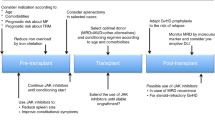
Abstract
Optimal clinical management of patients with primary myelofibrosis and post-essential thrombocythemia/polycythemia vera myelofibrosis is a challenge, given the typically advanced age of presentation and variability of the disease course and prognosis. Current medical therapeutic options have not demonstrated an impact on the disease course, which exceeds the palliation of disease-related extramedullary hematopoiesis and alleviation of cytopenias. In contrast, allogeneic stem cell transplantation (SCT) can lead to ‘cure’ but is limited due to patient's age or comorbidities. Currently, in patients, who are reasonable candidates, SCT (frequently with a reduced intensity conditioning regimen) is employed for intermediate- to high-risk disease. Current pharmaco-medical therapy is used as a bridge to transplant, or instead of transplant in poor transplant candidates. Pathogenetic insights, especially the discovery of the Janus kinase (JAK)2V617F mutation, have ushered in a host of new potential therapeutic agents that may augment the role of medical therapy. Similarly, the boundaries of transplantation continue to alter with strategies that decrease conditioning-related toxicity, improved antimicrobial prophylaxis and decreased graft-versus-host disease. The potential for continued improvements in both medical and transplant therapy suggests that for the immediate future the optimal choices for an individual patient will remain potentially volatile and present complex decisions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mesa RA, Verstovsek S, Cervantes F, Barosi G, Reilly JT, Dupriez B et al. Primary myelofibrosis (PMF), post polycythemia vera myelofibrosis (post-PV MF), post essential thrombocythemia myelofibrosis (post-ET MF), blast phase PMF (PMF-BP): Consensus on terminology by the international working group for myelofibrosis research and treatment (IWG-MRT). Leuk Res 2007; 31: 737–740.
Mesa RA, Silverstein MN, Jacobsen SJ, Wollan PC, Tefferi A . Population-based incidence and survival figures in essential thrombocythemia and agnogenic myeloid metaplasia: an Olmsted County Study, 1976–1995. Am J Hematol 1999; 61: 10–15.
Cervantes F, Barosi G, Hernandez-Boluda JC, Marchetti M, Montserrat E . Myelofibrosis with myeloid metaplasia in adult individuals 30 years old or younger: presenting features, evolution and survival. Eur J Haematol 2001; 66: 324–327.
Cervantes F, Barosi G, Demory JL, Reilly J, Guarnone R, Dupriez B et al. Myelofibrosis with myeloid metaplasia in young individuals: disease characteristics, prognostic factors and identification of risk groups. Br J Haematol 1998; 102: 684–690.
Fruehauf S, Buss EC, Topaly J, Kreipe HH, Ho AD . Myeloablative conditioning in myelofibrosis using i.v. treosulfan and autologous peripheral blood progenitor cell transplantation with high doses of CD34+ cells results in hematologic responses—follow-up of three patients. Haematologica 2005; 90: ECR08.
Anderson JE, Tefferi A, Craig F, Holmberg L, Chauncey T, Appelbaum FR et al. Myeloablation and autologous peripheral blood stem cell rescue results in hematologic and clinical responses in patients with myeloid metaplasia with myelofibrosis. Blood 2001; 98: 586–593.
Ngirabacu MC, Ravoet C, Dargent JL, Meuleman N, Ahmad I, Ysebrant L et al. Long term follow-up of autologous peripheral blood stem cell transplantation in the treatment of a patient with acute panmyelosis with myelofibrosis. Haematologica 2006; 91 (12 Suppl): ECR53.
Dokal I, Jones L, Deenmamode M, Lewis SM, Goldman JM . Allogeneic bone marrow transplantation for primary myelofibrosis. Br J Haematol 1989; 71: 158–160.
Creemers GO, Lowenberg B, Hagenbeek A . Allogeneic bone marrow transplantation for primary myelofibrosis. Br J Haematol 1992; 82: 772–773.
Byrne JL, Beshti H, Clark D, Ellis I, Haynes AP, Das-Gupta E et al. Induction of remission after donor leucocyte infusion for the treatment of relapsed chronic idiopathic myelofibrosis following allogeneic transplantation: evidence for a ‘graft vs myelofibrosis’ effect. Br J Haematol 2000; 108: 430–433.
Cervantes F, Rovira M, Urbano-Ispizua A, Rozman M, Carreras E, Montserrat E . Complete remission of idiopathic myelofibrosis following donor lymphocyte infusion after failure of allogeneic transplantation: demonstration of a graft-versus-myelofibrosis effect. Bone Marrow Transplant 2000; 26: 697–699.
Guardiola P, Anderson JE, Bandini G, Cervantes F, Runde V, Arcese W et al. Allogeneic stem cell transplantation for agnogenic myeloid metaplasia: a European Group for Blood and Marrow Transplantation, Societe Francaise de Greffe de Moelle, Gruppo Italiano per il Trapianto del Midollo Osseo, and Fred Hutchinson Cancer Research Center Collaborative Study. Blood 1999; 93: 2831–2838.
Deeg HJ, Gooley TA, Flowers ME, Sale GE, Slattery JT, Anasetti C et al. Allogeneic hematopoietic stem cell transplantation for myelofibrosis. Blood 2003; 102: 3912–3918.
Kerbauy DM, Gooley TA, Sale GE, Flowers ME, Doney KC, Georges GE et al. Hematopoietic cell transplantation as curative therapy for idiopathic myelofibrosis, advanced polycythemia vera, and essential thrombocythemia. Biol Blood Marrow Transplant 2007; 13: 355–365.
Ballen K, Sobocinski M, Zhang MJ, Arora M, Horowitz M, Giralt S . Outcome of bone marrow transplantation for myelofibrosis. Blood 2005; 106: Abstract no. 170.
Singhal S, Powles R, Treleaven J, Pollard C, Lumley H, Mehta J . Allogeneic bone marrow transplantation for primary myelofibrosis. Bone Marrow Transplant 1995; 16: 743–746.
Anderson JE, Sale G, Appelbaum FR, Chauncey TR, Storb R . Allogeneic marrow transplantation for primary myelofibrosis and myelofibrosis secondary to polycythaemia vera or essential thrombocytosis. Br J Haematol 1997; 98: 1010–1016.
Daly A, Song K, Nevill T, Nantel S, Toze C, Hogge D et al. Stem cell transplantation for myelofibrosis: a report from two Canadian centers. Bone Marrow Transplant 2003; 32: 35–40.
Mittal P, Saliba RM, Giralt SA, Shahjahan M, Cohen AI, Karandish S et al. Allogeneic transplantation: a therapeutic option for myelofibrosis, chronic myelomonocytic leukemia and Philadelphia-negative/BCR-ABL-negative chronic myelogenous leukemia. Bone Marrow Transplant 2004; 33: 1005–1009.
Ditschkowski M, Beelen DW, Trenschel R, Koldehoff M, Elmaagacli AH . Outcome of allogeneic stem cell transplantation in patients with myelofibrosis. Bone Marrow Transplant 2004; 34: 807–813.
Przepiorka D, Giralt S, Khouri I, Champlin R, Bueso-Ramos C . Allogeneic marrow transplantation for myeloproliferative disorders other than chronic myelogenous leukemia: review of forty cases. Am J Hematol 1998; 57: 24–28.
Guardiola P, Anderson JE, Gluckman E . Myelofibrosis with myeloid metaplasia. N Engl J Med 2000; 343: 659 (Letter).
Hessling J, Kröger N, Werner M, Zabelina T, Hansen A, Kordes U et al. Dose-reduced conditioning regimen followed by allogeneic stem cell transplantation in patients with myelofibrosis with myeloid metaplasia. Br J Haematol 2002; 119: 769–772.
Devine SM, Hoffman R, Verma A, Shah R, Bradlow BA, Stock W et al. Allogeneic blood cell transplantation following reduced-intensity conditioning is effective therapy for older patients with myelofibrosis with myeloid metaplasia. Blood 2002; 99: 2255–2258.
Greyz N, Miller WE, Andrey J, Mason J . Long-term remission of myelofibrosis following nonmyeloablative allogeneic peripheral blood progenitor cell transplantation in older age: the Scripps Clinic experience. Bone Marrow Transplant 2004; 34: 273–274.
Tanner ML, Hoh CK, Bashey A, Holman P, Sun C, Broome HE et al. FLAG chemotherapy followed by allogeneic stem cell transplant using nonmyeloablative conditioning induces regression of myelofibrosis with myeloid metaplasia. Bone Marrow Transplant 2003; 32: 581–585.
Rondelli D, Barosi G, Bacigalupo A, Prchal JT, Popat U, Alessandrino EP et al. Myeloproliferative Diseases-Research Consortium. Allogeneic hematopoietic stem-cell transplantation with reduced-intensity conditioning in intermediate- or high-risk patients with myelofibrosis with myeloid metaplasia. Blood 2005; 105: 4115–4119.
Kröger N, Zabelina T, Schieder H, Panse J, Ayuk F, Stute N et al. Pilot study of reduced-intensity conditioning followed by allogeneic stem cell transplantation from related and unrelated donors in patients with myelofibrosis. Br J Haematol 2005; 128: 690–697.
Merup M, Lazarevic V, Nahi H, Andreasson B, Malm C, Nilsson L et al. Swedish Group for Myeloproliferative Disorders. Different outcome of allogeneic transplantation in myelofibrosis using conventional or reduced-intensity conditioning regimens. Br J Haematol 2006; 135: 367–373.
Snyder DS, Palmer J, Stein AS, Pullarkat V, Sahebi F, Cohen S et al. Allogeneic hematopoietic cell transplantation following reduced intensity conditioning for treatment of myelofibrosis. Biol Blood Marrow Transplant 2006; 12: 1161–1168.
Kröger N, Holler E, Kobbe G, Bornhäuser M, Schwerdtfeger R, Nagler A et al. Dose-reduced conditioning followed by allogeneic stem cell transplantation in patients with myelofibrosis. Blood 2007; 110: Abstract no. 683.
Bacigalupo A, Dominetto A, Pozzi S, Piaggio G, van Lint MT, Zupo S et al. Allogeneic hematopoietic stem cell transplant for patients with idiopathic myelofibrosis using a reduced intensity thiotepa based conditioning regimen. Blood 2007; 110: Abstract no. 684.
Li Z, Deeg HJ . Pros and cons of splenectomy in patients with myelofibrosis undergoing stem cell transplantation. Leukemia 2001; 15: 465–467.
Tefferi A, Mesa RA, Nagorney DM, Schroeder G, Silverstein MN . Splenectomy in myelofibrosis with myeloid metaplasia: a single-institution experience with 223 patients. Blood 2000; 95: 2226–2233.
Barosi G, Ambrosetti A, Centra A, Falcone A, Finelli C, Foa P et al. Splenectomy and risk of blast transformation in myelofibrosis with myeloid metaplasia. Italian Cooperative Study Group on Myeloid with Myeloid Metaplasia. Blood 1998; 91: 3630–3636.
Baxter EJ, Scott LM, Campbell PJ, East C, Fourouclas N, Swanton S et al. Cancer Genome Project. Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet 2005; 365: 1054–1061.
Levine RL, Wadleigh M, Cools J, Ebert BL, Wernig G, Huntly BJ et al. Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocythemia, and myeloid metaplasia with myelofibrosis. Cancer Cell 2005; 7: 387–397.
James C, Ugo V, Le Couedic JP, Staerk J, Delhommeau F, Lacout C et al. A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemia vera. Nature 2005; 434: 1144–1148.
Jones AV, Silver RT, Waghorn K, Curtis C, Kreil S, Zoi K et al. Minimal molecular response in polycythemia vera patients treated with imatinib or interferon alpha. Blood 2006; 107: 3339–3341.
Kralovics R, Passamonti F, Buser AS, Teo SS, Tiedt R, Passweg JR et al. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med 2005; 352: 1779–1790.
Ditschkowski M, Elmaagacli AH, Trenschel R, Steckel NK, Koldehoff M, Beelen DW . No influence of V617F mutation in JAK2 on outcome after allogeneic hematopoietic stem cell transplantation (HSCT) for myelofibrosis. Biol Blood Marrow Transplant 2006; 12: 1350–1351.
Tefferi A, Barosi G, Mesa RA, Cervantes F, Deeg HJ, Reilly JT et al. International Working Group (IWG) consensus criteria for treatment response in myelofibrosis with myeloid metaplasia: On behalf of the IWG for myelofibrosis research and treatment (IWG-MRT). Blood 2006; 108: 1497–1503.
Kröger N, Badbaran A, Holler E, Hahn J, Kobbe G, Bornhaeuser M et al. Monitoring of the JAK2-V617F mutation by highly sensitive quantitative real-time PCR after allogeneic stem cell transplantation in patients with myelofibrosis. Blood 2007; 109: 1316–1321.
Thiele J, Kvasnicka HM, Fischer R, Diehl V . Clinicopathological impact of the interaction between megakaryocytes and myeloid stroma in chronic myeloproliferative disorders: a concise update. Leuk Lymphoma 1997; 24: 463–481.
Wang JC, Chang TH, Goldberg A, Novetsky AD, Lichter S, Lipton J . Quantitative analysis of growth factor production in the mechanism of fibrosis in agnogenic myeloid metaplasia. Exp Hematol 2006; 34: 1617–1623.
Katoh O, Kimura A, Itoh T, Kuramoto A . Platelet derived growth factor messenger RNA is increased in bone marrow megakaryocytes in patients with myeloproliferative disorders. Am J Hematol 1990; 35: 145–150.
Le Bousse-Kerdiles MC, Martyre MC, French INSERM research network on Idiopathic Myelofibrosis. Involvement of the fibrogenic cytokines, TGF-beta and bFGF, in the pathogenesis of idiopathic myelofibrosis. Pathol Biol (Paris) 2001; 49: 153–157.
Martyré MC . Critical review of pathogenetic mechanisms in myelofibrosis with myeloid metaplasia. Curr Hematol Rep 2003; 2: 257–263.
Centurione L, Di Baldassarre A, Zingariello M, Bosco D, Gatta V, Rana RA et al. Increased and pathologic emperipolesis of neutrophils within megakaryocytes associated with marrow fibrosis in GATA-1(low) mice. Blood 2004; 104: 3573–3580.
Thiele J, Kvasnicka HM, Dietrich H, Stein G, Hann M, Kaminski A et al. Dynamics of bone marrow changes in patients with chronic idiopathic myelofibrosis following allogeneic stem cell transplantation. Histol Histopathol 2005; 20: 879–889.
Ni H, Barosi G, Rondelli D, Hoffman R . Studies of the site and distribution of CD34+ cells in idiopathic myelofibrosis. Am J Clin Pathol 2005; 123: 833–839.
Kröger N, Thiele J, Kobbe G, Schwerdtfeger R, Bornhäuser M, Bethge W et al. Rapid Regression of bone marrow fibrosis after dode-reduced allogeneic stem cell transplantation. Exp Hematol 2007; 35: 1719–1722.
Sale GE, Deeg HJ, Porter BA . Regression of myelofibrosis and osteosclerosis following hematopoietic cell transplantation assessed by magnetic resonance imaging and histologic grading. Biol Blood Marrow Transplant 2006; 12: 1285–1294.
Mielcarek M, Martin PJ, Leisenring W, Flowers ME, Maloney DG, Sandmaier BM et al. Graft-versus-host disease after nonmyeloablative versus conventional hematopoietic stem cell transplantation. Blood 2003; 102: 756–762.
Perez-Simon JA, Diez-Campelo M, Martino R, Brunet S, Urbano A, Caballero MD et al. Influence of the intensity of the conditioning regimen on the characteristics of acute and chronic graft-versus-host disease after allogeneic transplantation. Br J Haematol 2005; 130: 394–403.
Weiden PL, Flournoy N, Thomas ED, Prentice R, Fefer A, Buckner CD et al. Antileukemic effect of graft-versus-host disease in human recipients of allogeneic-marrow grafts. N Engl J Med 1979; 300: 1068–1073.
Weiden PL, Sullivan KM, Flournoy N, Storb R, Thomas ED . Antileukemic effect of chronic graft-versus-host disease: contribution to improved survival after allogeneic marrow transplantation. N Engl J Med 1981; 304: 1529–1533.
Kröger N, Zabelina T, Krüger W, Renges H, Stute N, Rischewski J et al. In-vivo T-cell depletion with pretransplant anti-thymocyte globulin reduces graft versus host disease without increasing relapse in good risk myeloid leukemia patients after stem cell transplantation from matched related donors. Bone Marrow Transplant 2002; 29: 683–689.
Cervantes F, Alvarez-Larran A, Hernandez-Boluda JC, Sureda A, Torrebadell M, Montserrat E . Erythropoietin treatment of the anaemia of myelofibrosis with myeloid metaplasia: results in 20 patients and review of the literature. Br J Haematol 2004; 127: 399–403.
Cervantes F, Alvarez-Larran A, Hernandez-Boluda JC, Sureda A, Granell M, Vallansot R et al. Darbepoetin-alpha for the anaemia of myelofibrosis with myeloid metaplasia. Br J Haematol 2006; 134: 184–186.
Barosi G, Grossi A, Comotti B, Musto P, Gamba G, Marchetti M . Safety and efficacy of thalidomide in patients with myelofibrosis with myeloid metaplasia. Br J Haematol 2001; 114: 78–83.
Mesa RA, Steensma DP, Pardanani A, Li CY, Elliott M, Kaufmann SH et al. A phase 2 trial of combination low-dose thalidomide and prednisone for the treatment of myelofibrosis with myeloid metaplasia. Blood 2003; 101: 2534–2541.
Wickramasinghe SN, Peart S, Gill DS . Alpha-interferon in primary idiopathic myelofibrosis (letter). Lancet 1987; 2: 1524–1525.
Tefferi A, Cortes J, Verstovsek S, Mesa RA, Thomas D, Lasho TL et al. Lenalidomide therapy in myelofibrosis with myeloid metaplasia. Blood 2006; 108: 1158–1164.
Lofvenberg E, Wahlin A . Management of polycythaemia vera, essential thrombocythaemia and myelofibrosis with hydroxyurea. Eur J Haematol 1988; 41: 375–381.
Manoharan A, Pitney WR . Chemotherapy resolves symptoms and reverses marrow fibrosis in myelofibrosis. Scand J Haematol 1984; 33: 453–459.
Petti MC, Latagliata R, Spadea T, Spadea A, Montefusco E, Aloe Spiriti MA et al. Melphalan treatment in patients with myelofibrosis with myeloid metaplasia. Br J Haematol 2002; 116: 576–581.
Najean Y, Rain JD . Treatment of polycythemia vera: use of 32P alone or in combination with maintenance therapy using hydroxyurea in 461 patients greater than 65 years of age. The French Polycythemia Study Group. Blood 1997; 89: 2319–2327.
Tefferi A, Silverstein MN, Li CY . 2-Chlorodeoxyadenosine treatment after splenectomy in patients who have myelofibrosis with myeloid metaplasia. Br J Haematol 1997; 99: 352–357.
Rodriguez JN, Martino ML, Dieguez JC, Prados D . rHuEpo for the treatment of anemia in myelofibrosis with myeloid metaplasia. Experience in 6 patients and meta-analytical approach. Haematologica 1998; 83: 616–621.
Besa E, Nowell P, Geller N, Gardner F . Analysis of the androgen response of 23 patients with agnogenic myeloid metaplasia: the value of chromosomal studies in prediciting response and survival. Cancer 1982; 49: 308.
Cervantes F, Alvarez-Larran A, Domingo A, Arellano-Rodrigo E, Montserrat E . Efficacy and tolerability of danazol as a treatment for the anaemia of myelofibrosis with myeloid metaplasia: long-term results in 30 patients. Br J Haematol 2005; 129: 771–775.
Battegay EJ, Raines EW, Colbert T, Ross R . TNF-alpha stimulation of fibroblast proliferation. Dependence on platelet-derived growth factor (PDGF) secretion and alteration of PDGF receptor expression. J Immunol 1995; 154: 6040–6047.
Steensma DP, Mesa RA, Li CY, Gray L, Tefferi A . Etanercept, a soluble tumor necrosis factor receptor, palliates constitutional symptoms in patients with myelofibrosis with myeloid metaplasia: results of a pilot study. Blood 2002; 99: 2252–2254.
Faoro LN, Tefferi A, Mesa RA . Long-term analysis of the palliative benefit of 2-chlorodeoxyadenosine for myelofibrosis with myeloid metaplasia. Eur J Haematol 2005; 74: 117–120.
Huang J, Xu J . Proliferation of human marrow fibroblasts suppressed synergistically by interferon alpha and TGF-β1 in vitro. Blood 2003; 102: Abstract no. 5077.
Carlo-Stella C, Cazzola M, Gasner A, Barosi G, Dezza L, Meloni F et al. Effects of recombinant alpha and gamma interferons on the in vitro growth of circulating hematopoietic progenitor cells (CFU-GEMM, CFU-Mk, BFU-E, and CFU-GM) from patients with myelofibrosis with myeloid metaplasia. Blood 1987; 70: 1014–1019.
Dalla KP, Zeigler ZR, Shadduck RK . Alpha-Interferon in myelofibrosis: a case report (see comments). Br J Haematol 1994; 86: 654–656.
Tefferi A, Elliot MA, Yoon SY, Li CY, Mesa RA, Call TG et al. Clinical and bone marrow effects of interferon alfa therapy in myelofibrosis with myeloid metaplasia. Blood 2001; 97: 1896.
Heis-Vahidi-Fard N, Forberg E, Eichinger S, Chott A, Lechner K, Gisslinger H . Ineffectiveness of interferon-gamma in the treatment of idiopathic myelofibrosis: a pilot study. Ann Hematol 2001; 80: 79–82.
Verstovsek S, Lawhorn K, Giles F, Cortes J, Thomas D, Garcia-Manero G et al. PEG-Intron for Myeloproliferative Diseases: an update of ongoing phase II study. Blood 2004; 104: Abstract no. 1517.
Mesa RA, Li CY, Ketterling RP, Schroeder GS, Knudson RA, Tefferi A . Leukemic transformation in myelofibrosis with myeloid metaplasia: a single-institution experience with 91 cases. Blood 2005; 105: 973–977.
Tefferi A, Lasho TL, Mesa RA, Pardanani A, Ketterling RP, Hanson CA . Lenalidomide therapy in del(5)(q31)-associated myelofibrosis: cytogenetic and JAK2V617F molecular remissions. Leukemia 2007; 21: 1827–1828.
Schafer PH, Gandhi AK, Loveland MA, Chen RS, Man HW, Schnetkamp PP et al. Enhancement of cytokine production and AP-1 transcriptional activity in T cells by thalidomide-related immunomodulatory drugs. J Pharmacol Exp Ther 2003; 305: 1222–1232.
Wood JM, Bold G, Buchdunger E, Cozens R, Ferrari S, Frei J et al. PTK787/ZK 222584, a novel and potent inhibitor of vascular endothelial growth factor receptor tyrosine kinases, impairs vascular endothelial growth factor-induced responses and tumor growth after oral administration. Cancer Res 2000; 60: 2178–2189.
Wagner-Ballon O, Gastinne T, Tulliez M, Lacout C, Pisani D, Chagraoui H et al. Proteasome inhibitor bortezomib can inhibit bone marrow fibrosis development in a murine model of myelofibrosis. Blood 2005; 106: Abstract no. 2582.
Lombardo LJ, Lee FY, Chen P, Norris D, Barrish JC, Behnia K et al. Discovery of N-(2-chloro-6-methyl- phenyl)-2-(6-(4-(2-hydroxyethyl)- piperazin-1-yl)-2-methylpyrimidin-4-ylamino)thiazole-5-carboxamide (BMS-354825), a dual Src/Abl kinase inhibitor with potent antitumor activity in preclinical assays. J Med Chem 2004; 47: 6658–6661.
Yingling JM, Blanchard KL, Sawyer JS . Development of TGF-beta signalling inhibitors for cancer therapy. Nat Rev Drug Discov 2004; 3: 1011–1022.
Cardones AR, Banez LL . VEGF inhibitors in cancer therapy. Curr Pharm Des 2006; 12: 387–394.
O’Brien S, Kipps TJ, Faderl S, Crump M, Keating MJ, Anderson B et al. A phase I trial of the small molecule Pan-Bcl-2 family inhibitor GX15-070 administered intravenously (IV) every 3 weeks to patients with previously treated chronic lymphocytic leukemia (CLL). Blood 2005; 106: Abstract no. 446.
Issa JP, Gharibyan V, Cortes J, Jelinek J, Morris G, Verstovsek S et al. Phase II study of low-dose decitabine in patients with chronic myelogenous leukemia resistant to imatinib mesylate. J Clin Oncol 2005; 23: 3948–3956.
Giles FJ, List AF, Carroll M, Cortes JE, Valickas J, Chen BL et al. PTK787/ZK 222584, a small molecule tyrosine kinase receptor inhibitor of vascular endothelial growth factor (VEGF), has modest activity in myelofibrosis with myeloid metaplasia. Leu Res 2007; 31: 891–897.
Giles FJ, Cooper MA, Silverman L, Karp JE, Lancet JE, Zangari M et al. Phase II study of SU5416—a small-molecule, vascular endothelial growth factor tyrosine-kinase receptor inhibitor—in patients with refractory myeloproliferative diseases. Cancer 2003; 97: 1920–1928.
Mesa RA, Tefferi A, Elliott MA, Hoagland HC, Call TG, Schroeder GS et al. A phase II trial of pirfenidone (5-methyl-1-phenyl-2-[1H]-pyridone), a novel anti-fibrosing agent, in myelofibrosis with myeloid metaplasia. Br J Haematol 2001; 114: 111–113.
Schlingensiepen KH, Bischof A, Egger T, Hafner M, Herrmuth H, Jachimczak P et al. The TGF-Beta1 antisense oligonucleotide AP 11014 for the treatment of non-small cell lung, colorectal and prostate cancer: preclinical studies. J Clin Oncol 2004; 22 (14S): 2004 ASCO Annual Meeting Proceedings (Post-Meeting Edition); Abstract no. 3132.
Sawyer JS, Beight DW, Britt KS, Anderson BD, Campbell RM, Goodson Jr T et al. Synthesis and activity of new aryl- and heteroaryl-substituted 5,6-dihydro-4H-pyrrolo[1,2-b]pyrazole inhibitors of the transforming growth factor-beta type I receptor kinase domain. Bioorg Med Chem Lett 2004; 14: 3581–3584.
Gellibert F, Woolven J, Fouchet MH, Mathews N, Goodland H, Lovegrove V et al. Identification of 1,5-naphthyridine derivatives as a novel series of potent and selective TGF-beta type I receptor inhibitors. J Med Chem 2004; 47: 4494–4506.
Callahan JF, Burgess JL, Fornwald JA, Gaster LM, Harling JD, Harrington FP et al. Identification of novel inhibitors of the transforming growth factor beta1 (TGF-beta1) type 1 receptor (ALK5). J Med Chem 2002; 45: 999–1001.
Uhl M, Aulwurm S, Wischhusen J, Weiler M, Ma JY, Almirez R et al. SD-208, a novel transforming growth factor beta receptor I kinase inhibitor, inhibits growth and invasiveness and enhances immunogenicity of murine and human glioma cells in vitro and in vivo. Cancer Res 2004; 64: 7954–7961.
Kvasnicka HM, Thiele J . Bone marrow angiogenesis: methods of quantification and changes evolving in chronic myeloproliferative disorders. Histol Histopathol 2004; 19: 1245–1260.
Tefferi A, Mesa RA, Gray LA, Steensma DP, Camoriano JK, Elliott MA et al. Phase 2 trial of imatinib mesylate in myelofibrosis with myeloid metaplasia. Blood 2002; 99: 3854–3856.
Cortes J, Ault P, Koller C, Thomas D, Ferrajoli A, Wierda W et al. Efficacy of imatinib mesylate in the treatment of idiopathic hypereosinophilic syndrome. Blood 2003; 101: 4714–4716.
Cortes J, Albitar M, Thomas D, Giles F, Kurzrock R, Thibault A et al. Efficacy of the farnesyl transferase inhibitor R115777 in chronic myeloid leukemia and other hematologic malignancies. Blood 2003; 101: 1692–1697.
Mesa RA, Camoriano JK, Geyer SM, Wu W, Kaufmann SH, Rivera CE et al. A phase II trial of tipifarnib in myelofibrosis: primary, post-polycythemia vera and post-essential thrombocythemia. Leukemia 2007; 21: 1964–1970.
Baylin SB, Herman JG, Graff JR, Vertino PM, Issa JP . Alterations in DNA methylation: a fundamental aspect of neoplasia. Adv Cancer Res 1998; 72: 141–196.
Martyre MC, Steunou V, Le Bousse-Kerdiles MC, Wietzerbin J . Lack of alteration in GATA-1 expression in CD34+ hematopoietic progenitors from patients with idiopathic myelofibrosis (comment). Blood 2003; 101: 5087–5088; author reply 88–89.
Pardanini A . JAK2 inhibitor therapy in myeloproliferative disorders: rationale, preclinical studies and ongoing clinical trials. Leukemia 2007, 20 Sept; e-pub ahead of print.
Grandage VL, Everington T, Linch DC, Khwaja A . Go6976 is a potent inhibitor of the JAK 2 and FLT3 tyrosine kinases with significant activity in primary acute myeloid leukaemia cells. Br J Haematol 2006; 135: 303–316.
Li Z, Xu M, Xing S, Ho WT, Ishii T, Li Q et al. Erlotinib effectively inhibits JAK2V617F activity and polycythemia vera cell growth. J Biol Chem 2007; 282: 3428–3432.
Giles F, Bergstrom DA, Garcia-Manero G, Kornblau S, Andreeff M, Kantarjian H et al. MK-0457 is a novel aurora kinase and Janus kinase 2 (JAK2) inhibitor with activity in transformed JAK2-positive myeloproliferative disease (MPD). Blood 2006; 108: Abstract no. 4893.
Dobrzanski P, Hexner E, Serdikoff C, Mahfuza J, Swider C, Robinson C et al. CEP-701 is a JAK2 inhibitor which attenuates JAK2/STAT5 signaling pathway and the proliferation of primary cells from patients with myeloproliferative disorders. Blood 2006; 108: Abstract no. 3594.
Scott LM, Tong W, Levine RL, Scott MA, Beer PA, Stratton MR et al. JAK2 exon 12 mutations in polycythemia vera and idiopathic erythrocytosis. N Engl J Med 2007; 356: 459–468.
Dupriez B, Morel P, Demory JL, Lai JL, Simon M, Plantier I et al. Prognostic factors in agnogenic myeloid metaplasia: a report on 195 cases with a new scoring system. Blood 1996; 88: 1013–1018.
Elliot MA, Verstovsek S, Dingli D, Schwager SM, Mesa RA, Li CY et al. Monocytois is an adverse prognostic factor for survival in younger patients with primary myelofibrosis. Leuk Res 2007; 31: 1503–1509.
Tanimoto TE, Shimoda K, Yamaguchi T, Okamura T, Mizoguchi H, Omine M et al. Prognostic factors in primary chronic myelofibrosis in patients aged less than 70 years: a report on 207 patients with the description of a scoring system and its validation on 100 other patients. Blood 2004; 104: Abstract no. 1524.
Kvasnicka HM, Thiele J, Werden C, Zankovich R, Diehl V, Fischer R . Prognostic factors in idiopathic (primary) osteo-myelofibrosis. Cancer 1997; 80: 708–719.
Thiele J, Kvasnicka HM . Hematopathologic findings in chronic idiopathic myelofibrosis. Semin Oncol 2005; 32: 380–394.
Tefferi A, Dingli D, Li CY, Dewald GW . Prognostic diversity among cytogenetic abnormalities in myelofibrosis with myeloid metaplasia. Cancer 2005; 104: 1656–1660.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005; 106: 2912–2919.
Acknowledgements
We thank the staff of the clinics for providing excellent care of our patients and the medical technicians for their excellent work in the laboratories and Richard Brummel for proof editing.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kröger, N., Mesa, R. Choosing between stem cell therapy and drugs in myelofibrosis. Leukemia 22, 474–486 (2008). https://doi.org/10.1038/sj.leu.2405080
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2405080
Keywords
This article is cited by
-
Magnetic resonance enterography for assessment of intestinal graft-versus-host disease after allogeneic stem cell transplantation
European Radiology (2015)
-
18F-FDG PET/CT for Detection of Metachronous Hodgkin’s Disease in Patients with Myelofibrosis
Nuclear Medicine and Molecular Imaging (2015)
-
Assessment of bone marrow inflammation in patients with myelofibrosis: an 18F-fluorodeoxyglucose PET/CT study
European Journal of Nuclear Medicine and Molecular Imaging (2015)
-
How to manage the transplant question in myelofibrosis
Blood Cancer Journal (2012)
-
Refined cytogenetic-risk categorization for overall and leukemia-free survival in primary myelofibrosis: a single center study of 433 patients
Leukemia (2011)