abdominal aortic aneurysms by systemic treatment with a hydroxamate-based matrix metalloproteinase inhibitor (RS). J Vasc Surg 1999; 29: 522–532.
Baxter BT et al. Prolonged administration of doxycycline in patients with small asymptomatic abdominal aortic aneurysms: report of a prospective (Phase II) multicenter study. J Vasc Surg 2002; 36: 1–12.
Kadoglou NP, Liapis CD . Matrix metalloproteinases: contribution to pathogenesis, diagnosis, surveillance and treatment of abdominal aortic aneurysms. Curr Med Res Opin 2004; 20: 419–432.
Nakashima H et al. Inhibition of experimental abdominal aortic aneurysm in the rat by use of decoy oligodeoxynucleotides suppressing activity of NFkB and ets transcription factors. Circulation 2004; 109: 132–138.
Chase AJ, Bond M, Crook MF, Newby AC . Role of nuclear factor-kappa B activation in metalloproteinase-1, -3, and -9 secretion by human macrophages in vitro and rabbit foam cells produced in vivo. Arterioscler Thromb Vasc Biol 2002; 22: 765–771.
Zhang J et al. Effects of inflammatory infiltration on the formation of abdominal aortic aneurysm. Zhonghua Wai Ke Za Zhi 1999; 37: 177–179.
Bobryshev YV, Lord RS, Parsson H . Immunophenotypic analysis of the aortic aneurysm wall suggests that vascular dendritic cells are involved in immune responses. Cardiovasc Surg 1998; 6: 240–249.
Longo GM et al. Matrix metalloproteinases 2 and 9 work in concert to produce aortic aneurysms. J Clin Invest 2002; 110: 625–632.
Petersen E, Wagberg F, Angquist KA . Proteolysis of the abdominal aortic aneurysm wall and the association with rupture. Eur J Vasc Endovasc Surg 2002; 23: 153–157.
Wassef M et al. Pathogenesis of abdominal aortic aneurysms: a multidisciplinary research program supported by the National Heart, Lung, and Blood Institute. J Vasc Surg 2001; 34: 730–738.
MacSweeney ST, Powell JT, Greenhalgh RM . Pathogenesis of abdominal aortic aneurysm. Br J Surg 1994; 81: 935–941.
Anidjar S, Kieffer E . Pathogenesis of acquired aneurysms of the abdominal aorta. Ann Vasc Surg 1992; 6: 298–305.
Brophy CM, Reilly JM, Smith GJ, Tilson MD . The role of inflammation in nonspecific abdominal aortic aneurysm disease. Ann Vasc Surg 1991; 5: 229–233.
Freestone T et al. Inflammation and matrix metalloproteinases in the enlarging abdominal aortic aneurysm. Arterioscler Thromb Vasc Biol 1995; 15: 1145–1151.
Carrell TW et al. Stromelysin-1 (matrix metalloproteinase-3) and tissue inhibitor of metalloproteinase-3 are overexpressed in the wall of abdominal aortic aneurysms. Circulation 2002; 105: 477–482.
Wojtowicz-Praga SM, Dickson RB, Hawkins MJ . Matrix metalloproteinase inhibitors. Invest New Drugs 1997; 15: 61–75.
Elmore JR et al. Expression of matrix metalloproteinases and TIMPs in human abdominal aortic aneurysms. Ann Vasc Surg 1998; 12: 221–228.
Crowther M et al. Localization of matrix metalloproteinase 2 within the aneurysmal and normal aortic wall. Br J Surg 2000; 87: 1391–1400.
Newman KM et al. Identification of matrix metalloproteinases 3 (stromelysin-1) and 9 (gelatinase B) in abdominal aortic aneurysm. Arterioscler Thromb 1994; 14: 1315–1320.
Nishimura K et al. Relationships between matrix metalloproteinases and tissue inhibitor of metalloproteinases in the wall of abdominal aortic aneurysms. Int Angiol 2003; 22: 229–238.
Annabi B et al. Differential regulation of matrix metalloproteinase activities in abdominal aortic aneurysms. J Vasc Surg 2002; 35: 539–546.
Yamashita A et al. Enhanced expression of matrix metalloproteinase-9 in abdominal aortic aneurysms. World J Surg 2001; 25: 259–265.
Satta J et al. Chronic inflammation and elastin degradation in abdominal aortic aneurysm disease: an immunohistochemical and electron microscopic study. Eur J Vasc Endovasc Surg 1998; 15: 313–319.
Petersen E, Gineitis A, Wagberg F, Angquist KA . Activity of matrix metalloproteinase-2 and -9 in abdominal aortic aneurysms. Relation to size and rupture. Eur J Vasc Endovasc Surg 2000; 20: 457–461.
Lindholt JS et al. The plasma level of matrix metalloproteinase 9 may predict the natural history of small abdominal aortic aneurysms. A preliminary study. Eur J Vasc Endovasc Surg 2000; 20: 281–285.
MacLeod K, Leprince D, Stehelin D . The ets gene family. Trends Biochem Sci 1992; 17: 251–256.
Laudet V, Niel C, Stehelin D, Duterque-Coquillaud M . Molecular phylogeny of ets gene family. Oncogene 1999; 18: 1351–1359.
Kitange G et al. Ets-1 transcription factor-mediated urokinase type plasminogen activator expression and invasion in glioma cells stimulated by serum and basic fibroblast growth factor. Lab Invest 1999; 79: 407–416.
Goldberg G et al. Secreted proteases. Regulation of their activity and their possible role in metastasis. Ann N Y Acad Sci 1990; 580: 375–384.
Wernert N et al. Stromal expression of c-ets1 transcription factor correlates with tumor invasion. Cancer Res 1994; 54: 5683–5688.
Behrens P et al. Stromal expression of invasion-promoting, matrix-degrading proteases MMP-1 and -9 and the Ets 1 transcription factor in HNPCC carcinomas and sporadic colorectal cancers. Int J Cancer 20031; 107: 183–188.
Kobayashi M et al. Expression of angiogenesis and angiogenic factors in human aortic vascular disease. J Surg Res 2002; 106: 239–245.
Hashiya N et al. In vivo evidence of angiogenesis induced by transcription factor Ets-1: Ets-1 is located upstream of angiogenesis cascade. Circulation 2004; 109: 3035–3041.
Nuchprayoon I et al. An enhancer located between the neutrophil elastase and proteinase 3 promoters is activated by Sp1 and an Ets factor. J Biol Chem 1999; 274: 1085–1091.
Baeuerle PA . IkappaB-NF-kappaB structures: at the interface of inflammation control. Cell 1998; 95: 729–731.
Lawrence DM et al. Rapamycin suppresses experimental aortic aneurysm growth. J Vasc Surg 2004; 40: 334–338.
Aoki M et al. Survival of grafts of genetically modified cardiac myocytes transfected with FITC-labeled oligodeoxynucleotides and β-galactosidase gene in non-infarcted area, but not myocardial infarcted area. Gene Ther 1997; 4: 120–127.
Morishita R et al. In vivo transfection of cis element ‘decoy’ against NFkB binding site prevented myocardial infarction as gene therapy. Nat Med 1997; 3: 894–899.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta C(T)) method. Methods 2001; 25: 402–408.
Neish AS et al. Functional analysis of human vascular cell adhesion molecule 1 promoter. J Exp Med 1992; 176: 1583–1593.
Acknowledgements
This work was partially supported by a Grant-in-Aid from the Organization for Pharmaceutical Safety and Research, a Grant-in-Aid from The Ministry of Public Health and Welfare, a Grant-in-Aid from Japan Promotion of Science and through Special Coordination Funds of the Ministry of Education, Culture, Sports, Science and Technology, the Japanese Government.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Miwa, K., Nakashima, H., Aoki, M. et al. Inhibition of ets, an essential transcription factor for angiogenesis, to prevent the development of abdominal aortic aneurysm in a rat model. Gene Ther 12, 1109–1118 (2005). https://doi.org/10.1038/sj.gt.3302496
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302496
Keywords
This article is cited by
-
Angiopoietin-2 attenuates angiotensin II-induced aortic aneurysm and atherosclerosis in apolipoprotein E-deficient mice
Scientific Reports (2016)
-
The inhibitory effect of chimeric decoy oligodeoxynucleotide against NF-κB and Sp1 in renal interstitial fibrosis
Journal of Molecular Medicine (2013)
-
Ets-1 promotes the progression of cerebral aneurysm by inducing the expression of MCP-1 in vascular smooth muscle cells
Gene Therapy (2010)
-
Prevention of abdominal aortic aneurysms by simultaneous inhibition of NFκB and ets using chimeric decoy oligonucleotides in a rabbit model
Gene Therapy (2006)