Abstract
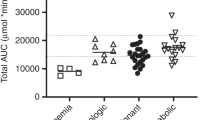
We report our experience with oral busulfan (BU) in 159 consecutive patients to evaluate the safety of home administration. Patients received a myeloablative BU-containing regimen, including oral anticonvulsant and antiemetic prophylaxis, followed by hematopoietic stem cell transplantation. Comprehensive verbal and written education was provided. Pharmacokinetic monitoring was performed and dose adjustments were made to target an area under the plasma concentration-time curve (AUC) of 900–1500 μmol.min/l. Safety was assessed by evaluating therapy-related toxicities, including seizures, venoocclusive disease (VOD) and patient tolerability. The utilization of pharmacokinetic monitoring was reviewed as a secondary end point. Of the 143 patients evaluated for BU-related seizures and VOD, only two (1.4%) experienced a generalized seizure and four patients (3%) were diagnosed with VOD. VOD resolved in three patients and was a contributing cause of death in one patient. Additional BU dosing owing to nausea and/or vomiting occurred in 28 patients (18%) and five patients (3%) were hospitalized. The median measured AUC was 1405 μmol.min/l, 68% of patients required a dose adjustment, and the median total administered BU dose was 13.6 mg/kg. In conclusion, high-dose oral BU can be safely administered on an outpatient basis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
McCune JS, Gibbs JP, Slattery JT . Plasma concentration monitoring of busulfan. Clin Pharmacokinet 2000; 39: 155–165.
Vassal G . Pharmacologically-guided dose adjustment of busulfan in high-dose chemotherapy regimens: rationale and pitfalls. Anticancer Res 1994; 14: 2363–2370.
Slattery JT, Clift RA, Buckner CD, Radich J, Storer B, Bensinger WI et al. Marrow transplantation for chronic myeloid leukemia: the influence of plasma busulfan levels on the outcome of transplantation. Blood 1997; 89: 3055–3060.
Slattery JT, Risler LJ . Therapeutic monitoring of busulfan in hematopoietic stem cell transplantation. Ther Drug Monit 1998; 20: 543–549.
Peters WP, Henner WD, Grochow LB, Olsen G, Edwards S, Stanbuck H et al. Clinical and pharmacological effects of high dose single agent busulfan with autologous bone marrow support in the treatment of solid tumors. Cancer Res 1987; 47: 6402–6406.
Jones RJ, Lee KS, Beschorner WE, Vogel VG, Grochow LB, Braine HG et al. Venoocclusive disease of the liver following bone marrow transplantation. Transplantation 1987; 44: 778–783.
McDonald GB, Slattery JT, Bouvier RB, Ren S, Batchelder A, Kalhorn T et al. Cyclophosphamide metabolism, liver toxicity, and mortality following hematopoietic stem cell transplantation. Blood 2003; 101: 2043–2048.
Grochow LB, Jones RJ, Brundrett RB, Braine HG, Chen TL, Saral R et al. Pharmacokinetics of busulfan; correlation with veno-occlusive disease in patients undergoing bone marrow transplantation. Cancer Chemother Pharmacol 1989; 25: 55–61.
Grochow L . Busulfan disposition: the role of therapeutic monitoring in bone marrow transplantation induction regimens. Semin Oncol 1993; 20: 18–21.
Dix SP, Wingard JR, Mullins RE, Jerkunica I, Davidson TG, Gilmore CE et al. Association of busulfan area under the curve with veno-occlusive disease following BMT. Bone Marrow Transplant 1996; 17: 225–230.
Deeg HJ, Storer B, Slattery JT, Anasetti C, Doney KC, Hansen JA et al. Conditioning with targeted busulfan and cyclophosphamide for hematopoietic stem cell transplantation from related and unrelated donors in patients with myelodysplastic syndrome. Blood 2002; 100: 1201–1207.
De Lima M, Couriel D, Thall P, Wang X, Madden T, Jones R et al. Once-daily intravenous busulfan and fludarabine: clinical and pharmacokinetic results of a myeloablative, reduced-toxicity conditioning regimen for allogeneic stem cell transplantation in AML and MDS. Blood 2004; 104: 857–864.
Kashyap A, Wingard J, Cagnoni P, Roy J, Tarantolo S, Hu W et al. Intravenous versus oral busulfan as part of a busulfan/cyclophosphamide preparative regimen for allogeneic hematopoietic stem cell transplantation: decreased incidence of hepatic venoocclusive disease (HVOD), HVOD-related mortality, and overall 100-day mortality. Biol Blood Marrow Transplant 2002; 8: 493–500.
Hassan M, Ehrsson H, Ljungman P . Aspects concerning busulfan pharmacokinetics and bioavailability. Leuk Lymphoma 1996; 22: 395–407.
Svahn B, Remberger M, Myrback K, Holmberg K, Eriksson B, Hentschke P et al. Home care during the pancytopenia phase after allogeneic hematopoietic stem cell transplantation is advantageous compared with hospital care. Blood 2002; 100: 4317–4324.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Matthews, R., Emami, M., Connaghan, D. et al. Home administration of high-dose oral busulfan in patients undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant 39, 397–400 (2007). https://doi.org/10.1038/sj.bmt.1705610
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705610
Keywords
This article is cited by
-
Outpatient myeloablative allo-SCT: a comprehensive approach yields decreased hospital utilization and low TRM
Bone Marrow Transplantation (2010)