Abstract
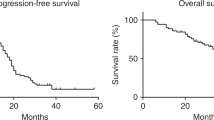
The role of high-dose chemotherapy (HDCT) in epithelial ovarian cancer (EOC) remains controversial. This study was initiated to compare the efficacy and tolerability of HDCT as a consolidation approach in women with chemosensitive advanced EOC (FIGO stages IIC–IV). Patients who had achieved their first clinical complete remission after six cycles of conventional paclitaxel and carboplatin combination chemotherapy were randomly assigned to receive or not high-dose melphalan. The primary objective was to compare time to disease progression (TTP). A total of 80 patients were enrolled onto the trial. Patients who were randomized to receive HDCT were initially treated with cyclophosphamide 4 g/m2 for PBPC mobilization. HDCT consisted of melphalan 200 mg/m2. Of the 37 patients who were allocated to HDCT, 11 (29.7%) did not receive melphalan either due to patient refusal (n=5) or due to failure of PBPC mobilization (n=6). In an intent-to-treat analysis, there were no significant differences between the two arms in TTP (P=0.059) as well as in overall survival (OS) (P=0.38).
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Landis SH, Murray T, Bolden S, Wingo PA . Cancer statistics, 1999. CA Cancer J Clin 1999; 49: 8–31.
Ozols EF, Rubin SC, Thomas G, Robboy S . Epithelial ovarian cancer. In: Hoskins WJ, Perez CA, Young RC (eds). Principles and Practice of Gynecologic Oncology. Lippincott Williams & Wilkins: Philadelphia, 2000, pp 981–1057.
Cannistra SA, Bast Jr RC, Berek JS, Bookman MA, Crum CP, DePriest PD et al. Progress in the management of gynecologic cancer: consensus summary statement. J Clin Oncol 2003; 21 (Suppl 10): 129–132.
Cannistra SA . Cancer of the ovary. N Engl J Med 2004; 351: 2519–2529.
Mc Guire WP, Hoskins WJ, Brady MF, Kucera PR, Partridge EE, Look KY et al. Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. N Engl J Med 1996; 334: 1–6.
Neijt JP, Engelholm SA, Tuxen MK, Sorensen PG, Hansen M, Sessa C et al. Exploratory phase III study of paclitaxel and cisplatin versus paclitaxel and carboplatin in advanced ovarian cancer. J Clin Oncol 2000; 18: 3084–3092.
Piccart MJ, Bertelsen K, James K, Cassidy J, Mangioni C, Simonsen E et al. Randomized intergroup trial of cisplatin-paclitaxel versus cisplatin-cyclophosphamide in women with advanced epithelial ovarian cancer: three-year results. J Natl Cancer Inst 2000; 92: 699–708.
Du Bois A, Lück HJ, Meier W, Adams HP, Möbus V, Costa S et al. A randomized clinical trial of cisplatin/paclitaxel versus carboplatin/paclitaxel as first-line treatment of ovarian cancer. J Natl Cancer Inst 2003; 95: 1320–1329.
ICON1, EORTC-ACTION. International Collaborative Ovarian Neoplasm Trial 1 and Adjuvant Chemotherapy In Ovarian Neoplasm Trial. Two parallel randomized phase III trials of adjuvant chemotherapy in patients with early-stage ovarian carcinoma. J Natl Cancer Inst 2003; 95: 105–112.
Vasey PA, Jayson GC, Gordon A, Gabra H, Coleman R, Atkinson R et al. Phase III randomized trial of docetaxel-carboplatin versus paclitaxel-carboplatin as first-line chemotherapy for ovarian carcinoma. J Natl Cancer Inst 2004; 96: 1682–1691.
Aravantinos G, Fountzilas G, Kosmidis P, Dimopoulos MA, Stathopoulos GP, Pavlidis N et al. Paclitaxel plus carboplatin versus paclitaxel plus alternating carboplatin and cisplatin for initial treatment of advanced ovarian cancer: long-term efficacy results. A Hellenic Cooperative Oncology Group (HeCOG) study. Ann Oncol 2005; 16: 1116–1122.
Du Bois A, Weber B, Rochon J, Meier W, Goupil A, Olbricht S et al. Addition of epirubicin as a third drug to carboplatin-paclitaxel in first-line treatment of advanced ovarian cancer: a prospectively randomized gynecologic cancer intergroup trial by the Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group and the Groupe d'Investigateurs Nationaux pur l'Etude des Cancer Ovariens. J Clin Oncol 2006; 24: 1127–1135.
Mc Guire III WP . High-dose chemotherapeutic approaches to ovarian cancer management. Semin Oncol 2000; 27 (Suppl 7): 41–46.
Perillo A, Bonanno G, Pierelli L, Rutella S, Scambia G . Mancuso S: stem cells in gynecology and obstetrics. Panminerva Medica 2004; 46: 49–59.
Aghajanian C, Fennelly D, Shapiro F, Waltzman R, Almadrones L, O'Flaherty C et al. Phase II study of ‘dose-dense’ high-dose chemotherapy treatment with peripheral-blood progenitor-cell support as primary treatment for patients with advanced ovarian cancer. J Clin Oncol 1998; 16: 1852–1860.
Schilder RJ, Brady MF, Spriggs D, Shea T . Pilot evaluation of high-dose carboplatin and paclitaxel followed by high-dose melphalan supported by peripheral blood stem cells in previously untreated advanced ovarian cancer: a Gynecologic Oncology Group study. Gynecol Oncol 2003; 88: 3–8.
Ledermann JA, Frickhofen N, Wandt H, Bengala C, Champion K, Hinke A et al. A phase III randomised trial of sequential high dose chemotherapy (HDC) with peripheral blood stem cell support or standard dose chemotherapy (SDC) for first-line treatment of ovarian cancer. J Clin Oncol 2005; 23 (Suppl 16): 456s; abstract 5006.
Legros M, Dauplat J, Flenny J, Cure H, Suzanne F, Chassagne J et al. High-dose chemotherapy with hematopoietic rescue in patients with stage III to IV ovarian cancer: long-term results. J Clin Oncol 1997; 15: 1302–1308.
Curé H, Battista C, Biron P, Bay JO, Lotz J, Lioures B et al. Phase III randomized trial of high-dose chemotherapy and peripheral blood stem cell support as consolidation in patients with advanced ovarian cancer: 5-year follow-up of a GINECO/FNCLCC/SFGMTC study. Bone Marrow Transplant 2005; 35 (Suppl 2): 552; abstract 0270.
Miller AB, Hoogerstraten B, Staquet M, Winkler A . Reporting results of cancer treatment. Cancer 1981; 47: 207–214.
Machin D, Campbell MJ, Fayers PM, Pinol A . Statistical Tables for the Design of Clinical Studies, 2nd edn. Blackwell Scientific Publications: Oxford, 1998.
Agresti A . Categorical Data Analysis. John Wiley & Sons: New York, 1990.
Kaplan E, Meier F . Non-parametric estimation from incomplete observations. J Am Stat Assoc 1958; 53: 457–481.
Peto R, Peto J . Asymptotically efficient rank invariant test procedures. J Stat Soc (Series A) 1972; 135: 185–198.
Cox DR . Regression models in life tables (with discussion). J R Stat Soc B 1972; 34: 187–220.
Shpall EJ, Clarke-Pearson D, Soper JT, Berchuck A, Jones RB, Bast RC et al. High-dose alkylating agent chemotherapy with autologous bone marrow support in patients with stage III/IV epithelial ovarian cancer. Gynecol Oncol 1990; 38: 386–391.
Brown ER, Belinson JL, Berek JS, McIntosh D, Hurd D, Ball H et al. Salvage therapy for recurrent and refractory ovarian cancer with high-dose chemotherapy and autologous bone marrow support: a Gynecologic Oncology Group pilot study. Gynecol Oncol 1994; 54: 142–146.
Stiff PJ, Bayer R, Kerger C, Potkul RK, Malhotra D, Peace DJ et al. High-dose chemotherapy with autologous transplantation for persistant/relapsed ovarian cancer: a multivariate analysis of survival for 100 consecutively treated patients. J Clin Oncol 1997; 15: 1309–1317.
Pujade-Lauraine E, Cure H, Battista C, Guastalla JP, Chiurazzi B, Fabbro M et al. High dose chemotherapy in ovarian cancer. Int J Gynecol Cancer 2001; 11 (Suppl 1): 64–67.
Bertucci F, Viens P, Delpero JR, Bardou VJ, Faucher C, Houvenaeghel G et al. High-dose melphalan-based chemotherapy and autologous stem cell transplantation after second look laparotomy in patients with chemosensitive advanced ovarian carcinoma: long-term results. Bone Marrow Transplant 2000; 26: 61–67.
Salerno MG, Ferrandina G, Greggi S, Pierelli L, Menichella G, Leone G et al. High-dose chemotherapy as a consolidation approach in advanced ovarian cancer: long-term results. Bone Marrow Transplant 2001; 27: 1017–1025.
Bengala C, Guarneri V, Ledermann J, Rosti G, Wandt H, Lotz JP et al. High-dose chemotherapy with autologous hematopoietic support for advanced ovarian cancer in first complete remission: retrospective analysis from the Solid Tumor Registry of the European Group for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplant 2005; 36: 25–31.
Lotz J-P, Bouleuc C, André T, Touboul E, Macovei C, Hannoun L et al. Tandem high-dose chemotherapy with ifosfamide, carboplatin, and teniposide with autologous bone marrow transplantation for the treatment of poor prognosis common epithelial ovarian carcinoma. Cancer 1996; 77: 2550–2559.
Grénman S, Wiklund T, Jalkanen J, Kuoppala T, Mäenpää J, Kuronen A et al. A randomized phase III study comparing high-dose chemotherapy to conventionally dosed chemotherapy for stage III ovarian cancer: The Finnish Ovarian Cancer (FINOVA) study. Eur J Cancer 2006; 42 (14): 2196–2199.
Piver MS . Ovarian carcinoma. A decade of progress. Cancer 1984; 54 (Suppl 11): 2706–2715.
Maraninchi D, Abecasis M, Gastant JA, Herve P, Sebahoun G, Flesch M et al. High-dose melphalan with autologous bone marrow rescue for the treatment of adult solid tumors. Cancer Treat Rep 1984; 68: 471–474.
Von Hoff DD, Clark GM, Weiss CR, Marshall MH, Buchok JB, Knight WA et al. Use of in vitro dose response effects to select antineoplastics for high-dose or regional administration regimens. J Clin Oncol 1986; 4: 1827–1834.
Frei III E, Teicher BA, Holden SA, Cathcart KN, Wang YY . Effect of alkylating agent dose: preclinical studies and possible clinical correlation. Cancer Res 1988; 48: 6417–6423.
Lazarus HM, Herzig RH, Graham-Pole J, Wolff SN, Phillips GL, Strandjord S et al. Intensive melphalan chemotherapy and cryopreserved autologous bone marrow transplantation for the treatment of refractory cancer. J Clin Oncol 1983; 1: 359–367.
Schabel Jr FM, Trader MW, Laster Jr WR, Wheeler GP, Witt MH . Patterns of resistance and therapeutic synergism among alkylating agents. Antibiot Chemother 1978; 23: 200–215.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Papadimitriou, C., Dafni, U., Anagnostopoulos, A. et al. High-dose melphalan and autologous stem cell transplantation as consolidation treatment in patients with chemosensitive ovarian cancer: results of a single-institution randomized trial. Bone Marrow Transplant 41, 547–554 (2008). https://doi.org/10.1038/sj.bmt.1705925
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705925
Keywords
This article is cited by
-
Are there candidates for high-dose chemotherapy in ovarian carcinoma?
Journal of Experimental & Clinical Cancer Research (2012)
-
Stem cells in clinical practice: applications and warnings
Journal of Experimental & Clinical Cancer Research (2011)