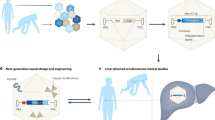
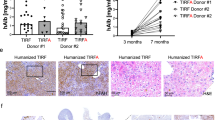
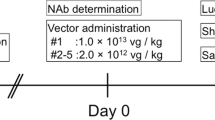
Abstract
Adenoviral vectors efficiently target normal liver cells; however, a clear-cut description of the safety boundaries for using adenovectors in hepatic cirrhosis has not been settled. With this in mind, we used a first-generation, replication-deficient adenoviral vector carrying the E. coli lacZ gene (Ad5βGal) to monitor therapeutic range, biodistribution, toxicity and transduction efficiency in Wistar rats made cirrhotic by two different experimental approaches resembling alcoholic cirrhosis and biliary cirrhosis in humans. Further, we show proof of concept on fibrosis reversion by a ‘therapeutic’ Ad-vector (AdMMP8) carrying a gene coding for a collagen-degrading enzyme. Dose–response experiments with Ad5βGal ranging from 1 × 108–3 × 1012 viral particles (vp) per rat (250 g), demonstrated that adenovirus-mediated gene transfer via iliac vein at 3 × 1011 vp/rat, resulted in an approximately 40% transduction in livers of rats made cirrhotic by chronic intoxication with carbon tetrachloride, compared with approximately 80% in control non-cirrhotic livers. In rats made cirrhotic by bile-duct obstruction only, 10% efficiency of transduction was observed. Biodistribution analyses showed that vector expression was detected primarily in liver and at a low level in spleen and kidney. Although there was an important increase in liver enzymes between the first 48 h after adenovirus injection in cirrhotic animals compared to non-transduced cirrhotic rats, this hepatic damage was resolved after 72–96 h. Then, the cDNA for neutrophil collagenase, also known as Matrix Metalloproteinase 8 (MMP8), was cloned in an Ad-vector and delivered to cirrhotic rat livers being able to reverse fibrosis in 44%. This study demonstrates the potential use of adenoviral vectors in safe transient gene therapy strategies for human liver cirrhosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Olaso E., Friedman S.L. . Molecular regulation of hepatic fibrogenesis J Hepatol 1998 29: 836 836
Armendariz-Borunda J., Seyer J.M., Kang A.H., Raghow R. . Regulation of TGFβ gene expression in rat liver intoxicated with carbon tetrachloride FASEB J 1990 4: 215 215
Terao R. et al. Supression of proliferation cholangitis in a rat model with direct adenovirus-mediated retinoblastoma gene transfer to the biliary tract Hepatology 1998 28: 605 605
Lusky M. et al. In vitro and in vivo biology of recombinant adenovirus vectors with E1, E1/E2A, or E1/E4 deleted J Virol 1998 72: 2022 2022
Worgall S., Wolff G., Falck-Pedersen E., Crystal R.G. . Innate immune mechanisms dominate elimination of adenoviral vectors following in vivo administration Hum Gene Ther 1997 8: 37 37
Drazan K.E. et al. Hepatic function is preserved following liver-directed, adenovirus-mediated gene transfer J Surg Res 1995 59: 299 299
Lieber A. et al. The role of Kupffer cell activation and viral gene expression in early liver toxicity after infusion of recombinant adenovirus vectors J Virol 1997 71: 8798 8798
Salgado S. et al. Liver cirrhosis is reverted by urokinase-type plasminogen activator gene therapy Mol Ther 2000 2: 545 545
Rudolph K.L. et al. Inhibition of experimental liver cirrhosis in mice by telomerase gene delivery Science 2000 287: 1253 1253
Hecht N. et al. Hyper-IL-6 gene therapy reverses fulminant hepatic failure Mol Ther 2001 3: 683 683
Nakatani T. et al. Assessment of efficiency and safety of adenovirus-mediated gene transfer into normal and damaged murine livers Gut 2000 47: 563 563
Ramm G.A. et al. Contribution of hepatic parenchymal and nonparnchymal cells to hepatic fibrogenesis in biliary atresia Am J Pathol 1998 153: 527 527
Kossakowska A.E. et al. Altered balance between matrix metalloproteinases and their inhibitors in experimental biliary fibrosis Am J Pathol 1998 53: 1895 1895
Desmouliere A. et al. Extracellular matrix deposition, lysyl oxidase expression and myofibroblastic differentiation during the initial stages of cholestatic fibrosis in rat Lab Invest 1997 76: 765 765
Zern M.A., Kresina T.F. . Hepatic drug delivery and gene therapy Hepatology 1997 25: 484 484
de Roos W.K. et al. Isolated-organ perfusion for local gene delivery: efficient adenovirus-mediated gene transfer into the liver Gene Therapy 1997 71: 55 55
Ferry N., Heard J.M. . Liver-directed gene transfer vectors Hum Gene Ther 1998 9: 1975 1975
Nakamura T., Akiyoshi H., Saito I., Sato K. . Adenovirus-mediated gene expression in the septal cells of cirrhotic rat livers J Hepatol 1999 30: 101 101
Aguilar L.K. et al. A prescription for gene therapy Mol Ther 2000 1: 385 385
Sullivan D.E. et al. Liver-directed gene transfer in non-human primates Hum Gene Ther 1997 8: 1195 1195
Siller-Lopez F. et al. Truncated active matrix metalloproteinase-8 gene expression in HepG2 cells is active against native type I collagen J Hepatol 2000 33: 758 758
Lee S.S. et al. Hemodynamic characterization of chronic bile duct-ligated rats: effect of pentobarbital sodium Am J Physiol 1986 251: 176 176
Armendariz-Borunda J., Katayama K., Seyer J.M. . Transcriptional mechanisms of type I collagen gene expression are differentially regulated by IL-1β TNF-α and TGFβ in Ito cells J Biol Chem 1992 267: 14316 14316
Nyberg-Hoffman C. et al. Sensitivity and reproducibility in adenoviral infectious titer determination Nat Med 1997 3: 808 808
Mizuguchi H., Kay M.A. . Efficient construction of a recombinant adenovirus vector by an improved in vitro ligation method Hum Gene Ther 198 9: 2577 2577
Weiss D.J., Liggitt D., Clark J.G. . In situ histochemical detection of β-galactosidase activity in lung: assessment of X-gal reagent in distinguishing lacZ gene expression and endogenus β-galactosidase activity Hum Gene Ther 1997 8: 1545 1545
Lopez-De Leon A., Rojkind M. . A simple micromethod for collagen and total protein determination in formalin-fixed paraffin-ambedded sections J Histochem Cytochem 1985 33: 737 737
Acknowledgements
This work was supported in part by a grant from CONACyT No. 28832-M to Juan Armendariz-Borunda. The authors are indebted to Dr Guillermo Grijalva and Jose M Vera for their invaluable assistance.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Garcia-Bañuelos, J., Siller-Lopez, F., Miranda, A. et al. Cirrhotic rat livers with extensive fibrosis can be safely transduced with clinical-grade adenoviral vectors. Evidence of cirrhosis reversion. Gene Ther 9, 127–134 (2002). https://doi.org/10.1038/sj.gt.3301647
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301647
Keywords
This article is cited by
-
Delivery of Anti-IFNAR1 shRNA to Hepatic Cells Decreases IFNAR1 Gene Expression and Improves Adenoviral Transduction and Transgene Expression
Molecular Biotechnology (2022)
-
Adenovirus Biodistribution is Modified in Sensitive Animals Compared to Naïve Animals
Molecular Biotechnology (2020)
-
Interferon-α Silencing by Small Interference RNA Increases Adenovirus Transduction and Transgene Expression in Huh7 Cells
Molecular Biotechnology (2018)
-
Regulation of hepatocyte identity and quiescence
Cellular and Molecular Life Sciences (2015)
-
AAV vectors transduce hepatocytes in vivo as efficiently in cirrhotic as in healthy rat livers
Gene Therapy (2012)