Abstract
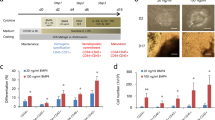
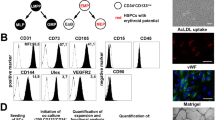
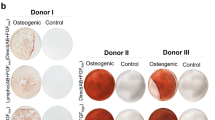
Bone marrow stromal cells (MSCs) are pluripotent cells capable of differentiation into several tissue types. This present study was performed to determine their functional neoangiogenic potential in vivo. Whole bone marrow was harvested from C57Bl/6 mice, and the adherent cellular fraction was culture expanded for 14 doublings. These MSCs were resuspended in Matrigel and their angiogenic effect assessed in isogenic recipients. At 2 weeks postimplantation, the mean vascular density in Matrigel plugs containing 2 × 106 MSCs/ml was 41±5.0 blood vessels (BVs)/mm2 compared to 0.5±0.7 for empty Matrigel (P<0.001). In comparison, Matrigel plugs containing either recombinant murine VEGF 165 at 50 ng/ml or bovine bFGF at 1000 ng/ml generated 21±5 and 11±2.0 BV/mm2, respectively. Arteriogenesis was observed only in the MSC-containing implants. With the use of LacZ retroviral labeling of ex vivo expanded MSCs, we show that ∼10% of LacZ+MSCs differentiated into CD31+ and VEGF+ endothelial cells. More than 99% of the neoangiogenic phenomena arose from recruitment of host-derived LacZnull vascular structures. Neutralizing anti-VEGF antibodies inhibited the MSC-initiated angiogenic response in vivo by 85% (P<0.001). In conclusion, MSCs have the ability to effectively recruit and participate in angiogenesis and arteriogenesis de novo and VEGF plays a central role in the observed host-derived angiogenic response. We propose that ex vivo expanded autologous MSCs may serve as cell therapy to promote therapeutic angiogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Ausprunk DH, Folkman J . Migration and proliferation of endothelial cells in preformed and newly formed blood vessels during tumor angiogenesis. Microvasc Res 1977; 14: 53–65.
Flamme I, Risau W . Induction of vasculogenesis and hematopoiesis in vitro. Development 1992; 116: 435–439.
Asahara T et al. Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circul Res 1999; 85: 221–228.
Reyes M et al. Purification and ex vivo expansion of postnatal human marrow mesodermal progenitor cells. Blood 2001 1; 98: 2615–2625.
Prockop DJ . Marrow stromal cells as stem cells for nonhematopoietic tissues. Science 1997; 276: 71–74 [Review, 32 refs].
Piersma AH, Ploemacher RE, Brockbank KG . Transplantation of bone marrow fibroblastoid stromal cells in mice via the intravenous route. Br J Haematol 1983; 54: 285–290.
Makino S et al. Cardiomyocytes can be generated from marrow stromal cells in vitro. J Clin Invest 1999; 103: 697–705.
Seshi B, Kumar S, Sellers D . Human bone marrow stromal cell: Coexpression of markers specific for multiple mesenchymal cell lineages. Blood Cells Mol Dis 2000; 26: 234–246.
Deans RJ, Moseley AB . Mesenchymal stem cells: biology and potential clinical uses. Exp Hematol 2000; 28: 875–884 [Review, 92 refs].
Jaalouk DE et al. Glucocorticoid-inducible retrovector for regulated transgene expression in genetically engineered bone marrow stromal cells. Hum Gene Ther 2000; 11: 1837–1849.
Eliopoulos N et al. Human cytidine deaminase as an ex vivo drug selectable marker in gene-modified primary bone marrow stromal cells. Gene Ther 2002; 9: 452–462.
Ory DS, Neugeboren BA, Mulligan RC . A stable human-derived packaging cell line for production of high titer retrovirus/vesicular stomatitis virus G pseudotypes. Proc Nat Acad Sci USA 1996; 93: 11400–11406.
Passaniti A et al. A simple quantitative method for assessing angiogenesis and antiangiogenic agents using reconstituted basement membrane, heparin, and fibroblast growth factor. Lab Invest 1992; 67: 519–528.
Brown G . An Introduction to Histotechnology. Appleton-Century-Crofts: NY, 1978.
Galmiche MC et al. Stromal cells from human long-term marrow cultures are mesenchymal cells that differentiate following a vascular smooth muscle differentiation pathway. Blood 1993; 82: 66–76.
Li J, Sensebe L, Herve P, Charbord P . Nontransformed colony-derived stromal cell lines from normal human marrows. II. Phenotypic characterization and differentiation pathway. Exp Hematol 1995; 23: 133–141.
Koc ON et al. Culture-expanded autologous human mesenchymal stem cells (MSCs) circulate in blood and retain proliferative capacity following i.v. infusion into breast cancer patients. Blood 1998; 92: 274a.
Horwitz EM et al. Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nat Med 1999; 5: 309–313.
Azizi SA et al. Engraftment and migration of human bone marrow stromal cells implanted in the brains of albino rats – similarities to astrocyte grafts. Proc Nat Acad Sci USA 1998; 95: 3908–3913.
Wang JS et al. Marrow stromal cells for cellular cardiomyoplasty: Feasibility and potential clinical advantages. J Thor Cardiovas Surg 2000; 120: 999–1006.
Reyes M et al. Purification and ex vivo expansion of postnatal human marrow mesodermal progenitor cells. Blood 2001; 98: 2615–2625.
Takahashi T et al. Ischemia- and cytokine-induced mobilization of bone marrow-derived endothelial progenitor cells for neovascularization. Nat Med 1999; 5: 434–438.
Quirici N et al. Differentiation and expansion of endothelial cells from human bone marrow CD133(+) cells. Br J Haematol 2001; 115: 186–194.
Murohara T et al. Transplanted cord blood-derived endothelial precursor cells augment postnatal neovascularization. J Clin Invest 2000; 105: 1527–1536.
Kocher AA et al. Neovascularization of ischemic myocardium by human bone marrow derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med 2001; 7; 430–436.
Phinney DG, Kopen G, Isaacson RL, Prockop DJ . Plastic adherent stromal cells from the bone marrow of commonly used strains of inbred mice: variations in yield, growth, and differentiation. J Cell Biochem 1999; 72: 570–585.
Tremain N et al. MicroSAGE analysis of 2,353 expressed genes in a single cell-derived colony of undifferentiated human mesenchymal stem cells reveals mRNAs of multiple cell lineages. Stem Cells 1902; 19: 408–418.
Reyes M et al. Origin of endothelian progenitors in human postnatal bone marrow. J Clin Invest 2002; 109: 337–346.
Friedenstein AJ, Gorskaja JF, Kulagina NN . Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp Hematol 1976; 4: 267–274.
Pittenger MF et al. Multilineage potential of adult human mesenchymal stem cells. Science 1999; 284: 143–147.
Waltenberger J, Mayr U, Pentz S, Hombach V . Functional upregulation of the vascular endothelial growth factor receptor KDR by hypoxia [see comments]. Circulation 1996; 94: 1647–1654.
Montrucchio G, Lupia E, Battaglia E et al. Platelet-activating factor enhances vascular endothelial growth factor-induced endothelial cell motility and neoangiogenesis in a murine matrigel model. Arterioscl, Thromb Vasc Biol 2000; 20: 80–88.
Schwarz ER et al. Evaluation of the effects of intramyocardial injection of DNA expressing vascular endothelial growth factor (VEGF) in a myocardial infarction model in the rat–angiogenesis and angioma formation. J Am Coll Cardiol 2000; 35: 1323–1330.
Biroccio A et al. Bcl-2 overexpression and hypoxia synergistically act to modulate vascular endothelial growth factor expression and in vivo angiogenesis in a breast carcinoma line. FASEB J 2000; 14: 652–660.
Acknowledgements
This work was supported by the Canadian Institutes of Health Research Grant MOP-15017. JG is recipient of a CIHR Clinician-Scientist Award and NE is supported by a Leukemia Research Fund of Canada Fellowship.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Al-Khaldi, A., Eliopoulos, N., Martineau, D. et al. Postnatal bone marrow stromal cells elicit a potent VEGF-dependent neoangiogenic response in vivo. Gene Ther 10, 621–629 (2003). https://doi.org/10.1038/sj.gt.3301934
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301934
Keywords
This article is cited by
-
In vivo delivery of VEGF RNA and protein to increase osteogenesis and intraosseous angiogenesis
Scientific Reports (2019)
-
Mesenchymal stem cells in suppression or progression of hematologic malignancy: current status and challenges
Leukemia (2019)
-
Value of pre-operative CTX serum levels in the prediction of medication-related osteonecrosis of the jaw (MRONJ): a retrospective clinical study
EPMA Journal (2019)
-
Tumour-associated mesenchymal stem/stromal cells: emerging therapeutic targets
Nature Reviews Drug Discovery (2017)
-
In-vivo evaluation of subcutaneously implanted cell-loaded apatite microcarriers for osteogenic potency
Journal of Materials Science: Materials in Medicine (2017)