Abstract
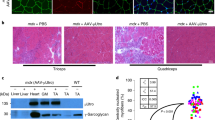
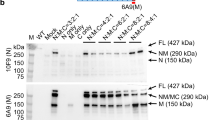
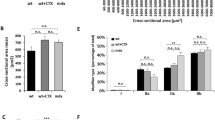
Utrophin is highly homologous and structurally similar to dystrophin, and in gene delivery experiments in mdx mice was able to functionally replace dystrophin. We performed mini-utrophin gene transfer in Golden Retriever dogs with canine muscular dystrophy (CXMD). Unlike the mouse model, the clinicopathological phenotype of CXMD is similar to that of Duchenne muscular dystrophy (DMD). We injected an adenoviral vector expressing a synthetic utrophin into tibialis anterior muscles of newborn dogs affected with CXMD and examined transgene expression by RNA and protein analysis at 10, 30 and 60 days postinjection in cyclosporin-treated and -untreated animals. Immunosuppression by cyclosporin was required to mitigate the immune response to viral and transgene antigens. RT-PCR analysis showed the presence of the exogenous transcript in the muscle of cyclosporin-treated and -untreated animals. The transgenic utrophin was efficiently expressed at the extrajunctional membrane in immunosuppressed dogs and this expression was stable for at least 60 days. We found reduced fibrosis and increased expression of dystrophin-associated proteins (DAPs) in association with muscle areas expressing the utrophin minigene, indicating that mini-utrophin can functionally compensate for lack of dystrophin in injected muscles. For this reason, utrophin transfer to dystrophin-deficient muscle appears as a promising therapeutic approach to DMD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ohtsuka Y et al. Dystrophin acts as a transplantation rejection antigen in dystrophin-deficient mice: implication for gene therapy. J Immunol 1998; 160: 4635–4640.
Ferrer A, Wells KE, Wells DJ . Immune responses to dystrophin: implications for gene therapy of Duchenne muscular dystrophy. Gene Ther 2000; 7: 1439–1446.
Love DR et al. An autosomal transcript in skeletal muscle with homology to dystrophin. Nature 1989; 339: 55–58.
Blake D, Tinsley JM, Davies KE . Utrophin: a structural and functional comparison to dystrophin. Brain Pathol 1996; 6: 37–47.
Khurana TS, Hoffmann EP, Kunkel LM . Identification of chromosome 6-encoded dystrophin-related protein. J Biol Chem 1990; 265: 16717–16720.
Nguyen TM et al. Localization of the DMDL gene-encoded dystrophin-related protein using a panel of nineteen monoclonal antibodies: presence at neuromuscular junctions, in the sarcolemma of dystrophic skeletal muscle, in vascular and other smooth muscles, and in proliferating brain cell lines. J Cell Biol 1991; 115: 1695–1700.
Helliwell TR, Nguyen TN, Morris GE, Davies KE . The dystrophin-related protein, utrophin, is expressed on the sarcolemma of regenerating human skeletal muscle fibers in dystrophies and inflammatory myopathies. Neuromuscul Disord 1992; 2: 177–184.
Karpati G et al. Localization and quantitation of the chromosome 6-encoded dystrophin-related protein in normal and pathological human muscle. J Neuropathol Exp Neurol 1993; 52: 119–128.
Ebihara S et al. Differential effects of dystrophin and utrophin gene transfer in immunocompetent muscular dystrophy (mdx) mice. Physiol Genomics 2000; 8: 133–144.
Tinsley JM et al. Amelioration of the dystrophic phenotype of mdx mice using a truncated utrophin transgene. Nature 1996; 384: 349–353.
Rafael JA et al. Skeletal muscle specific expression of a utrophin transgene rescues utrophin–dystrophin deficient mice. Nat Genet 1998; 19: 79–82.
Ohlendieck K et al. Duchenne muscular dystrophy: deficiency of dystrophin-associated proteins in the sarcolemma. Neurology 1993; 43: 795–800.
Yoshida M, Ozawa E . Glycoprotein complex anchoring dystrophin to sarcolemma. J Biochem 1990; 108: 748–752.
Ervasti JM, Campbell KP . A role for the dystrophin-glycoprotein complex as a transmembrane linker between laminin and actin. J Cell Biol 1993; 122: 809–823.
Gaschen FP et al. Dystrophin deficiency causes lethal muscle hypertrophy in cats. J Neurol Sci 1992; 110: 149–159.
Cooper BJ et al. The homologue of the Duchenne locus is defective in X-linked muscular dystrophy of dogs. Nature 1988; 334: 154–156.
Bulfield G, Siller WG, White PAL, Moore KJ . X chromosome-linked muscular dystrophy (mdx) in the mouse. Proc Natl Acad Sci USA 1984; 81: 1189–1192.
Lefaucheur JP, Pastoret C, Sebille A . Phenotype of dystrophinopathy in old mdx mice. Anat Rec 1995; 242: 70–76.
Valentine BA et al. Canine X-linked muscular dystrophy. An animal model of Duchenne muscular dystrophy: clinical studies. J Neurol Sci 1988; 88: 69–81.
Valentine BA et al. Canine X-linked muscular dystrophy as an animal model of Duchenne muscular dystrophy: a review. Am J Med Genet 1992; 42: 352–356.
Cozzi F et al. Development of muscle pathology in canine X-linked muscular dystrophy II. Quantitative characterization of histopathological progression during postnatal skeletal muscle development. Acta Neuropathol 2001; 101: 469–478.
Lanfossi M et al. Development of muscle pathology in canine X-linked muscular dystrophy I. Delayed postnatal maturation of affected and normal muscle as revealed by myosin isoform analysis and utrophin expression. Acta Neuropathol 1999; 97: 127–138.
Goldspink G, Fernandes K, Williams PE, Wells DJ . Age-related changes in collagen gene expression in the muscles of mdx dystrophic and normal mice. Neuromuscul Disord 1994; 4: 183–191.
Pearce M et al. The utrophin and dystrophin genes share similarities in genomic structure. Hum Mol Genet 1993; 2: 1765–1772.
Neuman S et al. The dystrophin/utrophin homologues in Drosophila and in sea urchin. Gene 2001; 263: 17–29.
Amann KJ, Guo AW-X, Ervasti JM . Utrophin lacks the rod domain actin binding activity of dystrophin. J Biol Chem 1999; 274: 35375–35380.
Rybakova IN et al. Utrophin binds laterally along actin filaments and can couple costameric actin with sarcolemma when overexpressed in dystrophin-deficient muscle. Mol Biol Cell 2002; 13: 1512–1521.
Zhao N, Liu DP, Liang CC . Hot topics in adeno-associated virus as gene transfer vector. Mol Biotechnol 2001; 19: 229–237.
Xiao X, Li J, Samulski RJ . Efficient long-term gene transfer into muscle tissue of immunocompetent mice by adeno-associated virus vector. J Virol 1996; 70: 8098–8108.
Wang B, Li J, Xiao X . Adeno-associated virus vector carrying human minidystrophin genes effectively ameliorates muscular dystrophy in mdx mouse model. Proc Natl Acad Sci USA 2000; 97: 13714–13719.
Tripathy SK, Black HB, Goldwasser E, Leiden JM . Immune responses to transgene-encoded proteins limit the stability of gene expression after injection of replication-defective adenovirus vectors. Nat Med 1996; 2: 545–550.
Lochmuller H et al. Transient immunosuppression by FK506 permits a sustained high-level dystrophin expression after adenovirus-mediated dystrophin minigene transfer to skeletal muscles of adult dystrophic (mdx) mice. Gene Ther 1996; 3: 706–716.
Gilbert R et al. Efficient utrophin expression following adenovirus gene transfer in dystrophic muscle. Biochem Biophys Res Comm 1998; 242: 244–247.
Acknowledgements
The authors thank Drs Rocco Lombardo and Gaia Luvoni for performing muscle biopsies and dog husbandry, and Don Ward for help with the English.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cerletti, M., Negri, T., Cozzi, F. et al. Dystrophic phenotype of canine X-linked muscular dystrophy is mitigated by adenovirus-mediated utrophin gene transfer. Gene Ther 10, 750–757 (2003). https://doi.org/10.1038/sj.gt.3301941
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301941
Keywords
This article is cited by
-
Endogenous bioluminescent reporters reveal a sustained increase in utrophin gene expression upon EZH2 and ERK1/2 inhibition
Communications Biology (2023)
-
Therapeutic potential of highly functional codon-optimized microutrophin for muscle-specific expression
Scientific Reports (2022)
-
Duchenne muscular dystrophy cell culture models created by CRISPR/Cas9 gene editing and their application in drug screening
Scientific Reports (2021)
-
Supplementation with a selective amino acid formula ameliorates muscular dystrophy in mdx mice
Scientific Reports (2018)
-
Whole genome sequencing reveals a 7 base-pair deletion in DMD exon 42 in a dog with muscular dystrophy
Mammalian Genome (2017)