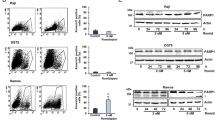
Abstract
Several histone deacetylase inhibitors (HDACi), which have recently entered early clinical trials, exert their anticancer activity in part through the induction of apoptosis although the precise mechanism of this induction is not known. Induction of apoptosis by structurally diverse HDACi in primary cells from patients with chronic lymphocytic leukemia (CLL) and different leukemic cell lines was mediated by the Bcl-2 regulated intrinsic pathway and demonstrated a requirement for de novo protein synthesis. A marked time-dependent induction of the pro-apoptotic BH3-only proteins, Bim, Noxa and Bmf was observed, which preceded the induction of apoptosis. A key role for both Bim and Noxa was proposed in HDACi-mediated apoptosis based on our findings that siRNA for Bim and Noxa but not Bmf largely prevented the HDACi-induced loss in mitochondrial membrane potential, caspase processing and phosphatidylserine externalization. Noxa, induced by HDACi, in CLL cells and tumor cell lines, bound extensively to Mcl-1, a major anti-apoptotic Bcl-2 family member present in CLL cells. Our data strongly suggests that HDACi induce apoptosis primarily through inactivation of anti-apoptotic Bcl-2 family members by increases in Bim and Noxa and highlights these increases as a potential clinical target for CLL/lymphoma therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Marks P, Rifkind RA, Richon VM, Breslow R, Miller T, Kelly WK . Histone deacetylases and cancer: causes and therapies. Nat Rev Cancer 2001; 1: 194–202.
Minucci S, Pelicci PG . Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat Rev Cancer 2006; 6: 38–51.
Rosato RR, Grant S . Histone deacetylase inhibitors: insights into mechanisms of lethality. Expert Opin Ther Targets 2005; 9: 809–824.
Strasser A . The role of BH3-only proteins in the immune system. Nat Rev Immunol 2005; 5: 189–200.
Cory S, Adams JM . The Bcl2 family: regulators of the cellular life-or-death switch. Nat Rev Cancer 2002; 2: 647–656.
Huang DC, Strasser A . BH3-Only proteins-essential initiators of apoptotic cell death. Cell 2000; 103: 839–842.
Willis SN, Adams JM . Life in the balance: how BH3-only proteins induce apoptosis. Curr Opin Cell Biol 2005; 17: 617–625.
Labi V, Erlacher M, Kiessling S, Villunger A . BH3-only proteins in cell death initiation, malignant disease and anticancer therapy. Cell Death Differ 2006; 13: 1325–1338.
Chen L, Willis SN, Wei A, Smith BJ, Fletcher JI, Hinds MG et al. Differential targeting of prosurvival Bcl-2 proteins by their BH3-only ligands allows complementary apoptotic function. Mol Cell 2005; 17: 393–403.
Willis SN, Chen L, Dewson G, Wei A, Naik E, Fletcher JI et al. Proapoptotic Bak is sequestered by Mcl-1 and Bcl-xL, but not Bcl-2, until displaced by BH3-only proteins. Genes Dev 2005; 19: 1294–1305.
Ruefli AA, Ausserlechner MJ, Bernhard D, Sutton VR, Tainton KM, Kofler R et al. The histone deacetylase inhibitor and chemotherapeutic agent suberoylanilide hydroxamic acid (SAHA) induces a cell-death pathway characterized by cleavage of Bid and production of reactive oxygen species. Proc Natl Acad Sci USA 2001; 98: 10833–10838.
Henderson C, Mizzau M, Paroni G, Maestro R, Schneider C, Brancolini C . Role of caspases, Bid, and p53 in the apoptotic response triggered by histone deacetylase inhibitors trichostatin-A (TSA) and suberoylanilide hydroxamic acid (SAHA). J Biol Chem 2003; 278: 12579–12589.
Zhang Y, Adachi M, Zhao X, Kawamura R, Imai K . Histone deacetylase inhibitors FK228, N-(2-aminophenyl)-4-[N-(pyridin-3-yl-methoxycarbonyl)amino- methyl]benzamide and m-carboxycinnamic acid bis-hydroxamide augment radiation-induced cell death in gastrointestinal adenocarcinoma cells. Int J Cancer 2004; 110: 301–308.
Inoue S, MacFarlane M, Harper N, Wheat LM, Dyer MJ, Cohen GM . Histone deacetylase inhibitors potentiate TNF-related apoptosis-inducing ligand (TRAIL)-induced apoptosis in lymphoid malignancies. Cell Death Differ 2004; 11: S193–S206.
Zhang Y, Adachi M, Kawamura R, Imai K . Bmf is a possible mediator in histone deacetylase inhibitors FK228 and CBHA-induced apoptosis. Cell Death Differ 2006; 13: 129–140.
Zhang Y, Adachi M, Kawamura R, Zou HC, Imai K, Hareyama M et al. Bmf contributes to histone deacetylase inhibitor-mediated enhancing effects on apoptosis after ionizing radiation. Apoptosis 2006; 11: 1349–1357.
Zhao Y, Tan J, Zhuang L, Jiang X, Liu ET, Yu Q . Inhibitors of histone deacetylases target the Rb-E2F1 pathway for apoptosis induction through activation of proapoptotic protein Bim. Proc Natl Acad Sci USA 2005; 102: 16090–16095.
Glick RD, Swendeman SL, Coffey DC, Rifkind RA, Marks PA, Richon VM et al. Hybrid polar histone deacetylase inhibitor induces apoptosis and CD95/CD95 ligand expression in human neuroblastoma. Cancer Res 1999; 59: 4392–4399.
Medina V, Edmonds B, Young GP, James R, Appleton S, Zalewski PD . Induction of caspase-3 protease activity and apoptosis by butyrate and trichostatin A (inhibitors of histone deacetylase): dependence on protein synthesis and synergy with a mitochondrial/cytochrome c-dependent pathway. Cancer Res 1997; 57: 3697–3707.
Rosato RR, Almenara JA, Grant S . The histone deacetylase inhibitor MS-275 promotes differentiation or apoptosis in human leukemia cells through a process regulated by generation of reactive oxygen species and induction of p21CIP1/WAF1 1. Cancer Res 2003; 63: 3637–3645.
Lucas DM, Davis ME, Parthun MR, Mone AP, Kitada S, Cunningham KD et al. The histone deacetylase inhibitor MS-275 induces caspase-dependent apoptosis in B-cell chronic lymphocytic leukemia cells. Leukemia 2004; 18: 1207–1214.
Aron JL, Parthun MR, Marcucci G, Kitada S, Mone AP, Davis ME et al. Depsipeptide (FR901228) induces histone acetylation and inhibition of histone deacetylase in chronic lymphocytic leukemia cells concurrent with activation of caspase 8-mediated apoptosis and down-regulation of c-FLIP protein. Blood 2003; 102: 652–658.
MacFarlane M, Harper N, Snowden RT, Dyer MJ, Barnett GA, Pringle JH et al. Mechanisms of resistance to TRAIL-induced apoptosis in primary B cell chronic lymphocytic leukaemia. Oncogene 2002; 21: 6809–6818.
Inoue S, Mai A, Dyer MJ, Cohen GM . Inhibition of histone deacetylase class I but not class II is critical for the sensitization of leukemic cells to tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis. Cancer Res 2006; 66: 6785–6792.
Inoue S, Twiddy D, Dyer MJ, Cohen GM . Upregulation of TRAIL-R2 is not involved in HDACi mediated sensitization to TRAIL-induced apoptosis. Cell Death Differ 2006; 13: 2160–2162.
MacFarlane M, Inoue S, Kohlhaas SL, Majid A, Harper N, Kennedy DB et al. Chronic lymphocytic leukemic cells exhibit apoptotic signaling via TRAIL-R1. Cell Death Differ 2005; 12: 773–782.
MacFarlane M, Kohlhaas SL, Sutcliffe MJ, Dyer MJ, Cohen GM . TRAIL Receptor-Selective Mutants Signal to Apoptosis via TRAIL-R1 in Primary Lymphoid Malignancies. Cancer Res 2005; 65: 11265–11270.
Estrov Z, Talpaz M, Ku S, Harris D, Van Q, Beran M et al. Z-138: a new mature B-cell acute lymphoblastic leukemia cell line from a patient with transformed chronic lymphocytic leukemia. Leuk Res 1998; 22: 341–353.
Ohgushi M, Kuroki S, Fukamachi H, O’Reilly LA, Kuida K, Strasser A et al. Transforming growth factor beta-dependent sequential activation of Smad, Bim, and caspase-9 mediates physiological apoptosis in gastric epithelial cells. Mol Cell Biol 2005; 25: 10017–10028.
Kawai Y, Arinze IJ . Valproic acid-induced gene expression through production of reactive oxygen species. Cancer Res 2006; 66: 6563–6569.
Qian DZ, Kato Y, Shabbeer S, Wei Y, Verheul HM, Salumbides B et al. Targeting tumor angiogenesis with histone deacetylase inhibitors: the hydroxamic acid derivative LBH589. Clin Cancer Res 2006; 12: 634–642.
Zhang XD, Gillespie SK, Borrow JM, Hersey P . The histone deacetylase inhibitor suberic bishydroxamate regulates the expression of multiple apoptotic mediators and induces mitochondria-dependent apoptosis of melanoma cells. Mol Cancer Ther 2004; 3: 425–435.
Duan H, Heckman CA, Boxer LM . Histone deacetylase inhibitors down-regulate bcl-2 expression and induce apoptosis in t(14;18) lymphomas. Mol Cell Biol 2005; 25: 1608–1619.
Nijhawan D, Fang M, Traer E, Zhong Q, Gao W, Du F et al. Elimination of Mcl-1 is required for the initiation of apoptosis following ultraviolet irradiation. Genes Dev 2003; 17: 1475–1486.
Brimmell M, Mendiola R, Mangion J, Packham G . BAX frameshift mutations in cell lines derived from human haemopoietic malignancies are associated with resistance to apoptosis and microsatellite instability. Oncogene 1998; 16: 1803–1812.
Cuconati A, Mukherjee C, Perez D, White E . DNA damage response and MCL-1 destruction initiate apoptosis in adenovirus-infected cells. Genes Dev 2003; 17: 2922–2932.
Byrd JC, Shinn C, Ravi R, Willis CR, Waselenko JK, Flinn IW et al. Depsipeptide (FR901228): a novel therapeutic agent with selective, in vitro activity against human B-cell chronic lymphocytic leukemia cells. Blood 1999; 94: 1401–1408.
Smit LA, Hallaert DY, Spijker R, de Goeij B, Jaspers A, Kater AP et al. Differential Noxa/Mcl-1 balance in peripheral versus lymph node chronic lymphocitic leukemia cells correlates with survival capacity. Blood 2007; 109: 1660–1668.
MacFarlane M, Cohen GM, Dickens M . JNK (c-Jun N-terminal kinase) and p38 activation in receptor-mediated and chemically-induced apoptosis of T-cells: differential requirements for caspase activation. Biochem J 2000; 348: 93–101.
Xu W, Ngo L, Perez G, Dokmanovic M, Marks PA . Intrinsic apoptotic and thioredoxin pathways in human prostate cancer cell response to histone deacetylase inhibitor. Proc Natl Acad Sci USA 2006; 103: 15540–15545.
Facchetti F, Previdi S, Ballarini M, Minucci S, Perego P, La Porta CA . Modulation of pro- and anti-apoptotic factors in human melanoma cells exposed to histone deacetylase inhibitors. Apoptosis 2004; 9: 573–582.
Su AI, Wiltshire T, Batalov S, Lapp H, Ching KA, Block D et al. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc Natl Acad Sci USA 2004; 101: 6062–6067.
Mackus WJ, Kater AP, Grummels A, Evers LM, Hooijbrink B, Kramer MH et al. Chronic lymphocytic leukemia cells display p53-dependent drug-induced Puma upregulation. Leukemia 2005; 19: 427–434.
Oda E, Ohki R, Murasawa H, Nemoto J, Shibue T, Yamashita T et al. Noxa, a BH3-only member of the Bcl-2 family and candidate mediator of p53-induced apoptosis. Science 2000; 288: 1053–1058.
Villunger A, Michalak EM, Coultas L, Mullauer F, Bock G, Ausserlechner MJ et al. p53- and drug-induced apoptotic responses mediated by BH3-only proteins puma and noxa. Science 2003; 302: 1036–1038.
Flinterman M, Guelen L, Ezzati-Nik S, Killick R, Melino G, Tominaga K et al. E1A activates transcription of p73 and Noxa to induce apoptosis. J Biol Chem 2005; 280: 5945–5959.
Hershko T, Ginsberg D . Up-regulation of Bcl-2 homology 3 (BH3)-only proteins by E2F1 mediates apoptosis. J Biol Chem 2004; 279: 8627–8634.
Fernandez Y, Verhaegen M, Miller TP, Rush JL, Steiner P, Opipari Jr AW et al. Differential regulation of noxa in normal melanocytes and melanoma cells by proteasome inhibition: therapeutic implications. Cancer Res 2005; 65: 6294–6304.
Perez-Galan P, Roue G, Villamor N, Montserrat E, Campo E, Colomer D . The proteasome inhibitor bortezomib induces apoptosis in mantle-cell lymphoma through generation of ROS and Noxa activation independent of p53 status. Blood 2006; 107: 257–264.
Qin JZ, Ziffra J, Stennett L, Bodner B, Bonish BK, Chaturvedi V et al. Proteasome inhibitors trigger NOXA-mediated apoptosis in melanoma and myeloma cells. Cancer Res 2005; 65: 6282–6293.
Seo YW, Shin JN, Ko KH, Cha JH, Park JY, Lee BR et al. The molecular mechanism of Noxa-induced mitochondrial dysfunction in p53-mediated cell death. J Biol Chem 2003; 278: 48292–48299.
Rampino N, Yamamoto H, Ionov Y, Li Y, Sawai H, Reed JC et al. Somatic frameshift mutations in the BAX gene in colon cancers of the microsatellite mutator phenotype. Science 1997; 275: 967–969.
Kitada S, Andersen J, Akar S, Zapata JM, Takayama S, Krajewski S et al. Expression of apoptosis-regulating proteins in chronic lymphocytic leukemia: correlations with In vitro and In vivo chemoresponses. Blood 1998; 91: 3379–3389.
Luo X, Budihardjo I, Zou H, Slaughter C, Wang X . Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 1998; 94: 481–490.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Leukemia Web site (http://www.nature.com/leu)
Rights and permissions
About this article
Cite this article
Inoue, S., Riley, J., Gant, T. et al. Apoptosis induced by histone deacetylase inhibitors in leukemic cells is mediated by Bim and Noxa. Leukemia 21, 1773–1782 (2007). https://doi.org/10.1038/sj.leu.2404760
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404760
Keywords
This article is cited by
-
BCL-2 protein family: attractive targets for cancer therapy
Apoptosis (2023)
-
Identification of NOXA as a pivotal regulator of resistance to CAR T-cell therapy in B-cell malignancies
Signal Transduction and Targeted Therapy (2022)
-
The combination of CUDC-907 and gilteritinib shows promising in vitro and in vivo antileukemic activity against FLT3-ITD AML
Blood Cancer Journal (2021)
-
Targeting multiple signaling pathways: the new approach to acute myeloid leukemia therapy
Signal Transduction and Targeted Therapy (2020)
-
NOXA-dependent contextual synthetic lethality of BCL-XL inhibition and “osmotic reprogramming” in colorectal cancer
Cell Death & Disease (2020)