Abstract
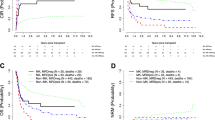
Cytogenetic studies were performed as part of all diagnostic and surveillance bone marrow examinations in 956 newly diagnosed patients with multiple myeloma (MM) receiving total therapy (TT) protocols and in 1085 previously treated patients enrolled in non-TT protocols. In both groups, cytogenetic abnormalities (CA) were present in one-third at baseline and persisted in 14% prior to first and 10% prior to second transplant (TT, 5%; non-TT, 15%); post-transplant detection rates increased progressively with time, from 7% within 6 months to 21% within 24 months to 28% at relapse. According to multivariate analyses, overall survival was adversely affected by the presence of CA at baseline (hazard ratio (HR)=7.20, P<0.001) and the development of CA both prior to (HR=3.28, P<0.001) and after first transplant (HR=6.24, P<0.001), whereas suppression of CA pretransplant was favorable (HR=0.38, P<0.001). The presence of CA at relapse further distinguished patients with a short median post-relapse survival of only 11 versus 47 months in those without CA (P<0.0001). Post-relapse survival was independently adversely affected by the detection of CA both at baseline (HR=1.35, P=0.044) and relapse (HR=2.47, P<0.001). Collectively, these results underscore the importance of monitoring for CA and attest to the favorable prognostic consequences of CA suppression with effective therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gould J, Alexanian R, Goodacre A, Pathak S, Hecht B, Barlogie B . Plasma cell karyotype in multiple myeloma. Blood 1988; 71: 453–456.
Tricot G, Barlogie B, Jagannath S, Bracy D, Mattox S, Vesole DH et al. Poor prognosis in multiple myeloma is associated only with partial or complete deletions of chromosome 13 or abnormalities involving 11q and not with other karyotype abnormalities. Blood 1995; 86: 4250–4256.
Tricot G, Sawyer JR, Jagannath S, Desikan KR, Siegel D, Naucke S et al. The unique role of cytogenetics in the prognosis of patients with myeloma receiving high dose therapy and autotransplants. J Clin Oncol 1997; 15: 2659–2666.
Seong C, Delasalle K, Hayes K, Weber D, Dimopoulos M, Swantkowski J et al. Prognostic value of cytogenetics in multiple myeloma. Br J Haematol 1998; 101: 189–195.
Tricot G, Spencer T, Sawyer J, Spoon D, Desikan R, Fassas A et al. Predicting long-term (⩾5 years) event-free survival in multiple myeloma patients following planned tandem autotransplants. Br J Haematol 2002; 116: 211–217.
Jacobson J, Barlogie B, Shaughnessy J, Drach J, Tricot G, Fassas A et al. MDS-type abnormalities within myeloma signature karyotype (MM-MDS): only 13% 1-year survival despite tandem transplants. Br J Haematol 2003; 122: 430–440.
Desikan R, Barlogie B, Sawyer J, Ayers D, Tricot G, Badros A et al. Results of high-dose therapy for 1000 patients with multiple myeloma: durable complete remissions and superior survival in the absence of chromosome 13 abnormalities. Blood 2000; 95: 4008–4010.
Fonseca R, Barlogie B, Bataille R, Bastard C, Bergsagel PL, Chesi M et al. Genetics and cytogenetics of MM: a workshop report. Cancer Res 2004; 64: 1546–1558.
Shaughnessy J, Jacobson J, Sawyer J, McCoy J, Fassas A, Zhan F et al. Continuous absence of metaphase-defined cytogenetic abnormalities especially of chromosome 13 and hypodiploidy assures long-term survival in multiple myeloma treated with Total Therapy I; interpretation in the context of global gene expression. Blood 2003; 101: 3849–3856.
Barlogie B, Alexanian R, Pershouse M, Smallwood L, Smith L . Cytoplasmic immunoglobulin content in multiple myeloma. J Clin Invest 1985; 76: 765–769.
Drach J, Schuster J, Nowotny H, Angerler J, Rosenthal F, Fiegl M et al. Multiple myeloma: high incidence of chromosomal aneuploidy as detected by interphase fluorescence in situ hybridization. Cancer Res 1995; 55: 3854–3859.
Shaughnessy J, Tian E, Sawyer J, Bumm K, Landes R, Badros A Sawyer J et al. High incidence of chromosome 13 deletions in multiple myeloma detected by multiprobe interphase FISH. Blood 2000; 96: 1505–1511.
Shaughnessy Jr J, Tian E, Sawyer J, McCoy J, Tricot G, Jacobson J et al. Prognostic impact of cytogenetic and interphase fluorescence in situ hybridization-defined chromosome 13 deletion in multiple myeloma: early results of total therapy II. Br J Haematol 2003; 120: 44–52.
Zhan F, Huang Y, Colla S, Stewart JP, Hanamura I, Gupta S Colla S et al. The molecular classification of multiple myeloma. Blood 2006; 108: 2020–2028.
Shaughnessy Jr JD, Zhan F, Burington BE, Huang Y, Colla S, Hanamura I et al. A validated gene expression model of high-risk multiple myeloma is defined by deregulated expression of genes mapping to chromosome 1. Blood 2007; 109: 2276–2284.
Shaughnessy Jr JD, Haessler J, van Rhee F, Anaissie E, Pineda-Roman M, Cottler-Fox M et al. Testing standard and genetic parameters in 220 patients with multiple myeloma with complete data sets: superiority of molecular genetics. Br J Haematol 2007; 137: 530–536.
Haessler J, Shaughnessy JD, Zhan F, Crowley J, Epstein J, van Rhee F et al. Benefit of complete response in multiple myeloma limited to high-risk subgroup identified by gene expression profiling. Clin Cancer Res 2007; 13: 7073–7079.
Jacobson J, Barlogie B, Shaughnessy J, Drach J, Tricot G, Fassas A et al. MDS-type abnormalities within myeloma signature karyotype (MM-MDS): only 13% 1-year survival despite tandem transplants. Br J Haematol 2003; 133: 430–440.
Barlogie B, Jagannath S, Vesole DH, Naucke S, Cheson B, Mattox S et al. Superiority of tandem autologous transplantation over standard therapy for previously untreated multiple myeloma. Blood 1997; 89: 789–793.
Barlogie B, Jagannath S, Desikan KR, Mattox S, Vesole D, Siegel D et al. Total therapy with tandem transplants for newly diagnosed multiple myeloma. Blood 1999; 93: 55–65.
Barlogie B, Tricot GJ, van Rhee F, Angtuaco E, Walker R, Epstein J et al. Long-term outcome results of the first tandem autotransplant trial for multiple myeloma. Br J Haematol 2006; 135: 158–164.
Barlogie B, Tricot G, Anaissie E, Shaughnessy J, Rasmussen E, van Rhee F et al. Thalidomide and hematopoietic-cell transplantation for multiple myeloma. N Engl J Med 2006; 354: 1021–1030.
van Rhee F, Bolejack V, Hollmig K, Pineda-Roman M, Anaissie E, Epstein J et al. High serum-free light chain levels and their rapid reduction in response to therapy define an aggressive multiple myeloma subtype with poor prognosis. Blood 2007; 110: 827–832.
Barlogie B, Anaissie E, van Rhee F, Haessler J, Hollmig K, Pineda-Roman M et al. Incorporating bortezomib into upfront treatment for multiple myeloma: early results of total therapy 3. Br J Haematol 2007; 138: 176–185.
Pineda M, Zangari M, Haessler J, Anaissie E, Tricot J, van Rhee F et al. Sustained complete remissions in multiple myeloma linked to bortezomib in total therapy 3: comparison with total therapy 2. Br J Haematol (in press).
Pineda M, Barlogie B, Anaissie E, Zangari M, Bolejack V, van Rhee F et al. The Arkansas experience since 1989 in 3077 patients. Cancer (in press).
Sawyer J, Waldron J, Jagannath S, Barlogie B . Cytogenetic findings in 200 patients with multiple myeloma. Cancer Genet Cytogenet 1995; 82: 41–49.
Kaplan EL, Meier P . Nonparametric estimation for incomplete observations. J Am Stat Assoc 1958; 53: 457–481.
Mantel N . Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep 1966; 50: 163–170.
Cox DR . Regression models and life-tables (with discussion). J Royal Stat Soc, Series B 1972; 34: 187–220.
Greipp PR, San Miguel J, Durie BG, Crowley JJ, Barlogie B, Bladé J et al. International staging system for multiple myeloma. J Clin Oncol 2005; 23: 3412–3420.
Acknowledgements
This work was supported in part by CA55819 from the National Cancer Institute, Bethesda, MD, USA.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions: BB and JS designed the study; VA and BB wrote the paper; JS performed and supervised cytogenetic analyses; CB and JG collected data into myeloma database; VA, FvR, EA and BB treated patients on protocols and AH and JC performed statistical analyses.
Rights and permissions
About this article
Cite this article
Arzoumanian, V., Hoering, A., Sawyer, J. et al. Suppression of abnormal karyotype predicts superior survival in multiple myeloma. Leukemia 22, 850–855 (2008). https://doi.org/10.1038/sj.leu.2405091
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2405091
Keywords
This article is cited by
-
Attainment of at least a very good partial response after induction treatment is an important surrogate of longer survival for multiple myeloma
Bone Marrow Transplantation (2010)
-
The research mission in myeloma
Leukemia (2009)
-
Making progress in treating multiple myeloma with total therapies: issue of complete remission and more
Leukemia (2008)
-
A kinder, gentler way: control of the proliferative tumor compartment, not cosmetic complete response, should be the goal of myeloma therapy
Leukemia (2008)