Abstract
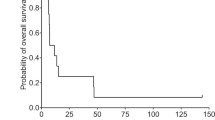
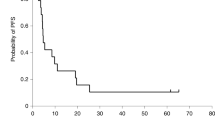
The herpes simplex virus (HSV) ICP34.5 null mutant 1716 replicates selectively in actively dividing cells and has been proposed as a potential treatment for cancer, particularly brain tumours. We present a clinical study to evaluate the safety of 1716 in patients with relapsed malignant glioma. Following intratumoural inoculation of doses up to l05 p.f.u., there was no induction of encephalitis, no adverse clinical symptoms, and no reactivation of latent HSV. Of nine patients treated, four are currently alive and well 14–24 months after 1716 administration. This study demonstrates the feasibility of using replication-competent HSV in human therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Rampling R . Modern aspects of radiation therapy for glial tumours of the brain Forum 1998 8: 289–301
Rampling R . Chemotherapy. Determining the appropriate treatment. In: Hopkins A, Davies EH (eds) Improving Care for Patients with Malignant Glioma Royal College of Physicians: London 1997 pp 63–74
Rajan B . et al. Survival in patients with recurrent glioma as a measure of treatment efficacy: prognostic factors following nitrosourea chemotherapy Eur J Cancer 1994 30A: 1809–1815
Gutin P, Leibel S . Stereotaxic interstitial irradiation of malignant brain tumours Neurol Clin 1985 3: 883–893
Mbidde EK et al. High dose BCNU chemotherapy with autologous bone marrow transplantation and full dose radiotherapy for grade IV astrocytoma Br J Cancer 1988 58: 779–782
Lucas GL et al. Treatment results of stereotactic interstitial brachytherapy for primary and metastatic brain tumours Int J Radiat Oncol Biol Phys 1991 21: 715–721
MacLean AR et al. Herpes simplex virus type 1 variants 1714 and 1716 pinpoint neurovirulence related sequences in Glasgow strain 17+ between immediate early gene 1 and the ‘a’ sequence J Gen Virol 1991 72: 631–639
Dolan A, McKie E, MacLean A, McGeoch D . Status of ICP34.5 gene in herpes simplex virus type 1 strain 17 J Gen Virol 1992 73: 971–973
Ackermann M et al. Identification by antibody to a synthetic peptide of a protein specified by a diploid gene located in the terminal repeats of the L component of the herpes simplex virus genome J Virol 1986 58: 843–850
Brown SM et al. Cell type and cell state determine differential in vitro growth of non virulent ICP34.5 negative herpes simplex virus J Gen Virol 1994 75: 2367–2377
Randazzo RB et al. Treatment of experimental intracranial murine melanoma with neuroattenuated herpes simplex virus type 1 mutant Virology 1995 211: 94–101
Kesari S et al. Therapy of experimental human brain tumors using a neuroattenuated herpes simplex virus mutant Lab Invest 1995 73: 636–648
Kucharczuk J et al. Use of a ‘replication-restricted’ herpes virus to treat experimental human malignant mesothelioma Cancer Res 1997 57: 466–471
McKie EA, Brown SM, MacLean A, Graham DI . Histopathological responses in the CNS following innoculation with a non-neurovirulent mutant (1716) of herpes simplex virus type 1 (HSV1): relevence for gene and cancer therapy Neuropath Appl Neurobiol 1998 24: 367–372
McKie EA et al. Selective in vitro killing of primary CNS tumours using herpes simplex virus type1 (HSV-1), ICP34.5 null mutants – a potentially effective clinical therapy Br J Cancer 1996 74: 745–752
Brown SM, MacLean A, McKie E, Harland J . The herpes simplex virulence factor ICP34.5 and the cellular protein MyD116 complex with proliferating cell nuclear antigen through the 63 amino acid domain conserved in ICP34.5, MyD116 and GADD34 J Virol 1997 71: 9442–9449
Chou J, Roizman B . The gamma 34.5 gene of herpes simplex virus type 1 precludes neuroblastoma cells from triggering total shut off of protein synthesis characteristic of programmed cell death in neuronal cells Proc Nat Acad Sci USA 1992 89: 3266–3270
Chou J, Roitzman B . HSV1 34.5 gene function which blocks the host response to infection maps in the homologous domain of genes expressed during growth arrest and DNA damage Proc Natl Acad Sci USA 1994 91: 5247–5251
He B et al. The carboxyl terminus of the murine MyD116 gene substitutes for the corresponding domain of the gamma 34.5 gene of herpes simplex virus to preclude the premature shutoff of total protein synthesis in infected human cells J Virol 1996 70: 84–90
Mulvey M, Poppers J, Ladd J, Mohr I . A herpes virus ribosome associated RNA binding protein confers a growth advantage upon mutants deficient in GADD 34 related function J Virol 1999 73: 3375–3385
Valyi-Nagi T et al. The HSV-1 strain 17+gamma 34.5 deletion mutant 1716 is avirulent in SCID mice J Gen Virol 1994 75: 2059–2063
Harland J, Brown SM . HSV growth preparation and assay. In: Brown SM, MacLean AR (eds) Herpes Simplex Virus Protocols. Methods in Molecular Medicine Humana Press 1997
Arbuck SG . Workshop on phase 1 study design Ninth NCI/EORTC New Drug Development Symposium, Amsterdam, March 1996 Ann Oncol 1996 7: 567–573
Steward WS . Phase 1 studies–alternative endpoints: when is toxicity not appropriate? In 9th NCI/EORTC Symposium on New Drugs in Cancer Therapy, Amsterdam 1996
Lehrman S . Virus treatment questioned after gene therapy death Nature 1999 401: 517–518
Cruickshank GS . The use of SPECT in the analysis of brain tumours, In: Duncan R (ed) SPECT Imaging of the Brain Kluwer Academic Publishers: Dordrecht 1997 pp 161–178
Macdonald DR, Terrance LC, Cascino S, Schold C . Responsecriteria for phase II studies of supratentorial malignant glioma J Clin Oncol 1990 8: 1277–1280
Kesari S et al. Selective vulnerability of mouse CNS neurons to latent infection with neuroattenuated HSV1 J Neurosci 1996 16: 5644–5653
Kesari S et al. A neuroattenuated ICP34.5-deficient herpes simplex type 1 replicates in ependymal cells of the murine central nervous system J Gen Virol 1998 79: 525–536
Lasner TM et al. Toxicity and neuronal infection of a HSV-1 ICP34.5 mutant in nude mice J Neurovirol 1998 4: 100–105
Chambers R et al. Comparison of genetically engineered herpes simplex viruses for the treatment of brain tumours in a scid mouse model of human malignant glioma Proc Natl Acad Sci USA 1995 92: 1411–1415
Markert JM et al. Initial report of phase 1 trial of genetically-engineered HSV-1 in patients with malignant glioma. In: 23rd International Herpesvirus Workshop. York, 1998.
Brown SM, Ritchie DA, Subak-Sharpe JH . Genetic studies with Herpes Simplex virus type 1. The isolation of temperature sensitive mutants, their arrangement into complementation groups and recombination analysis leading to a linkage map J Gen Virol 1973 18: 329–346
Cone RW et al. Extended duration of Herpes Simplex Virus DNA in genital lesions detected by polymerase chain reaction J Infect Dis 1991 164: 757
Acknowledgements
This work was supported by the UK Medical Research Council project grant No. G9539438N to RR, GC and MB. GMP grade 1716 was generously supplied without charge from Q1 Biotech, Glasgow and we are indebted to David Onions and Gillian Lees for their support. We are grateful to Howard Marsden, MRC Virology Unit, Glasgow who kindly supplied the ZlF11 antibody. Dr A Farrell and Mr E Galloway gave invaluable help and advise with respect to the immunology measurements. We also thank the GTAC committee and the MCA for allowing this study to proceed.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rampling, R., Cruickshank, G., Papanastassiou, V. et al. Toxicity evaluation of replication-competent herpes simplex virus (ICP 34.5 null mutant 1716) in patients with recurrent malignant glioma. Gene Ther 7, 859–866 (2000). https://doi.org/10.1038/sj.gt.3301184
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301184
Keywords
This article is cited by
-
Clinical advances in oncolytic virus therapy for malignant glioma: a systematic review
Discover Oncology (2023)
-
Oncolytic Viral Therapy for Malignant Glioma and Their Application in Clinical Practice
Neurotherapeutics (2022)
-
Implications of immune cells in oncolytic herpes simplex virotherapy for glioma
Brain Tumor Pathology (2022)
-
A bibliometric review of oncolytic virus research as a novel approach for cancer therapy
Virology Journal (2021)
-
Glioblastoma infiltration of both tumor- and virus-antigen specific cytotoxic T cells correlates with experimental virotherapy responses
Scientific Reports (2020)