Summary:
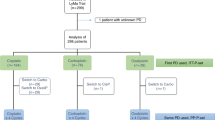
The majority of advanced ovarian cancer patients achieve an objective response following chemotherapy; however, only 20–30% are in remission after 5 years. Intraperitoneal or high-dose chemotherapy (HDC) may prolong disease-free and overall survival (OS) in patients with platinum-sensitive, small volume disease. To better define the subsets of patients who might benefit from HDC, we performed a retrospective analysis on 91 patients in 1st complete remission (CR) treated from 21 centres of the EBMT group. At a median follow-up of 48 months, median time-to-progression (TTP) and OS were 21.2 and 44.4 months, respectively. Tumour grade, stage, residual disease, disease status before HDC, type and year of transplant, source of haemopoietic progenitors and use of haemopoietic growth factors (HGF) after transplant were analysed for TTP and OS. The only significant parameter was the use of HGF: median OS for patients receiving or not receiving HGF was 46.2 vs 17.8 months, respectively (P: 0.035); this difference was maintained after multivariate analysis (P: 0.02). Our analysis does not identify any subgroup of patients in 1st CR who can benefit from HDC; however, median survival of patient with no residual disease has not been reached. The role of HGF after HDC deserves further investigation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ozols RF . Management of advanced ovarian cancer consensus summary. Advanced Ovarian Cancer Consensus Faculty. Semin Oncol 2000; 27 (Suppl. 7): 47–49.
Conte PF, Cianci C, Gadducci A . Update in the management of advanced ovarian carcinoma. Crit Rev Oncol Hematol 1999; 32: 49–58.
Bruzzone M, Repetto L, Chiara S et al. Chemotherapy vs radiotherapy in the management of ovarian cancer patients with pathological complete response or minimal residual disease at second look. Gynecol Oncol 1990; 38: 392–395.
Varia MA, Stehman FB, Bundy BN et al. Intraperitoneal radioactive phosphorus (32P) vs observation after negative second-look laparoptomy for stage III ovarian carcinoma: a randomised trial of the gynaecologic oncology group. J Clin Oncol 2003; 21: 2849–2855.
Markman M, Liu PY, Wilczynski S et al. Phase III randomized trial of 12 vs 3 months of maintenance paclitaxel in patients with advanced ovarian cancer after complete response to platinum and paclitaxel-based chemotherapy: a Southwest Oncology Group and Gynecologic Oncology Group trial. J Clin Oncol 2003; 21: 2460–2465.
Pignata S, De Placido S, Scambia G et al. Topotecan vs nihil after response to carboplatin and paclitaxel in advanced ovarian cancer. Early results of the MITO-1 (Multicenter Italian Trials in Ovarian Cancer) study. Proc Am Soc Clin Oncol 2003; 22: 446 (Abstr).
Pfisterer J, Lortholary A, Kimmig R et al. Paclitaxel/carboplatin (TC) vs Paclitaxel/carboplatin followed by topotecan (TC-Top) in first line treatment of ovarian cancer FIGO stages IIb–IV Interim results of a gynecologic cancer intergroup phase III trial of the AGO Ovarian Cancer Study Group and GINECO. Proc Am Soc Clin Oncol 2003; 22: 446 (abstr).
Barakat B, Sabbatini P, Bhaskaran D et al. Intraperitoneal chemotherapy for ovarian carcinoma: results of long-term follow-up. J Clin Oncol 2002; 20: 694–698.
Bruzzone M, Rubagotti A, Gadducci A et al. Intraperitoneal carboplatin with or without interferon-alpha in advanced ovarian cancer with minimal residual disease at second-look: a prospective randomised trial of 111 patients. Gynecol Oncol 1997; 65: 499–505.
Markman M, Bundy BN, Alberts DS et al. Phase III trial of standard-dose intravenous cisplatin plus paclitaxel vs moderately high-dose carboplatin followed by intravenous paclitaxel and intraperitoneal cisplatin in small-volume stage III ovarian carcinoma: an intergroup study of the Gynecologic Oncology Group, Southwestern Oncology Group and Eastern Cooperative Oncology Group. J Clin Oncol 2001; 19: 1001–1007.
Alberts DS, Liu PY, Hannigan EV et al. Intraperitoneal cisplatin plus intravenous cyclophosphamide vs intravenous cisplatin plus intravenous cyclophosphamide for stage III ovarian cancer. N Engl J Med 1996; 335: 1950–1955.
Gadducci A, Carnino F, Chiara S et al. Intraperitoneal vs intravenous cisplatin in combination with intravenous cyclophosphamide and epidoxorubicin in optimally cytoreduced advanced epithelial ovarian cancer: a randomized trial of the Gruppo Oncologico Nord-Ovest. Gynecol Oncol 2000; 76: 157–162.
Armstrong DK, Bundy BN, Baergen R et al. Randomized phase III study of intravenous (IV) paclitaxel and cisplatin vs IV paclitaxel, intraperitoneal (IP) cisplatin and IP paclitaxel in optimal stage III epithelial ovarian cancer (OC): a Gynecologic Oncology Group trial (GOG 172). Proc Am Soc Clin Oncol 2002; 21: 201a (Abstr).
Lotz J-P, Bouleuc C, Andrè T et al. Tandem high-dose chemotherapy with ifosfamide, carboplatin and teniposide with autologous bone marrow transplantation for the treatment of poor prognosis common epithelial ovarian carcinoma. Cancer 1996; 77: 2550–2559.
Salerno MG, Ferrandina G, Greggi S et al. High-dose chemotherapy as a consolidation approach in advanced ovarian cancer: long-term results. Bone Marrow Transplant 2001; 27: 1017–1025.
Schilder RJ, Brady MF, Spriggs D et al. Pilot evaluation of high-dose carboplatin and paclitaxel followed by high-dose melphalan supported by peripheral blood stem cells in previously untreated advanced ovarian cancer: a Gynecologic Oncology Group study. Gynecol Oncol 2003; 88: 3–8.
Bertucci F, Viens P, Delpero JR et al. High-dose melphalan-based chemotherapy and autologous stem cell transplantation after second look laparotomy in patients with chemosensitive advanced ovarian carcinoma: long-term results. Bone Marrow Transplant 2000; 26: 61–67.
Curè H, Battista C, Guastalla J et al. Phase III randomized trial of high-dose chemotherapy (HDC) and peripheral blood stem cell (PBSC) support as consolidation in patients (pts) with advanced ovarian cancer (AOC): 5-year follow-up of a GINECO/FNCLCC/SFGM-TC study. Proc Am Soc Clin Oncol 2004; 23 Abstr. 5006).
Stiff PJ, Bayer R, Kerger C et al. High-dose chemotherapy with autologous transplantation for persistent/relapsed ovarian cancer: a multivariate analysis of survival for 100 consecutively treated patients. J Clin Oncol 1997; 15: 1309–1317.
Ledermann JA, Herd R, Maraninchi D et al. High-dose chemotherapy for ovarian carcinoma: long-term results from the Solid Tumour Registry of the European Group for Blood and Marrow Transplantation (EBMT). Ann Oncol 2001; 12: 693–699.
Donato ML, Aleman A, Champlin RE et al. Analysis of 96 patients with advanced ovarian carcinoma treated with high-dose chemotherapy and autologous stem cell transplantation. Bone Marrow Transplant 2004; 33: 1219–1224.
Piccart MJ, Floquet A, Scarfone G et al. Intraperitoneal cisplatin vs no further treatment: 8-year results of EORTC 55875, a randomized phase III study in ovarian cancer patients with a pathologically complete remission after platinum-based intravenous chemotherapy. Int J Gynecol Cancer 2003; 13 (Suppl. 2): 196–203.
Brun J-L, Feyler A, Chene G et al. Long-term results and prognostic factors in patients with epithelial ovarian cancer. Gynecol Oncol 2000; 78: 21–27.
Chi DS, Liao JB, Leon LF et al. Identification of prognostic factors in advanced epithelial ovarian carcinoma. Gynecol Oncol 2001; 82: 532–537.
Heintz APM, Odicino F, Maissonneuve P et al. Carcinoma of the ovary. In: FIGO Annual Report on the results of treatment in Gynecological Cancer. J Epidemol Biostat 2001; 6: 107–138.
Norton L, Day R . Potential innovations in scheduling of cancer chemotherapy. In: DeVita Jr VT, Hellman S, Rosenberg SA (eds). Important Advances in Oncology. Lippincott: Philadelphia, PA, 1991, pp 57–72.
Aghajanian C, Fennelly D, Shapiro F et al. Phase II study of ‘dose dense’ high-dose chemotherapy treatment with peripheral blood progenitor cell support as primary treatment for patients with advanced ovarian cancer. J Clin Oncol 1998; 16: 1852–1860.
Wandt H, Birkmann J, Denzel T et al. Sequential cycles of high-dose chemotherapy with dose escalation of carboplatin with or without paclitaxel supported by G-CSF mobilized peripheral blood progenitor cells: a phase I/II study in advanced ovarian cancer. Bone Marrow Transplant 1999; 23: 763–770.
Pierelli L, Perillo A, Ferrnadina G et al. The role of the growth factor administration and T-cell recovery after peripheral blood progenitor cell transplantation in the treatment of solid tumors: results from a randomised comparison of G-CSF and GM-CSF. Transfusion 2001; 41: 1577–1585.
Fattorossi A, Battaglia A, Pierelli L et al. Effects of granulocyte-colony-stimulating factor and granulocyte/macrophage-colony-stimulating factor administration on T-cell proliferation and phagocyte cell-surface molecules during hematopoietic reconstitution after autologous peripheral blood progenitor cell transplantation. Cancer Immunol Immunother 2001; 49: 641–648.
Author information
Authors and Affiliations
Consortia
Corresponding author
Appendix
Appendix
The following Institutions provide information on their patients and we are grateful to all contributors who assisted us in the collection of these data: University College London Hospital (AH Goldstone, JA Ledermann), London, UK; Centre Val D'Aaurelle (M Fabbro), Montpellier, France; Centre Hospitalier Louis Pasteur (B Audhuy), Colmar, France; Hopital Laennec (J Andrieu), Paris, France; Centre Leon Bernard (P Biron), Lyon, France; Wilhelminenspital (H Ludwig, Vienna, Austria; Centre Jean Pierrin, (H Cure) Clermond–Ferrand, France; Hopital Tenon (JP Lotz), Paris, France; Klinikum Nurnberg, (H Wandt), Nurnberg, Germany; Policlinico Università (R Sabbatini), Modena, Italy; Hopital De Hautepierre (B Lioure), Strasbourg, France; St. Savas Hospital (A Efremidis), Athens, Greece; Ospedale Civile (G Rosti), Ravenna, Italy; Hospital Infantil La Paz (AM Martinez-Rubio), Madrid, Spain; Ospedale S Camillo (L De Rosa), Rome, Italy; Charles University Hospital, (T Kozak), Prague, Czech Republic; University Hospital (P Bojko), Essen, Germany; Hopital Erasme (W Feremans), Brussels, Belgium; Stadt. Kliniken Oldenburg (B Metzner), Oldenburg, Germany; Ludwig Boltzmann Institut fur Stammzelltransplantation, (W Hinterberger), Vienna, Austria; Hopital d'Enfants, (P Bordignoni), Nancy, France; University Hospital, (C Bengala), Pisa, Italy.
Rights and permissions
About this article
Cite this article
Bengala, C., Guarneri, V., Ledermann, J. et al. High-dose chemotherapy with autologous haemopoietic support for advanced ovarian cancer in first complete remission: retrospective analysis from the Solid Tumour Registry of the European Group for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplant 36, 25–31 (2005). https://doi.org/10.1038/sj.bmt.1705007
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705007
Keywords
This article is cited by
-
The inverted pyramid of biomarker-driven trials
Nature Reviews Clinical Oncology (2011)
-
Bloodless cancer treatment results of patients who do not want blood transfusion: single center experience of 77 cases
Supportive Care in Cancer (2010)
-
Phase I trial of autologous hematopoietic SCT with escalating doses of topotecan combined with CY and carboplatin in patients with relapsed or persistent ovarian or primary peritoneal carcinoma
Bone Marrow Transplantation (2010)
-
High-dose melphalan and autologous stem cell transplantation as consolidation treatment in patients with chemosensitive ovarian cancer: results of a single-institution randomized trial
Bone Marrow Transplantation (2008)