Abstract
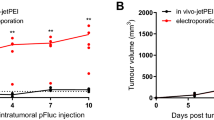
In vivo electroporation is an efficient means of increasing plasmid DNA delivery to normal tissues, such as skin and muscle, as well as directly to tumors. In the experiments described here, plasmid DNA was delivered by in vivo electroporation to B16 mouse melanomas using two very different pulsing protocols. Reporter expression increased 21- or 42-fold, respectively with electroporation over injection alone. The growth of experimental melanomas with an approximate diameter of 4 mm on the day of treatment was monitored after electroporation delivery of reporter plasmid DNA. Remarkably, short-term complete regressions using one of these pulsing protocols occurred in up to 100% of mice. These regressions continued long term in up to 83% of animals. 70% of these mice were resistant to challenge with B16 melanoma cells. Histological analysis revealed large numbers of apoptotic cells 24 h after treatment. This antitumor effect did not require therapeutic cDNA expression or eukaryotic sequences.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Jaroszeski MJ, Gilbert R, Heller R . Electrochemotherapy: an emerging drug delivery method for the treatment of cancer Adv Drug Del Rev 1997 26: 185–197
Heller R, Gilbert R, Jaroszeski M . Clinical applications of electrochemotherapy Adv Drug Del Rev 1999 35: 119–129
Heller L, Lucas ML . Delivery of plasmid DNA by in vivo electroporation Gene Ther Mol Biol 2000 5: 50–55
Titomirov AV, Sukharev S, Kistanova E . In vivo electroporation and stable transformation of newborn mice by plasmid DNA Biochim Biophys Acta 1991 1088: 131–134
Glasspool-Malone J, Somiari S, Drabick JJ, Malone RW . Efficient nonviral cutaneous transfection Mol Ther 2000 2: 140–146
Drabick JJ et al. Cutaneous transfection and immune responses to intradermal nucleic acid vaccination are significantly enhanced by in vivo electropermeabilization Mol Ther 2000 3: 249–255
Heller R et al. Intradermal delivery of IL-12 plasmid DNA by in vivo electroporation DNA Cell Biol 2001 20: 21–26
Heller R et al. In vivo gene electroinjection and expression in rat liver FEBS Lett 1996 389: 225–228
Suzuki T et al. Direct gene transfer into rat liver cells by in vivo electroporation FEBS Lett 1998 425: 436–440
Muramatsu T et al. Foreign gene expression in the mouse testis by localized in vivo gene transfer Biochem Biophys Res Com 1997 233: 45–49
Wolff JA et al. Direct gene transfer into mouse muscle in vivo Science 1990 247: 1465–1468
Aihara H, Miyazaki J-I . Gene transfer into muscle by electroporation in vivo Nat Biotech 1998 6: 867–870
Mir LM et al. High-efficiency gene transfer into skeletal muscle mediated by electric pulses Proc Natl Acad Sci USA 1999 96: 4262–4267
Mathiesen I . Electropermeabilization of skeletal muscle enhances gene transfer in vivo Gene Therapy 1999 6: 508–514
Rizzuto G et al. Efficient and regulated erythropoietin production by naked DNA injection and muscle electroporation Proc Natl Acad Sci USA 1999 96: 6417–6422
Vicat JM et al. Muscle transfection by electroporation with high-voltage and short-pulse currents provides high-level and long-lasting gene expression Hum Gene Ther 2000 11: 909–916
Maruyama H et al. Continuous erythropoietin delivery by muscle-targeted gene transfer using in vivo electroporation Hum Gene Ther 2000 11: 429–437
Widera G et al. Increased DNA vaccine delivery and immunogenicity by electroporation in vivo J Immunol 2000 164: 4635–4640
Zucchelli S et al. Enhancing B- and T-cell immune response to a hepatitis C virus E2 DNA vaccine by intramuscular electrical gene transfer J Virol 2000 74: 11598–11607
Kadowaki S-E et al. Protection against influenza virus infection in mice immunized by administration of hemagglutinin-expressing DNAs with electroporation Vaccine 2000 18: 2779–2788
Chen Z et al. Cross protection against a lethal influenza virus infection by DNA vaccine to neuraminidase Vaccine 2000 18: 3214–3222
Lucas ML, Heller R . Immunomodulation by electrically enhanced delivery of plasmid DNA encoding IL-12 to murine skeletal muscle Mol Ther 2001 3: 47–53
Lucas ML, Jaroszeski MJ, Gilbert R, Heller R . In vivo electroporation using an exponentially enhanced pulse, a new wave form DNA Cell Biol 2001 20: 183–188
Nishi T et al. High-efficiency in vivo gene transfer using intraarterial plasmid DNA injection following in vivo electroporation Cancer Res 1996 56: 1050–1055
Rols M-P et al. In vivo electrically mediated protein and gene transfer in murine melanoma Nat Biotech 1998 16: 168–171
Wells JM et al. Electroporation-enhanced gene delivery in mammary tumors Gene Therapy 2000 7: 541–547
Heller L et al. Electrically mediated plasmid DNA delivery to hepatocellular carcinomas in vivo Gene Therapy 2000 7: 826–829
Niu G et al. Gene therapy with dominant-negative Stat3 suppresses growth of the murine melanoma B16 tumor in vivo Cancer Res 1999 59: 5059–5063
Goto T et al. Highly efficient electro-gene therapy of solid tumor by using an expression plasmid for the herpes simplex virus thymidine kinase gene Proc Natl Acad Sci USA 2000 97: 354–359
Yamashita Y-I et al. Electroporation-mediated interleukin-12 gene therapy for hepatocellular carcinoma in the mice model Cancer Res 2001 61: 1005–1012
Tamura T et al. Intratumoral delivery of interleukin 12 expression plasmids with in vivo electroporation is effective for colon and renal cancer Human Gene Ther 2001 12: 1265–1276
Kishida T et al. In vivo electroporation-mediated transfer of interleukin-12 and interleukin-18 genes induces significant antitumor effects against melanoma in mice Gene Therapy 2001 8: 1234–1240
Heller L et al. In vivo electroporation of plasmids encoding GM-CSF or interleukin-2 into existing B16 melanomas combined with electrochemotherapy induces long-term antitumor immunity Melanoma Res 2000 10: 577–583
Mendiratta SK et al. Therapeutic tumor immunity induced by polyimmunization with melanoma antigens gp100 and TRP-2 Cancer Res 2001 61: 859–863
Li S et al. Intramuscular electroporation delivery of IFN α gene therapy for inhibition of tumor growth located at a distant site Gene Therapy 2001 8: 400–407
Gilbert RA, Jaroszeski MJ, Heller R . Novel electrode designs for electrochemotherapy Biochim Biophys Acta 1997 1334: 9–14
Brasier AP . Reporter system using firefly luciferase Ausubel FM (eds); Short Protocols in Molecular Biology John Wiley 1992 pp 9–21
Bird AP . CpG rich islands and the function of DNA methylation Nature 1986 321: 209–213
Krieg AM et al. CpG motifs in bacterial DNA trigger direct B-cell activation Nature 1995 374: 546–549
Sato Y et al. Immunostimulatory DNA sequences necessary for effective intradermal gene immunization Science 1996 273: 352–354
Krieg AM, Hartmann G, Yi AK . Mechanism of action of CpG DNA Curr Top Micro Immunol 1999 247: 1–21
Tokunaga T et al. Antitumor activity of deoxyribonucleic acid fraction from Mycobacterium bovis BCG. I. Isolation, physiochemical characterization, and antitumor activity J Natl Cancer Inst 1984 72: 955–962
Yamamoto S, Kuramoto E, Shimada S, Tokunaga T . In vitro augmentation of natural killer cell activity and production of interferon-alpha/beta and -gamma with deoxyribonucleic acid fraction from Mycobacterium bovis BCG Jpn J Cancer Res 1988 79: 866–873
Weiner GJ et al. Immunostimulatory oligodeoxynucleotides containing the CpG motif are effective as immune adjuvants in tumor antigen immunization Proc Natl Acad Sci USA 1997 94: 10833–10837
Liu H-M et al. Immunostimulatory CpG oligodeoxynucleotides enhance the immune response to vaccine strategies involving granulocyte-macrophage colony-stimulating factor Blood 1998 92: 3730–3736
Dow SW et al. Lipid-DNA complexes induce potent activation of innate immune responses and antitumor activity when administered intravenously J Immunol 1999 163: 1552–1561
McMahon JM et al. Inflammatory responses following direct injection of plasmid DNA into skeletal muscle Gene Therapy 1998 5: 1283–1290
Acknowledgements
The T820 Electrosquare porator and autoswitcher and plasmid VR1255 were kind gifts of BTX (San Diego, CA, USA) and Vical, Inc (San Diego, CA, USA) respectively. This project was supported in part by the University of South Florida Research and Creative Scholarship Grant Program, the American Cancer Society, Florida Division, Inc, the Pathology Core Facility, Histology Laboratory, at the University of South Florida College of Medicine and at the H Lee Moffitt Cancer Center and Research Institute, and the Center for Molecular Delivery.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Heller, L., Coppola, D. Electrically mediated delivery of vector plasmid DNA elicits an antitumor effect. Gene Ther 9, 1321–1325 (2002). https://doi.org/10.1038/sj.gt.3301802
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301802
Keywords
This article is cited by
-
Modification of the tumor microenvironment enhances immunity with plasmid gene therapy
Cancer Gene Therapy (2024)
-
Intratumoral DNA-based delivery of checkpoint-inhibiting antibodies and interleukin 12 triggers T cell infiltration and anti-tumor response
Cancer Gene Therapy (2022)
-
APOBEC3A intratumoral DNA electroporation in mice
Gene Therapy (2017)
-
Cytosolic DNA Sensor Upregulation Accompanies DNA Electrotransfer in B16.F10 Melanoma Cells
Molecular Therapy - Nucleic Acids (2016)
-
Electrotransfer of single-stranded or double-stranded DNA induces complete regression of palpable B16.F10 mouse melanomas
Cancer Gene Therapy (2013)