Abstract
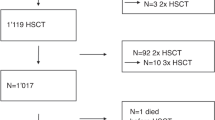
A cohort of 138 children with 144 hematopoietic stem cell transplantation (HSCT) performed in 1997–2006 were analyzed to evaluate risk factors and mortality predictors of hepatic veno-occlusive disease (VOD). Nineteen patients (13.2%) developed VOD (nine boys, median age 3.5 years) at 1–21 days after HSCT (median 13 days). Age ⩽2 years at transplant (odds ratio (OR)=5.25, P=0.011), BU–CY conditioning (OR=5.16, P=0.001), thalassemia major (OR=3.97, P=0.015), platelet engraftment beyond day +21 (OR=8.67, P=0.025) were univariate risk factors for VOD. The first two remained significant in multivariate regression. Seven patients (36.8%) with VOD died, at a median of 44 days post transplant (range, 30–421 days). The 5-year survival was 62%. All surviving patients had normal liver function on follow-up at 0.5–9 years. Patients with VOD had higher 100-day mortality (16.3 vs 9.6%, P=0.024). Mortality predictors included donors other than autologous or matched sibling (hazard ratio (HR)=23.6, P=0.006), hepatic and cutaneous GVHD (HR=8.15, P=0.038), maximal weight gain >9% (HR=6.81, P=0.023), pleural effusion, intensive care unit admission, peak bilirubin >300 μmol l−1 (HR=13.6, P=0.016), day +21 bilirubin >200 μmol l−1 (HR=33.9, P=0.001), and rise of bilirubin >15 μmol l−1 per day within the first week (HR=19.8, P=0.006). Mortality was substantially higher if >3 predictors were present (HR=33.9, P=0.001). Meticulous monitoring in high-risk patients and early treatment should be considered before VOD progresses beyond salvage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Brugieres L, Hartmann O, Benhamou E, Zafrani ES, Caillaud JM, Patte C et al. Veno-occlusive disease of the liver following high-dose chemotherapy and autologous bone marrow transplantation in children with solid tumors: incidence, clinical course and outcome. Bone Marrow Transplant 1988; 3: 53–58.
Lapierre V, Mahe C, Auperin A, Stambouli F, Oubouzar N, Tramalloni D et al. Platelet transfusion containing ABO-incompatible plasma and hepatic veno-occlusive disease after hematopoietic transplantation in young children. Transplantation 2005; 80: 314–319.
Cesaro S, Pillon M, Talenti E, Toffolutti T, Calore E, Tridello G et al. A prospective survey on incidence, risk factors and therapy of hepatic veno-occlusive disease in children after hematopoietic stem cell transplantation. Haematologica 2005; 90: 1396–1404.
Barker CC, Butzner JD, Anderson RA, Brant R, Sauve RS . Incidence, survival and risk factors for the development of veno-occlusive disease in pediatric hematopoietic stem cell transplant recipients. Bone Marrow Transplant 2003; 32: 79–87.
Reiss U, Cowan M, McMillan A, Horn B . Hepatic venoocclusive disease in blood and bone marrow transplantation in children and young adults: incidence, risk factors, and outcome in a cohort of 241 patients. J Pediatr Hematol Oncol 2002; 24: 746–750.
Hasegawa S, Horibe K, Kawabe T, Kato K, Kojima S, Matsuyama T et al. Veno-occlusive disease of the liver after allogeneic bone marrow transplantation in children with hematologic malignancies: incidence, onset time and risk factors. Bone Marrow Transplant 1998; 22: 1191–1197.
Bulley SR, Strahm B, Doyle J, Dupuis LL . Defibrotide for the treatment of hepatic veno-occlusive disease in children. Pediatr Blood Cancer 2006; 48: 700–704.
Corbacioglu S, Greil J, Peters C, Wulffraat N, Laws HJ, Dilloo D et al. Defibrotide in the treatment of children with veno-occlusive disease (VOD): a retrospective multicentre study demonstrates therapeutic efficacy upon early intervention. Bone Marrow Transplant 2004; 33: 189–195.
Jones RJ, Lee KS, Beschorner WE, Vogel VG, Grochow LB, Braine HG et al. Venoocclusive disease of the liver following bone marrow transplantation. Transplantation 1987; 44: 778–783.
McDonald GB, Hinds MS, Fisher LD, Schoch HG, Wolford JL, Banaji M et al. Veno-occlusive disease of the liver and multiorgan failure after bone marrow transplantation: a cohort study of 355 patients. Ann Intern Med 1993; 118: 255–267.
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA et al. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation 1974; 18: 295–304.
Blostein MD, Paltiel OB, Thibault A, Rybka WB . A comparison of clinical criteria for the diagnosis of veno-occlusive disease of the liver after bone marrow transplantation. Bone Marrow Transplant 1992; 10: 439–443.
Pihusch M, Wegner H, Goehring P, Salat C, Pihusch V, Hiller E et al. Diagnosis of hepatic veno-occlusive disease by plasminogen activator inhibitor-1 plasma antigen levels: a prospective analysis in 350 allogeneic hematopoietic stem cell recipients. Transplantation 2005; 80: 1376–1382.
Grochow LB, Jones RJ, Brundrett RB, Braine HG, Chen TL, Saral R et al. Pharmacokinetics of busulfan: correlation with veno-occlusive disease in patients undergoing bone marrow transplantation. Cancer Chemother Pharmacol 1989; 25: 55–61.
Meresse V, Hartmann O, Vassal G, Benhamou E, Valteau-Couanet D, Brugieres L et al. Risk factors for hepatic veno-occlusive disease after high-dose busulfan-containing regimens followed by autologous bone marrow transplantation: a study in 136 children. Bone Marrow Transplant 1992; 10: 135–141.
Gibbs JP, Yang JS, Slattery JT . Comparison of human liver and small intestinal glutathione S-transferase-catalyzed busulfan conjugation in vitro. Drug Metab Dispos 1998; 26: 52–55.
Lee JH, Choi SJ, Lee JH, Kim SE, Park CJ, Chi HS et al. Decreased incidence of hepatic veno-occlusive disease and fewer hemostatic derangements associated with intravenous busulfan vs oral busulfan in adults conditioned with busulfan+cyclophosphamide for allogeneic bone marrow transplantation. Ann Hematol 2005; 84: 321–330.
Zwaveling J, den Hartigh J, Lankester AC, Guchelaar HJ, Egeler RM, Bredius RG . Once-daily intravenous busulfan in children prior to stem cell transplantation: study of pharmacokinetics and early clinical outcomes. Anticancer Drugs 2006; 17: 1099–1105.
Lee MY, Chiou TJ, Bai LY, Hsiao LT, Hung GY, Chang CY et al. Intravenous busulfan as preparative regimen in pediatric patients receiving hematopoietic stem cell transplantation: the preliminary experience in Taiwan. J Chin Med Assoc 2004; 67: 117–122.
Slattery JT, Kalhorn TF, McDonald GB, Lambert K, Buckner CD, Bensinger WI et al. Conditioning regimen-dependent disposition of cyclophosphamide and hydroxycyclophosphamide in human marrow transplantation patients. J Clin Oncol 1996; 14: 1484–1494.
Brodsky R, Topolsky D, Crilley P, Bulova S, Brodsky I . Frequency of veno-occlusive disease of the liver in bone marrow transplantation with a modified busulfan/cyclophosphamide preparative regimen. Am J Clin Oncol 1990; 13: 221–225.
Beelen DW, Trenschel R, Casper J, Freund M, Hilger RA, Scheulen ME et al. Dose-escalated treosulphan in combination with cyclophosphamide as a new preparative regimen for allogeneic haematopoietic stem cell transplantation in patients with an increased risk for regimen-related complications. Bone Marrow Transplant 2005; 35: 233–241.
Dix SP, Wingard JR, Mullins RE, Jerkunica I, Davidson TG, Gilmore CE et al. Association of busulfan area under the curve with veno-occlusive disease following BMT. Bone Marrow Transplant 1996; 17: 225–230.
Yeager AM, Wagner Jr JE, Graham ML, Jones RJ, Santos GW, Grochow LB . Optimization of busulfan dosage in children undergoing bone marrow transplantation: a pharmacokinetic study of dose escalation. Blood 1992; 80: 2425–2428.
Kletzel M, Jacobsohn D, Duerst R . Pharmacokinetics of a test dose of intravenous busulfan guide dose modifications to achieve an optimal area under the curve of a single daily dose of intravenous busulfan in children undergoing a reduced-intensity conditioning regimen with hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2006; 12: 472–479.
Tran H, Petropoulos D, Worth L, Mullen CA, Madden T, Andersson B et al. Pharmacokinetics and individualized dose adjustment of intravenous busulfan in children with advanced hematologic malignancies undergoing allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2004; 10: 805–812.
Ozkaynak MF, Weinberg K, Kohn D, Sender L, Parkman R, Lenarsky C . Hepatic veno-occlusive disease post-bone marrow transplantation in children conditioned with busulfan and cyclophosphamide: incidence, risk factors, and clinical outcome. Bone Marrow Transplant 1991; 7: 467–474.
Srivastava A, Poonkuzhali B, Shaji RV, George B, Mathews V, Chandy M et al. Glutathione S-transferase M1 polymorphism: a risk factor for hepatic venoocclusive disease in bone marrow transplantation. Blood 2004; 104: 1574–1577.
Tay J, Tinmouth A, Fergusson D, Huebsch L, Allan DS . Systematic review of controlled clinical trials on the use of ursodeoxycholic acid for the prevention of hepatic veno-occlusive disease in hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2007; 13: 206–217.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheuk, D., Wang, P., Lee, T. et al. Risk factors and mortality predictors of hepatic veno-occlusive disease after pediatric hematopoietic stem cell transplantation. Bone Marrow Transplant 40, 935–944 (2007). https://doi.org/10.1038/sj.bmt.1705835
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705835
Keywords
This article is cited by
-
EASIX predicts non-relapse mortality after haploidentical transplantation with post-transplant cyclophosphamide
Bone Marrow Transplantation (2023)
-
Analysis of risk factors for hepatic sinusoidal obstruction syndrome following allogeneic hematopoietic stem cell transplantation in pediatric patients
Journal of Cancer Research and Clinical Oncology (2022)
-
Risk factors and outcomes of diffuse alveolar haemorrhage after allogeneic haematopoietic stem cell transplantation
Bone Marrow Transplantation (2021)
-
Hepatic veno-occlusive disease/sinusoidal obstruction syndrome after hematopoietic stem cell transplantation for thalassemia major: incidence, management, and outcome
Bone Marrow Transplantation (2021)
-
Hematopoietic stem cell transplantation in children with Griscelli syndrome type 2: a single-center report on 35 patients
Bone Marrow Transplantation (2020)