Abstract
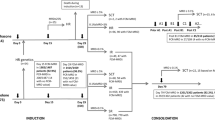
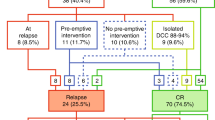
Most patients with acute myeloid leukemia (AML) and t(8;21) or inv(16) have a good prognosis with current anthracycline- and cytarabine-based protocols. Tandem analysis with flow cytometry (FC) and real-time RT-PCR (RQ-PCR) was applied to 55 patients, 28 harboring a t(8;21) and 27 an inv(16), including one case with a novel CBFbeta/MYH11 transcript. A total of 31% (n=17) of CR patients relapsed: seven with t(8;21) and 10 with inv(16). The mean amount of minimal residual disease (MRD) detected by FC in relapsed and nonrelapsed patients was markedly different: 0.3 vs 0.08% (P=0.002) at the end of treatment. The mean number of fusion transcript copies/ABLx104 also differed between relapsed and non-relapsed patients: 2385 vs 122 (P=0.001) after induction, 56 vs 7.6 after intensification (P=0.0001) and 75 vs 3.3 (P=0.0001) at the end of chemotherapy. Relapses were more common in patients with FC MRD level >0.1% at the end of treatment than in patients with ⩽0.1%: cumulative incidence of relapse (CIR) was 67 and 21% (P=0.03), respectively. Likewise, using RQ-PCR, a cutoff level of >10 copies at the end of treatment correlated with a high risk of relapse: CIR was 75% for patients with RQ-PCR >10 compared to 21% for patients with RQ-PCR levels ⩽10 (P=0.04). Combined use of FC and RQ-PCR may improve MRD detection, and provide useful clinical information on relapse kinetics in AML patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Grimwade D, Walker H, Oliver F, Wheatley K, Harrison C, Harrison G et al. The importance of diagnostic cytogenetics on outcome in AML: analysis of 1,612 patients entered into the MRC AML 10 trial. Blood 1998; 92: 2322–2333.
Baer MR, Stewart CC, Lawrence D, Arthur DC, Byrd JC, Davey FR et al. Expression of the neural cell adhesion molecule CD56 is associated with short remission duration and survival in acute myeloid leukemia with t(8;21)(q22;q22). Blood 1997; 90: 1643–1648.
Nguyen S, Leblanc T, Fenaux P, Witz F, Blaise D, Pigneux A et al. A white blood cell index as the main prognostic factor in t(8;21) acute myeloid leukemia (AML): a survey of 161 cases from the French AML Intergroup. Blood 2002; 99: 3517–3523.
Delaunay J, Vey N, Leblanc T, Fenaux P, Rigal-Huguet F, Witz F et al. Prognosis of inv(16)/t(16;16) acute myeloid leukemia (AML): a survey of 110 cases from the French AML Intergroup. Blood 2003; 102: 462–469.
Schlenk RF, Benner A, Krauter J, Büchner T, Sauerland C, Ehninger G et al. Individual patient data-based meta-analysis of patients aged 16 to 60 years with core binding factor acute myeloid leukemia: a survey of the German Acute Myeloid Leukemia Intergroup. J Clin Oncol 2004; 22: 1–11.
San Miguel JF, Martínez A, Macedo A, Vidriales MB, López-Berges C, González M et al. Immunophenotyping investigation of minimal residual disease is a useful approach for predicting relapse in acute myeloid leukemia patients. Blood 1997; 90: 2465–2470.
San Miguel JF, Vidriales MB, López-Berges C, Díaz-Mediavilla J, Gutiérrez N, Cañizo C et al. Early immunophenotypical evaluation of minimal residual disease in acute myeloid leukemia identifies different patient risk groups and may contribute to postinduction treatment stratification. Blood 2001; 98: 1746–1751.
Sievers EL, Lange BJ, Alonzo TA, Gerbing RB, Bernstein ID, Smith FO et al. Immunophenotypic evidence of leukemia after induction therapy predicts relapse: results from a prospective Children's Cancer Group study of 252 patients with acute myeloid leukemia. Blood 2003; 101: 3398–3406.
Kern W, Voskova D, Schoch C, Hiddemann W, Schnittger S, Haferlach T . Determination of relapse risk based on assessment of minimal residual disease during complete remission by multiparameter flow cytometry in unselected patients with acute myeloid leukemia. Blood 2004; 104: 3078–3085.
Feller N, van der Pol MA, van Stijn A, Weijers GWD, Westra AH, Evertse BW et al. MRD parameters using immunophenotypic detection methods are highly reliable in predicting survival in acute myeloid leukemia. Leukemia 2004; 18: 1380–1390.
Buonamici S, Ottaviani E, Testoni N, Montefusco V, Visani G, Bonifazi F et al. Real-time quantitation of minimal residual disease in inv(16)-positive acute myeloid leukemia may indicate risk for clinical relapse and may identify patients in a curable state. Blood 2002; 99: 443–449.
Guerrasio A, Pilatrino C, De Micheli D, Cilloni D, Serra A, Gottardi E et al. Assessment of minimal residual disease (MRD) in CBFbeta/MYH11-positive acute myeloid leukemias by qualitative and quantitative RT-PCR amplification of fusion transcripts. Leukemia 2002; 16: 1176–1181.
Morschhauser F, Cayuela JM, Martini S, Baruchel A, Rousselot P, Sociè G et al. Evaluation of minimal residual disease using reverse-transcription polymerase chain reaction in t(8;21) acute myeloid leukemia: a multicenter study of 51 patients. J Clin Oncol 2000; 18: 778–794.
Krauter J, Görlich K, Ottmann O, Lübbert M, Döhner H, Heit W et al. Prognostic value of minimal residual disease quantification by real-time reverse transcriptase polymerase chain reaction in patients with core binding factor leukemias. J Clin Oncol 2003; 21: 4413–4422.
Marcucci G, Caligiuri MA, Bloomfield CD . Core binding factor (CBF) acute myeloid leukemia: is molecular monitoring by RT-PCR useful clinically? Eur J Haematol 2003; 71: 143–154.
Tobal K, Newton J, Macheta M, Chang J, Morgenstern G, Evans PAS et al. Molecular quantitation of minimal residual disease in acute myeloid leukemia with t(8;21) can identify patients in durable remission and predict clinical relapse. Blood 2000; 95: 815–819.
Jurlander J, Caligiuri MA, Ruutu T, Baer MR, Strout MP, Oberkircher AR et al. Persistence of the AML1/ETO fusion transcript in patients treated with allogeneic bone marrow transplantation for t(8;21) leukemia. Blood 1996; 88: 2183–2191.
Viehmann S, Teigler-Schlegel A, Bruch J, Langebrake C, Reinhardt D, Harbott J . Monitoring of minimal residual disease (MRD) by real-time quantitative reverse transcription PCR (RQ-RT-PCR) in childhood acute myeloid leukemia with AML1/ETO rearrangement. Leukemia 2003; 17: 1130–1136.
Van Dongen JJM, Macintyre EA, Gabert JA, Delabesse E, Rossi V, Saglio G et al. Standardized RT-PCR analysis of fusion gene transcripts from chromosome aberrations in acute leukemia for detection of minimal residual disease. Report of the BIOMED-1 Concerted Action: investigation of minimal residual disease in acute leukemia. Leukemia 1999; 13: 1901–1928.
Brunet S, Esteve J, Berlanga J, Ribera JM, Bueno J, Martí JM et al. Treatment of primary acute myeloid leukemia: results of a prospective multicenter trial including high-dosecytarabine or stem cell transplantation as post-remission strategy. Haematologica 2004; 89: 940–949.
ISCN. An International System for Human Cytogenetic Nomenclature, Mitelman F (ed). Karger: Basel, 1995.
Muñoz L, Nomdedéu JF, Villamor N, Guardia R, Colomer D, Ribera JM et al. Acute myeloid leukemia with MLL rearrangements: clinicobiological features, prognostic impact and value of flow cytometry in the detection of residual leukemic cells. Leukemia 2003; 17: 76–82.
Muñoz L, Aventín A, Villamor N, Juncà J, Acebedo G, Domingo A et al. Immunophenotypic findings in acute myeloid leukemia with FLT3 internal tandem duplication. Haematologica 2003; 88: 637–645.
Macedo A, Orfao A, Gonzalez M, Vidriales MB, López-Berges MC, Martínez A et al. Immunological detection of blast cell subpopulations in acute myeloblastic leukemia at diagnosis: implications for minimal residual disease studies. Leukemia 1995; 9: 993–998.
Macedo A, Orfao A, Martínez MB, Valverde B, López-Berges MC, San Miguel JF . Immunophenotype of c-kit cells in normal human bone marrow: implications for the detection of minimal residual disease in AML. Br J Hematol 1995; 89: 338–341.
Gabert J, Beillard E, van der Velden VH, Bi W, Grimwade D, Pallisgaard N et al. Standardization and quality control studies of ‘real-time’ quantitative reverse transcriptase polymerase chain reaction of fusion gene transcripts for residual disease detection in leukemia- A Europe against cancer program. Leukemia 2003; 17: 2318–2357.
Cheson BD, Bennett JM, Kopecky KJ, Buchner T, Willman CL, Estey EH et al. Revised recommendations of the International Working Group for diagnosis, standardization of response criteria, treatment outcomes, and reporting standards for therapeutic trials in acute myeloid leukaemia. J Clin Oncol 2003; 21: 4642–4649.
Liu PP, Hajra A, Wijmenga C, Collins FS . Molecular pathogenesis of the chromosome 16 inversion in the M4Eo subtype of acute myeloid leukemia. Blood 1995; 85: 2289–2302, Erratum in: Blood 1997; 89: 1842.
Aventín A, La Starza R, Nomdedeu J, Brunet S, Sierra J, Mecucci C . Typical CBFbeta/MYH11 fusion due to insertion of the 3′-MYH11 gene into 16q22 in acute monocytic leukemia with normal chromosomes 16 and trisomies 8 and 22. Cancer Genet Cytogenet 2000; 123: 137–139.
Van der Reijden BA, Simons A, Luiten E, van der Poel SC, Hogenbirk PE, Tönnissen E et al. Minimal residual disease quantification in patients with acute myeloid leukemia and inv(16)/CBFB/MYH11 gene fusion. Br J Haematol 2002; 118: 411–418.
Baer MR, Stewart CC, Dodge RK, Leget G, Sulé N, Mrózek K et al. High frequency of immunophenotype changes in acute myeloid leukemia at relapse: implications for residual disease detection (Cancer and Leukemia Group B Study 8361). Blood 2001; 97: 3574–3580.
Campana D . Determination of minimal residual disease in leukaemia patients. Br J Haematol 2003; 121: 823–838.
Marcucci G, Livak KJ, Bi W, Strout MP, Bloomfield CD, Caligiuri MA . Detection of minimal residual disease in patients with AML1/ETO-associated acute myeloid leukemia using a novel quantitative reverse transcription polymerase chain reaction assay. Leukemia 1998; 12: 1482–1489.
Schnittger S, Weisser M, Schoch C, Hiddemann W, Haferlach T, Kern W . New score predicting for prognosis in PML-RARA+, AML1-ETO+, or CBFB-MYH11+ acute myeloid leukemia based on quantification of fusion transcripts. Blood 2003; 102: 2746–2755.
Diverio D, Rossi V, Avvisati G, De Santis S, Pistilli A, Pane F et al. Early detection of relapse by prospective reverse transcriptase-polymerase chain reaction analysis of the PML/RARalpha fusion gene in patients with acute promyelocytic leukemia enrolled in the GIMEMA-AIEOP multicenter ‘AIDA’ trial, GIMEMA.AIEOP Multicenter ‘AIDA’ Trial. Blood 1998; 92: 784–789.
Pallisgaard N, Clausen N, Schroder H, Hokland P . Rapid and sensitive minimal residual disease detection in acute leukemia by quantitative real-time RT-PCR exemplified by t(12;21) TEL-AML1 fusion transcript. Genes Chromosomes Cancer 1999; 26: 355–365.
Neale GAM, Coustan-Smith E, Stow P, Pan Q, Chen X, Pui CH et al. Comparative analysis of flow cytometry and polymerase chain reaction for the detection of minimal residual disease in childhood acute lymphoblastic leukemia. Leukemia 2004; 18: 934–938.
Leroy H, de Botton S, Grardel-Duflos N, Darre S, Leleu X, Roumier C et al. Prognosis value of real-time quantitative PCR (RQ-PCR) in AML with t(8;21). Leukemia 2005; 19: 367–372.
Malec M, Van Der Velden VH, Bjorklund E, Wijkhuijs JM, Soderhall S, Mazur J et al. Analysis of minimal residual disease in childhood acute lymphoblastic leukemia: comparison between RQ-PCR analysis of Ig/TcR gene rearrangements and multicolor flow cytometric immunophenotyping. Leukemia 2004; 18: 1630–1636.
Acknowledgements
This study was supported by Fondo de Investigaciones Sanitarias (FIS) Grants (GO3/008 and C03/010) and XT2004/00058 from Generalitat of Catalonia. GP is a recipient of a grant of the Fundación Española de Hematología y Hemoterapia (FEHH).
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Perea, G., Lasa, A., Aventín, A. et al. Prognostic value of minimal residual disease (MRD) in acute myeloid leukemia (AML) with favorable cytogenetics [t(8;21) and inv(16)]. Leukemia 20, 87–94 (2006). https://doi.org/10.1038/sj.leu.2404015
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404015
Keywords
This article is cited by
-
Association of hematologic response and assay sensitivity on the prognostic impact of measurable residual disease in acute myeloid leukemia: a systematic review and meta-analysis
Leukemia (2022)
-
Dynamic assessment of measurable residual disease in favorable-risk acute myeloid leukemia in first remission, treatment, and outcomes
Blood Cancer Journal (2021)
-
Prognostic value of lymphoid marker CD7 expression in acute myeloid leukemia patients undergoing allogeneic hematopoietic cell transplantation in first morphological complete remission
International Journal of Hematology (2021)
-
Application of a digital PCR method for WT1 to myeloid neoplasms in CR and deep ELN WT1 molecular response (< 10 copies)
Annals of Hematology (2020)
-
Low Rate of Invasive Fungal Infections During Induction and Consolidation Chemotherapy for Adults with De Novo Acute Myeloid Leukemia Without Anti-mold Prophylaxis: Single-Center 2002–2018 Empirical/Pre-emptive Approach
Mycopathologia (2020)