Abstract
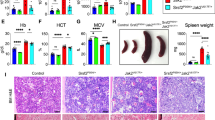
An acquired JAK2 V617F mutation is found in most patients with polycythemia vera (PV), and about half of patients with essential thrombocythemia (ET) or primary myelofibrosis (PMF). Mice transplanted with bone marrow cells in which JAK2 V617F was retrovirally expressed developed PV-like features, but not ET or PMF. To address the contribution of this mutation to the pathogenesis of these three MPDs, we generated two lines of JAK2 V617F transgenic mice. One line showed granulocytosis after 4 months of age. Among 43 mice, 8 (19%) showed polycythemia and 15 (35%) showed thrombocythemia. The second line showed extreme leukocytosis and thromobocytosis. They showed anemia that means Hb value from 9 to 10 g per 100 ml when 1 month old. Myeloid cells and megakaryocytes were predominant in the bone marrow of these animals, and splenomegaly was observed. The expression of JAK2 V617F mRNA in bone marrow cells was 0.45 and 1.35 that of endogenous wild-type JAK2 in the two lines, respectively. In vitro analysis of bone marrow cells from both lines showed constitutive activation of ERK1/2, STAT5 and AKT, and augmentation of their phosphorylations by cytokine stimulation. We conclude that in vivo expression of JAK2 V617F results in ET-, PMF- and PV-like disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Witthuhn BA, Quelle FW, Silvennoinen O, Yi T, Tang B, Miura O et al. JAK2 associates with the erythropoietin receptor and is tyrosine phosphorylated and activated following stimulation with erythropoietin. Cell 1993; 74: 227–236.
Parganas E, Wang D, Stravopodis D, Topham DJ, Marine JC, Teglund S et al. Jak2 is essential for signaling through a variety of cytokine receptors. Cell 1998; 93: 385–395.
Ihle JN . Cytokine receptor signalling. Nature 1995; 377: 591–594.
Ihle JN . STATs: signal transducers and activators of transcription. Cell 1996; 84: 331–334.
Ihle JN, Nosaka T, Thierfelder W, Quelle FW, Shimoda K . Jaks and Stats in cytokine signaling. Stem Cells 1997; 15 (Suppl 1): 105–111; discussion 112.
Wu H, Liu X, Jaenisch R, Lodish HF . Generation of committed erythroid BFU-E and CFU-E progenitors does not require erythropoietin or the erythropoietin receptor. Cell 1995; 83: 59–67.
Cui Y, Riedlinger G, Miyoshi K, Tang W, Li C, Deng CX et al. Inactivation of Stat5 in mouse mammary epithelium during pregnancy reveals distinct functions in cell proliferation, survival, and differentiation. Mol Cell Biol 2004; 24: 8037–8047.
Baxter EJ, Scott LM, Campbell PJ, East C, Fourouclas N, Swanton S et al. Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet 2005; 365: 1054–1061.
James C, Ugo V, Le Couedic JP, Staerk J, Delhommeau F, Lacout C et al. A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemia vera. Nature 2005; 434: 1144–1148.
Kralovics R, Passamonti F, Buser AS, Teo SS, Tiedt R, Passweg JR et al. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med 2005; 352: 1779–1790.
Levine RL, Wadleigh M, Cools J, Ebert BL, Wernig G, Huntly BJ et al. Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocythemia, and myeloid metaplasia with myelofibrosis. Cancer Cell 2005; 7: 387–397.
Jones AV, Kreil S, Zoi K, Waghorn K, Curtis C, Zhang L et al. Widespread occurrence of the JAK2 V617F mutation in chronic myeloproliferative disorders. Blood 2005; 106: 2162–2168.
Shide K, Shimoda K, Kamezaki K, Kakumitsu H, Kumano T, Numata A et al. Tyk2 mutation homologous to V617F Jak2 is not found in essential thrombocythaemia, although it induces constitutive signaling and growth factor independence. Leuk Res 2007; 31: 1085–1092.
Dameshek W . Some speculations on the myeloproliferative syndromes. Blood 1951; 6: 372–375.
Reeder TL, Bailey RJ, Dewald GW, Tefferi A . Both B and T lymphocytes may be clonally involved in myelofibrosis with myeloid metaplasia. Blood 2003; 101: 1981–1983.
Prchal JT, Guan YL, Prchal JF, Barany F . Transcriptional analysis of the active X-chromosome in normal and clonal hematopoiesis. Blood 1993; 81: 269–271.
Prchal JF, Axelrad AA . Letter: Bone-marrow responses in polycythemia vera. N Engl J Med 1974; 290: 1382.
Reid CD . The significance of endogenous erythroid colonies (EEC) in haematological disorders. Blood Rev 1987; 1: 133–140.
Tefferi A, Thiele J, Orazi A, Kvasnicka HM, Barbui T, Hanson CA et al. Proposals and rationale for revision of the World Health Organization diagnostic criteria for polycythemia vera, essential thrombocythemia, and primary myelofibrosis: recommendations from an ad hoc international expert panel. Blood 2007; 110: 1092–1097.
Wernig G, Mercher T, Okabe R, Levine RL, Lee BH, Gilliland DG . Expression of Jak2V617F causes a polycythemia vera-like disease with associated myelofibrosis in a murine bone marrow transplant model. Blood 2006; 107: 4274–4281.
Lacout C, Pisani DF, Tulliez M, Gachelin FM, Vainchenker W, Villeval JL . JAK2V617F expression in murine hematopoietic cells leads to MPD mimicking human PV with secondary myelofibrosis. Blood 2006; 108: 1652–1660.
Bumm TG, Elsea C, Corbin AS, Loriaux M, Sherbenou D, Wood L et al. Characterization of murine JAK2V617F-positive myeloproliferative disease. Cancer Res 2006; 66: 11156–11165.
Zaleskas VM, Krause DS, Lazarides K, Patel N, Hu Y, Li S et al. Molecular pathogenesis and therapy of polycythemia induced in mice by JAK2 V617F. PLoS ONE 2006; 1: e18.
Domen J, Gandy KL, Weissman IL . Systemic overexpression of BCL-2 in the hematopoietic system protects transgenic mice from the consequences of lethal irradiation. Blood 1998; 91: 2272–2282.
Kakumitsu H, Kamezaki K, Shimoda K, Karube K, Haro T, Numata A et al. Transgenic mice overexpressing murine thrombopoietin develop myelofibrosis and osteosclerosis. Leuk Res 2005; 29: 761–769.
Riley J, Butler R, Ogilvie D, Finniear R, Jenner D, Powell S et al. A novel, rapid method for the isolation of terminal sequences from yeast artificial chromosome (YAC) clones. Nucleic Acids Res 1990; 18: 2887–2890.
Schessl C, Rawat VP, Cusan M, Deshpande A, Kohl TM, Rosten PM et al. The AML1-ETO fusion gene and the FLT3 length mutation collaborate in inducing acute leukemia in mice. J Clin Invest 2005; 115: 2159–2168.
Noguchi A, Takekawa N, Einarsdottir T, Koura M, Noguchi Y, Takano K et al. Chromosomal mapping and zygosity check of transgenes based on flanking genome sequences determined by genomic walking. Exp Anim 2004; 53: 103–111.
Shimoda K, Feng J, Murakami H, Nagata S, Watling D, Rogers NC et al. Jak1 plays an essential role for receptor phosphorylation and Stat activation in response to granulocyte colony-stimulating factor. Blood 1997; 90: 597–604.
Lu X, Levine R, Tong W, Wernig G, Pikman Y, Zarnegar S et al. Expression of a homodimeric type I cytokine receptor is required for JAK2V617F-mediated transformation. Proc Natl Acad Sci USA 2005; 102: 18962–18967.
Kralovics R, Teo SS, Li S, Theocharides A, Buser AS, Tichelli A et al. Acquisition of the V617F mutation of JAK2 is a late genetic event in a subset of patients with myeloproliferative disorders. Blood 2006; 108: 1377–1380.
Andrieux J, Demory JL, Dupriez B, Quief S, Plantier I, Roumier C et al. Dysregulation and overexpression of HMGA2 in myelofibrosis with myeloid metaplasia. Genes Chromosomes Cancer 2004; 39: 82–87.
Komura E, Chagraoui H, Mansat de Mas V, Blanchet B, de Sepulveda P, Larbret F et al. Spontaneous STAT5 activation induces growth factor independence in idiopathic myelofibrosis: possible relationship with FKBP51 overexpression. Exp Hematol 2003; 31: 622–630.
Jones LC, Tefferi A, Idos GE, Kumagai T, Hofmann WK, Koeffler HP . RARbeta2 is a candidate tumor suppressor gene in myelofibrosis with myeloid metaplasia. Oncogene 2004; 23: 7846–7853.
Chagraoui H, Komura E, Tulliez M, Giraudier S, Vainchenker W, Wendling F . Prominent role of TGF-beta 1 in thrombopoietin-induced myelofibrosis in mice. Blood 2002; 100: 3495–3503.
Roberts AB, Sporn MB . Physiological actions and clinical applications of transforming growth factor-beta (TGF-beta). Growth Factors 1993; 8: 1–9.
Chagraoui H, Tulliez M, Smayra T, Komura E, Giraudier S, Yun T et al. Stimulation of osteoprotegerin production is responsible for osteosclerosis in mice overexpressing TPO. Blood 2003; 101: 2983–2989.
Yan XQ, Lacey D, Fletcher F, Hartley C, McElroy P, Sun Y et al. Chronic exposure to retroviral vector encoded MGDF (mpl-ligand) induces lineage-specific growth and differentiation of megakaryocytes in mice. Blood 1995; 86: 4025–4033.
Acknowledgements
We thank Dr N Watanabe (Department of Hematopoietic Factors, Institute of Medical Science, the University of Tokyo) for helpful advice and discussion concerning bubble LM-PCR. We also thank M Matsushita, K Tsugura and K Toyama for their excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shide, K., Shimoda, H., Kumano, T. et al. Development of ET, primary myelofibrosis and PV in mice expressing JAK2 V617F. Leukemia 22, 87–95 (2008). https://doi.org/10.1038/sj.leu.2405043
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2405043
Keywords
This article is cited by
-
Insufficiency of non-canonical PRC1 synergizes with JAK2V617F in the development of myelofibrosis
Leukemia (2022)
-
Circulating CD34+ cells of primary myelofibrosis patients contribute to myeloid-dominant hematopoiesis and bone marrow fibrosis in immunodeficient mice
International Journal of Hematology (2022)
-
Neoplastic fibrocytes play an essential role in bone marrow fibrosis in Jak2V617F-induced primary myelofibrosis mice
Leukemia (2021)
-
Clonal hematopoiesis with JAK2V617F promotes pulmonary hypertension with ALK1 upregulation in lung neutrophils
Nature Communications (2021)
-
Bone: a key aspect to understand phenomena in clinical hematology
Journal of Bone and Mineral Metabolism (2020)