Abstract
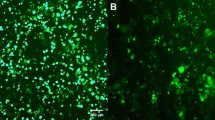
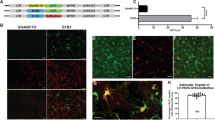
Lentiviral vectors have turned out to be an efficient method for stable gene transfer in vitro and in vivo. Not only do fields of application include cell marking and tracing following transplantation in vivo, but also the stable delivery of biological active proteins for gene therapy. A variety of cells, however, need immediate transplantation after preparation, for example, to prevent cell death, differentiation or de-differentiation. Although these cells are usually washed several times following lentiviral transduction, there may be the risk of viral vector shuttle via transplanted cells resulting in undesired in vivo transduction of recipient cells. We investigated whether infectious lentiviral particles are transmitted via ex vivo lentivirally transduced cells. To this end, we explored potential viral shuttle via ex vivo lentivirally transduced cardiomyocytes in vitro and following transplantation into the brain and peripheral muscle. We demonstrate that, even after extensive washing, infectious viral vector particles can be detected in cell suspensions. Those lentiviral vector particles were able to transduce target cells in transwell experiments. Moreover, transmitted vector particles stably transduced resident cells of the recipient central nervous system and muscle in vivo. Our results of lentiviral vector shuttle via transduced cardiomyocytes are significant for both ex vivo gene therapy and for lentiviral cell tracing, in particular for investigation of stem cell differentiation in transplantation models and co-cultivation systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Naldini L et al. Efficient transfer, integration, and sustained long-term expression of the transgene in adult rat brains injected with a lentiviral vector. Proc Natl Acad Sci USA 1996; 93: 11382–11388.
Zennou V et al. Loss of viral fitness associated with multiple Gag and Gag-Pol processing defects in human immunodeficiency virus type 1 variants selected for resistance to protease inhibitors in vivo. J Virol 1998; 72: 3300–3306.
Fleury S et al. Multiply attenuated, self-inactivating lentiviral vectors efficiently deliver and express genes for extended periods of time in adult rat cardiomyocytes in vivo. Circulation 2003; 107: 2375–2382.
Stitz J et al. A novel lentivirus vector derived from apathogenic simian immunodeficiency virus. Virology 2001; 291: 191–197.
Blomer U et al. Highly efficient and sustained gene transfer in adult neurons with a lentivirus vector. J Virol 1997; 71: 6641–6649.
Kafri T et al. Sustained expression of genes delivered directly into liver and muscle by lentiviral vectors. Nat Genet 1997; 17: 314–317.
Scherr M, Eder M . Gene transfer into hematopoietic stem cells using lentiviral vectors. Curr Gene Ther 2002; 2: 45–55.
Englund U et al. The use of a recombinant lentiviral vector for ex vivo gene transfer into the rat CNS. Neuroreport 2000; 11: 3973–3977.
Condorelli G et al. Cardiomyocytes induce endothelial cells to trans-differentiate into cardiac muscle: implications for myocardium regeneration. Proc Natl Acad Sci USA 2001; 98: 10733–10738.
Ericson C, Wictorin K, Lundberg C . Ex vivo and in vitro studies of transgene expression in rat astrocytes transduced with lentiviral vectors. Exp Neurol 2002; 173: 22–30.
Arsenijevic Y, Weiss S . Insulin-like growth factor-I is a differentiation factor for postmitotic CNS stem cell-derived neuronal precursors: distinct actions from those of brain-derived neurotrophic factor. J Neurosci 1998; 18: 2118–2128.
Arsenijevic Y et al. Isolation of multipotent neural precursors residing in the cortex of the adult human brain. Exp Neurol 2001; 170: 48–62.
Bonci D et al. ‘Advanced’ generation lentiviruses as efficient vectors for cardiomyocyte gene transduction in vitro and in vivo. Gene Therapy 2003; 10: 630–636.
Sakoda T, Kasahara N, Hamamori Y, Kedes L . A high-titer lentiviral production system mediates efficient transduction of differentiated cells including beating cardiac myocytes. J Mol Cell Cardiol 1999; 31: 2037–2047.
Zhao J et al. Lentiviral vectors for delivery of genes into neonatal and adult ventricular cardiac myocytes in vitro and in vivo. Basic Res Cardiol 2002; 97: 348–358.
Blömer U et al. Lentiviral gene transfer and stable transgene expression in cardiomyoctes in vitro and in vivo. Mol Ther 2002; 5: 242.
Conti C, Mastromarino P, Orsi N . Role of membrane phospholipids and glycolipids in cell-to-cell fusion by VSV. Comp Immunol Microbiol Infect Dis 1991; 14: 303–313.
Carneiro FA et al. Membrane fusion induced by vesicular stomatitis virus depends on histidine protonation. J Biol Chem 2003; 278: 13789–13794.
Nagata S, Kurata T, Ueno Y, Chiba J . Vesicular stomatitis virus-mediated cell fusion subsequent to virus adsorption at different pH values. Jpn J Med Sci Biol 1991; 44: 171–180.
Takehara M . Effect of certain inhibitors of glycoprotein synthesis on cell fusion induced by vesicular stomatitis virus. Microbiol Immunol 1979; 23: 167–176.
Haas DL, Case SS, Crooks GM, Kohn DB . Critical factors influencing stable transduction of human CD34(+) cells with HIV-1-derived lentiviral vectors. Mol Ther 2000; 2: 71–80.
Zufferey R et al. Self-inactivating lentivirus vector for safe and efficient in vivo gene delivery. J Virol 1998; 72: 9873–9880.
Zufferey R, Donello JE, Trono D, Hope TJ . Woodchuck hepatitis virus posttranscriptional regulatory element enhances expression of transgenes delivered by retroviral vectors. J Virol 1999; 73: 2886–2892.
Scherr M et al. Quantitative determination of lentiviral vector particle numbers by real-time PCR. Biotechniques 2001; 31: 520 522, 524, passim.
Kofidis T et al. In vitro engineering of heart muscle: artificial myocardial tissue. J Thorac Cardiovasc Surg 2002; 124: 63–69.
Acknowledgements
This work was supported by the HILF program of the Hannover Medical School. Ulrike Blömer was supported by ‘Deutsche Stiftung Querschnittslähmung’ and Ina Gruh by the ‘Graduiertenförderung’ of the Hannover Medical School and Niedersachsen Stiftung/CORTISS. pHR'SIN-CEW, pCMVΔR8.91 and pMD.G were kind gifts of D Trono. We are grateful to M Schweizer, ZMNH Hamburg, for providing time on the confocal microscope and to Patricia Schult-Dietrich, who performed the p24 assay.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Blömer, U., Gruh, I., Witschel, H. et al. Shuttle of lentiviral vectors via transplanted cells in vivo. Gene Ther 12, 67–74 (2005). https://doi.org/10.1038/sj.gt.3302384
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302384
Keywords
This article is cited by
-
Lineage tracing: technology tool for exploring the development, regeneration, and disease of the digestive system
Stem Cell Research & Therapy (2020)
-
Lentiviral vectors and cardiovascular diseases: a genetic tool for manipulating cardiomyocyte differentiation and function
Gene Therapy (2012)
-
The perforin-dependent immunological synapse allows T-cell activation-dependent tumor targeting by MLV vector particles
Gene Therapy (2006)
-
Rapid 1-hour transduction of whole bone marrow leads to long-term repopulation of murine recipients with lentivirus-modified hematopoietic stem cells
Gene Therapy (2006)
-
Tumor-targeted, systemic delivery of therapeutic viral vectors using hitchhiking on antigen-specific T cells
Nature Medicine (2005)