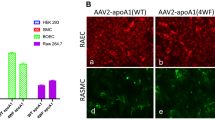
Abstract
Metallic stents coated with a polyurethane emulsion containing plasmid DNA were implanted in rabbit iliac arteries to evaluate transgene delivery and expression in the vessel wall. The expression of the plasmid-encoded marker genes, β-galactosidase, luciferase and green fluorescence protein (GFP), were evaluated at 7 days after implantation. In all cases, plasmid transfer was confined to the vessel wall at the site of stent implantation, plasmid DNA was not observed in vessel segments immediately proximal or distal to the stent and dissemination of plasmid DNA to lung, liver or spleen was not observed. Expression of transgenes occurred only in vessel segments in contact with the stent and analysis of the GFP expression pattern revealed a high frequency of marker protein-positive cells occurring at or near the luminal surface. The extent of transgene expression was dependent upon the quantity of DNA loaded onto the stent and no signal was detected in vessel segments that received polymer-coated stents lacking plasmid DNA. Of significance, colocalization studies identified transgene expression not only in vascular smooth muscle cells but also in macrophages. Hence, polymer-coated stents provide a new capability for transgene delivery to immune cells that are believed to contribute to the development of in-stent restenosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Topol EJ . Coronary-artery stents – gauging, gorging, and gouging. N Engl J Med 1998; 339: 1702–1704.
Fischman DL et al. A randomized comparison of coronary-stent-placement and balloon angioplasty in the treatment of coronary artery disease. N Engl J Med 1994; 331: 469–501.
Serruys P et al. A comparison of balloon expandable-stent implantation with balloon angioplasty in patients with coronary artery disease. N Engl J Med 1994; 331: 489–495.
Kornowski R et al. In-stent restenosis: contributions of inflammatory responses and arterial injury to neointimal hyperplasia. J Am Coll Cardiol 1998; 31: 224–230.
Lincoff A, Furst J, Ellis S, Tuch R et al. Sustained local delivery of dexamethasone by a novel intravascular eluting stent to prevent restenosis in the porcine coronary injury model. J Am Coll Cardiol 1997; 29: 808–816.
Karas S et al. Coronary intimal proliferation after balloon injury and stenting in swine: an animal model of restenosis. J Am Coll Cardiol 1992; 20: 467–474.
Kearney M et al. Histopathology of in-stent restenosis in patients with peripheral artery disease. Circulation 1997; 95: 1998–2002.
Drachman D et al. Neointimal thickening after stent delivery of paclitaxel: change in composition and arrest of growth over six months. J Am Coll Cardiol 2000; 36: 2325–2332.
Farb A et al. Pathological analysis of local delivery of paclitaxel via a polymer-coated stent. Circulation 2001; 104: 473–479.
Suzuki T et al. Stent-based delivery of sirolimus reduces neointimal formation in a porcine coronary model. Circulation 2001; 104: 1188–1193.
Sousa J et al. Sustained suppression of neointimal proliferation by sirolimus-eluting stents: one-year angiographic and intravascular ultrasound follow-up. Circulation 2001; 104: 2007–2011.
Sousa J et al. Lack of neointimal proliferation after implantation of sirolimus-coated stents in human coronary arteries: a quantitative coronary angiography and three-dimensional intravascular ultrasound study. Circulation 2001; 103: 192–195.
Sata M et al. Hematopoietic stem cells differentiate into vascular cells that participate in the pathogenesis of atherosclerosis. Nat Med 2002; 8: 403–409.
Klugherz BD et al. Gene delivery from a DNA controlled-release stent in porcine coronary arteries. Nat Biotechnol 2000; 18: 1181–1184.
Horvath C et al. Targeting CCR2 or CD18 inhibits experimental in-stent restenosis in primates: inhibitory potential depends on type of injury and leukocytes targeted. Circ Res 2002; 90: 488–494.
Feldman LJ et al. Interleukin-10 inhibits intimal hyperplasia after angioplasty or stent implantation in hypercholesterolemic rabbits. Circulation 2000; 101: 908–916.
Kollum M et al. Apoptosis after stent implantation compared with balloon angioplasty in rabbits. Role of macrophage. Arterioscler Thromb Vasc Biol 1997; 17: 2382–2388.
Rogers C, Edelman ER, Simon DI . A mAb to the beta2-leukocyte integrin Mac-1 (CD11b/CD18) reduces intimal thickening after angioplasty or stent implantation in rabbits. Proc Natl Acad Sci USA 1998; 95: 10134–10139.
Van der Giessen W et al. Marked inflammatory sequelae to implantation of biodegradable and nonbiodegradable polymers in porcine coronary arteries. Circulation 1996; 94: 1690–1697.
De Scheerder I et al. Biocompatibility of polymer-coated oversized metallic stents implanted in normal porcine coronary arteries. Atherosclerosis 1995; 114: 105–114.
Smith RC, Walsh K . Gene therapy for restenosis. Curr Cardiol Rep 2000; 2: 13–23.
Smith R, Walsh K . Local gene delivery to the vessel wall. Acta Physiol Scand 2001; 173: 93–102.
Sata M et al. Fas ligand gene transfer to the vessel wall inhibits neointima formation and overrides the adenovirus-mediated T cell response. Proc Natl Acad Sci USA 1998; 95: 1213–1217.
Luo Z et al. Adenovirus-mediated delivery of Fas ligand inhibits intimal hyperplasia after balloon injury in immunologically primed animals. Circulation 1999; 99: 1776–1779.
Newman KD et al. Adenovirus-mediated gene transfer into normal rabbit arteries results in prolonged vascular cell activation, inflammation, and neointimal hyperplasia. J Clin Invest 1995; 96: 2955–2965.
Palasis M, Luo Z, Barry JJ, Walsh K . Analysis of adenoviral transport mechanisms in the vessel wall and optimization of gene transfer using local devices. Hum Gene Ther 2000; 11: 237–246.
Simari RD et al. Regulation of cellular proliferation and intimal formation following balloon injury in atherosclerotic rabbit arteries. J Clin Invest 1996; 98: 225–235.
Marx SO, Jayaraman T, Go LO, Marks AR . Rapamycin-FKBP inhibits cell cycle regulators of proliferation in vascular smooth muscle cells. Circ Res 1995; 76: 412–417.
Poon M et al. Rapamycin inhibits vascular smooth muscle cell migration. J Clin Invest 1996; 98: 2277–2283.
Lim CS et al. Direct in vivo gene transfer into the coronary artery and peripheral vasculature of the intact dog. Circulation 1991; 83: 2007–2011.
Acknowledgements
This study was supported by National Institutes of Health Grants AR-40197, AG-15052, AG-17241, HD-23681 and HL-66957 to KW.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takahashi, A., Palmer-Opolski, M., Smith, R. et al. Transgene delivery of plasmid DNA to smooth muscle cells and macrophages from a biostable polymer-coated stent. Gene Ther 10, 1471–1478 (2003). https://doi.org/10.1038/sj.gt.3302010
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302010
Keywords
This article is cited by
-
Gene delivery technologies for cardiac applications
Gene Therapy (2012)
-
Quantitation of plasmid DNA deposited on gold particles for particle-mediated epidermal delivery using ICP-MS
Analytical and Bioanalytical Chemistry (2010)
-
Cardiovascular gene therapy: current status and therapeutic potential
British Journal of Pharmacology (2007)