Abstract
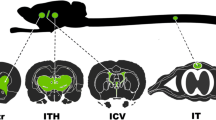
Baculoviruses have recently been shown to be effective gene transfer vectors in mammalian cells. However, very little information is available about their target cell tropism in the central nervous system. We studied transduction efficiency, tropism and biodistribution of baculoviruses after local delivery to rat brain and compared their properties to adenoviruses. It was found that baculoviruses specifically transduced cuboid epithelium of the choroid plexus in ventricles and that the transduction efficiency was as high as 76±14%, whereas adenoviruses showed preference to corpus callosum glial cells and ventricular ependymal lining. Only a modest microglia response was seen after the baculovirus transduction whereas the adenovirus gene transfer led to a strong microglia response. Sensitive nested RT-PCR revealed transgene expression in the hindbrain and in ectopic organs including spleen, heart and lung, which indicates that some escape of both vectors occurs to ectopic organs after local gene transfer to the brain. We conclude that both baculovirus and adenovirus vectors can be used for local intracerebral gene therapy. The knowledge of the cell type specificity of the vectors may offer a possibility to achieve targeted gene delivery to distinct brain areas. Baculoviruses seem to be especially useful for the targeting of choroid plexus cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hofmann C et al. Efficient gene transfer into human hepatocytes by baculovirus vectors Proc Natl Acad Sci USA 1995 92: 10 099–10 103
Airenne KJ et al. Baculovirus-mediated periadventitial gene transfer to rabbit carotid artery Gene Ther 2000 7: 1499–1504
Sarkis C et al. Efficient transduction of neural cells in vitro and in vivo by a baculovirus-derived vector Proc Natl Acad Sci USA 2000 97: 14 638–14 643
Pieroni L, Maione D, La Monica N . In vivo gene transfer in mouse skeletal muscle mediated by baculovirus vectors Hum Gene Ther 2001 12: 871–881
Wang P, Hammer DA, Granados RR . Binding and fusion of Autographa californica nucleopolyhedrovirus to cultured insect cells J Gen Virol 1997 78: (Pt 12) 3081–3089
van Loo ND et al. Baculovirus infection of nondividing mammalian cells: mechanisms of entry and nuclear transport of capsids J Virol 2001 75: 961–970
Boyce FM, Bucher NL . Baculovirus-mediated gene transfer into mammalian cells Proc Natl Acad Sci USA 1996 93: 2348–2352
Gröner A . Specificity and safety of baculoviruses In: Granados RR et al, (eds) The Biology of Baculoviruses Vol. 1: CRC Press: Boca Raton 1986 pp 177–202
Burges HD, Croizier G, Huber J . A review of safety tests on baculoviruses Entomophaga 1980 25: 329–340
Ignoffo CM, Huang HT, Shapiro M, Woodard G . Insuspectibility of the rhesus monkey, macaca mulatta, to an insect virus, baculovirus heliothis Environ Entomol 1975 4: 569–573
Driesse MJ et al. Distribution of recombinant adenovirus in the cerebrospinal fluid of nonhuman primates Hum Gene Ther 1999 10: 2347–2354
Ooboshi H et al. Adenovirus-mediated gene transfer in vivo to cerebral blood vessels and perivascular tissue Circ Res 1995 77: 7–13
Bajocchi G, Feldman SH, Crystal RG, Mastrangeli A . Direct in vivo gene transfer to ependymal cells in the central nervous system using recombinant adenovirus vectors Nat Genet 1993 3: 229–234
Hofmann C, Lehnert W, Strauss M . The baculovirus vector system for gene delivery into hepatocytes Gene Ther Mol Biol 1998 1: 231–239
Huser A, Rudolph M, Hofmann C . Incorporation of decay-accelerating factor into the baculovirus envelope generates complement-resistant gene transfer vectors Nat Biotechnol 2001 19: 451–455
Condreay JP, Witherspoon SM, Clay WC, Kost TA . Transient and stable gene expression in mammalian cells transduced with a recombinant baculovirus vector Proc Natl Acad Sci USA 1999 96: 127–132
Ma L et al. Baculovirus-mediated gene transfer into pancreatic islet cells Diabetes 2000 49: 1986–1991
Sandmair AM et al. Low efficacy of gene therapy for rat BT4C malignant glioma using intra-tumoural transduction with thymidine kinase retrovirus packaging cell injections and ganciclovir treatment Acta Neurochir (Wien) 1999 141: 867–872
Hakumaki JM et al. 1H MRS detects polyunsaturated fatty acid accumulation during gene therapy of glioma: implica-tions for the in vivo detection of apoptosis Nat Med 1999 5: 1323–1327
Yagi T et al. Rescue of ischemic brain injury by adenoviral gene transfer of glial cell line-derived neurotrophic factor after transient global ischemia in gerbils Brain Res 2000 885: 273–282
Thomas CE et al. Acute direct adenoviral vector cytotoxicity and chronic, but not acute, inflammatory responses correlate with decreased vector-mediated transgene expression in the brain Mol Ther 2001 3: 36–46
Ohmoto Y et al. Variation in the immune response to adenoviral vectors in the brain: influence of mouse strain, environmental conditions and priming Gene Ther 1999 6: 471–481
Lohr F et al. Systemic vector leakage and transgene expression by intratumorally injected recombinant adenovirus vectors Clin Cancer Res 2001 7: 3625–3628
Bramson JL, Hitt M, Gauldie J, Graham FL . Pre-existing immunity to adenovirus does not prevent tumor regression following intratumoral administration of a vector expressing IL-12 but inhibits virus dissemination Gene Ther 1997 4: 1069–1076
Laitinen M et al. Gene transfer into the carotid artery using an adventitial collar: comparison of the effectiveness of the plasmid–liposome complexes, retroviruses, pseudotyped retroviruses and adenoviruses Hum Gene Ther 1997 8: 1645–1650
Laitinen M et al. Adenovirus-mediated gene transfer to lower limb artery of patients with chronic critical leg ischemia Hum Gene Ther 1998 9: 1481–1486
Bjerkvig R, Laerum OD, Mella O . Glioma cell interactions with fetal rat brain aggregates in vitro and with brain tissue in vivo Cancer Res 1986 46: 4071–4079
Acknowledgements
This study was supported by grants from the Finnish Academy, Sigrid Juselius Foundation and Ark Therapeutics, Ltd. We would like to thank Ms Seija Sahrio, Ms Mervi Nieminen and Mr Tommi Heikura for excellent technical assistance.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lehtolainen, P., Tyynelä, K., Kannasto, J. et al. Baculoviruses exhibit restricted cell type specificity in rat brain: a comparison of baculovirus- and adenovirus-mediated intracerebral gene transfer in vivo. Gene Ther 9, 1693–1699 (2002). https://doi.org/10.1038/sj.gt.3301854
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301854
Keywords
This article is cited by
-
Baculovirus: an Insect-derived Vector for Diverse Gene Transfer Applications
Molecular Therapy (2013)
-
Screening of Complement Inhibitors: Shielded Baculoviruses Increase the Safety and Efficacy of Gene Delivery
Molecular Therapy (2010)
-
Ependymal cells: biology and pathology
Acta Neuropathologica (2010)
-
High Mobility Group Box2 Promoter-controlled Suicide Gene Expression Enables Targeted Glioblastoma Treatment
Molecular Therapy (2009)
-
(Strept)avidin-displaying lentiviruses as versatile tools for targeting and dual imaging of gene delivery
Gene Therapy (2009)