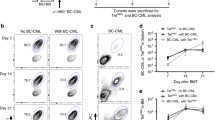
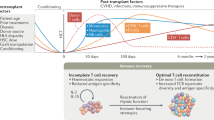
Summary:
Adoptive immunotherapy is the isolation and infusion of antigen-specific or nonspecific lymphocytes. Adoptive therapy with T cells may have a role in replacing, repairing, or enhancing immune function damaged by cytotoxic therapies, and rapid lymphocyte recovery may improve outcome after autologous and allogeneic stem cell transplantation (SCT). Recently, a plethora of information on the basic mechanisms of T-cell biology and regulation of cellular immune responses has emerged, permitting the development of new forms of adoptive cell therapy. Efficient ex vivo culture method for T-cell subsets affords the possibility of adoptive transfer of T cells engineered with enhanced capacity for central memory, effector cytotoxicity, Th1, Th2, veto cell, and T regulatory functions. Studies show that homeostatic T-cell proliferation is important for effective adoptive immunotherapy and pretreatment with chemotherapy may enhance the effects of infused T cells. Replicative senescence, in part due to telomere erosion, likely limits successful adoptive immunotherapy, though it may be possible to maintain T-cell pools by enforced expression of telomerase. Clinical trials now demonstrate that it is possible to enhance immune reconstitution after SCT with cytokines or infusions of ex vivo costimulated expanded T cells. These data all support the premise that adoptive therapy can accelerate reconstitution of cellular immunity with enhanced antitumor effects following SCT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Takaki A, Wiese M, Maertens G et al. Cellular immune responses persist and humoral responses decrease two decades after recovery from a single-source outbreak of hepatitis C. Nat Med 2000; 6: 578–582.
Lum LG . The kinetics of immune reconstitution after human marrow transplantation. Blood 1987; 69: 369–380.
Avigan D, Wu Z, Joyce R et al. Immune reconstitution following high-dose chemotherapy with stem cell rescue in patients with advanced breast cancer. Bone Marrow Transplant 2000; 26: 169–176.
Maury S, Mary JY, Rabian C et al. Prolonged immune deficiency following allogeneic stem cell transplantation: risk factors and complications in adult patients. Br J Haematol 2001; 115: 630–641.
Mackall CL, Stein D, Fleisher TA et al. Prolonged CD4 depletion after sequential autologous peripheral blood progenitor cell infusions in children and young adults. Blood 2000; 96: 754–762.
Avigan D, Pirofski L, Lazarus HM . Vaccination against infectious disease following hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2001; 7: 171–183.
Douek DC, Vescio RA, Betts MR et al. Assessment of thymic output in adults after haematopoietic stem-cell transplantation and prediction of T-cell reconstitution [see comments]. Lancet 2000; 355: 1875–1881.
Weinberg K, Blazar BR, Wagner JE et al. Factors affecting thymic function after allogeneic hematopoietic stem cell transplantation. Blood 2001; 97: 1458–1466.
Roux E, Dumont-Girard F, Starobinski M et al. Recovery of immune reactivity after T-cell-depleted bone marrow transplantation depends on thymic activity. Blood 2000; 96: 2299–2303.
Kim DH, Kim JG, Sohn SK et al. Clinical impact of early absolute lymphocyte count after allogeneic stem cell transplantation. Br J Haematol 2004; 125: 217–224.
Kumar S, Chen MG, Gastineau DA et al. Lymphocyte recovery after allogeneic bone marrow transplantation predicts risk of relapse in acute lymphoblastic leukemia. Leukemia 2003; 17: 1865–1870.
Porrata LF, Ingle JN, Litzow MR et al. Prolonged survival associated with early lymphocyte recovery after autologous hematopoietic stem cell transplantation for patients with metastatic breast cancer. Bone Marrow Transplant 2001; 28: 865–871.
Porrata LF, Gertz MA, Inwards DJ et al. Early lymphocyte recovery predicts superior survival after autologous hematopoietic stem cell transplantation in multiple myeloma or non-Hodgkin lymphoma. Blood 2001; 98: 579–585.
Chakrabarti S, Brown J, Guttridge M et al. Early lymphocyte recovery is an important determinant of outcome following allogeneic transplantation with CD34+selected graft and limited T-cell addback. Bone Marrow Transplant 2003; 32: 23–30.
Ferrandina G, Pierelli L, Perillo A et al. Lymphocyte recovery in advanced ovarian cancer patients after high-dose chemotherapy and peripheral blood stem cell plus growth factor support: clinical implications. Clin Cancer Res 2003; 9: 195–200.
Zhang L, Conejo-Garcia JR, Katsaros D et al. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N Engl J Med 2003; 348: 203–213.
Murali-Krishna K, Lau LL, Sambhara S et al. Persistence of memory CD8 T cells in MHC class I-deficient mice. Science 1999; 286: 1377–1381.
Freitas AA, Rocha B . Population biology of lymphocytes: the flight for survival. Annu Rev Immunol 2000; 18: 83–111.
Dummer W, Niethammer AG, Baccala R et al. T cell homeostatic proliferation elicits effective antitumor autoimmunity. J Clin Invest 2002; 110: 185–192.
Dudley ME, Wunderlich JR, Robbins PF et al. Cancer regression and autoimmunity in patients after clonal repopulation with antitumor lymphocytes. Science 2002; 298: 850–854.
Laport GG, Levine BL, Stadtmauer EA et al. Adoptive transfer of costimulated T cells induces lymphocytosis in patients with relapsed/refractory non-Hodgkin lymphoma following CD34+-selected hematopoietic cell transplantation. Blood 2003; 102: 2004–2013.
Geginat J, Lanzavecchia A, Sallusto F . Proliferation and differentiation potential of human CD8(+) memory T-cell subsets in response to antigen or homeostatic cytokines. Blood 2003; 101: 4260–4266.
Labrecque N, Whitfield LS, Obst R et al. How much TCR does a T cell need? Immunity 2001; 15: 71–82.
Bui JD, Calbo S, Hayden-Martinez K et al. A role for CaMKII in T cell memory. Cell 2000; 100: 457–467.
Dai ZH, Lakkis FG . Cutting edge: secondary lymphoid organs are essential for maintaining the CD4, but not CD8, naive T cell pool. J Immunol 2001; 167: 6711–6715.
Wynn RF, Cross MA, Hatton C et al. Accelerated telomere shortening in young recipients of allogeneic bone-marrow transplants. Lancet 1998; 351: 178–181.
Kapasi ZF, Murali-Krishna K, Mcrae ML et al. Defective generation but normal maintenance of memory T cells in old mice. Eur J Immunol 2002; 32: 1567–1573.
Greenberg PD . Adoptive T cell therapy of tumors: mechanisms operative in the recognition and elimination of tumor cells. Adv Immunol 1991; 49: 281–355.
Awwad M, North RJ . Cyclophosphamide (Cy)-facilitated adoptive immunotherapy of a Cy-resistant tumour. Evidence that Cy permits the expression of adoptive T-cell mediated immunity by removing suppressor T cells rather than by reducing tumour burden. Immunology 1988; 65: 87–92.
Matzinger P . Tolerance, danger, and the extended family. Annu Rev Immunol 1994; 12: 991–1045.
Huang AY, Golumbek P, Ahmadzadeh M et al. Role of bone marrow-derived cells in presenting MHC class I-restricted tumor antigens. Science 1994; 264: 961–965.
Effros RB, Pawelec G . Replicative senescence of T cells: does the Hayflick Limit lead to immune exhaustion? Immunol Today 1997; 18: 450–454.
Pawelec G, Rehbein A, Haehnel K et al. Human T-cell clones in long-term culture as a model of immunosenescence. Immunol Rev 1997; 160: 31–42.
Levine BL, Bernstein W, Craighead N et al. Effects of CD28 costimulation on long term proliferation of CD4+ T cells in the absence of exogenous feeder cells. J Immunol 1997; 159: 5921–5930.
Weng N-P, Levine BL, June CH et al. Human naive and memory T lymphocytes differ in telomeric length and replicative potential. Proc Natl Acad Sci USA 1995; 92: 11091–11094.
Epel ES, Blackburn EH, Lin J et al. Accelerated telomere shortening in response to life stress. Proc Natl Acad Sci USA 2004; 101: 17312–17315.
Weng N-P, Palmer LD, Levine BL et al. Tales of tails: regulation of telomere length and telomerase activity during lymphocyte development, differentiation, activation, and aging. Immunol Rev 1997; 160: 43–54.
Wynn R, Thornley I, Freedman M et al. Telomere shortening in leucocyte subsets of long-term survivors of allogeneic bone marrow transplantation. Br J Haematol 1999; 105: 997–1001.
Akbar AN, Beverley PC, Salmon M . Will telomere erosion lead to a loss of T-cell memory? Nat Rev Immunol 2004; 4: 737–743.
Verra NC, Jorritsma A, Weijer K et al. Human telomerase reverse transcriptase-transduced human cytotoxic T cells suppress the growth of human melanoma in immunodeficient mice. Cancer Res 2004; 64: 2153–2161.
Freitas AA, Rocha B . Peripheral T cell survival. Curr Opin Immunol 1999; 11: 152–156.
Ku CC, Murakami M, Sakamoto A et al. Control of homeostasis of CD8+ memory T cells by opposing cytokines. Science 2000; 288: 675–678.
Freitas AA, Rocha BB . Lymphocyte lifespans: homeostasis, selection and competition. Immunol Today 1995; 14: 25–29.
Michie CA, McLean A, Alcock C et al. Lifespan of human lymphocyte subsets defined by CD45 isoforms. Nature 1992; 360: 264–265.
Hellerstein MK . Measurement of T-cell kinetics: recent methodologic advances. Immunol Today 1999; 20: 438–441.
Hellerstein M, Hanley MB, Cesar D et al. Directly measured kinetics of circulating T lymphocytes in normal and HIV-1-infected humans. Nat Med 1999; 5: 83–89.
Mclean AR, Michie CA . In vivo estimates of division and death rates of human T lymphocytes. Proc Natl Acad Sci USA 1995; 92: 3707–3711.
McCune JM, Hanley MB, Cesar D et al. Factors influencing T-cell turnover in HIV-1-seropositive patients. J Clin Invest 2000; 105: R1–R8.
Blaese RM, Culver KW, Miller AD et al. T lymphocyte-directed gene therapy for ADA-SCID: initial trial results after 4 years. Science 1995; 270: 475–480.
Heslop HE, Ng CY, Li C et al. Long-term restoration of immunity against Epstein–Barr virus infection by adoptive transfer of gene-modified virus-specific T lymphocytes. Nat Med 1996; 2: 551–555.
Walker RE, Carter CS, Muul L et al. Peripheral expansion of pre-existing mature T cells is an important means of CD4+ T-cell regeneration HIV-infected adults. Nat Med 1998; 4: 852–856.
Riddell SR, Elliott M, Lewinsohn DA et al. T-cell mediated rejection of gene-modified HIV-specific cytotoxic T lymphocytes in HIV-infected patients. Nat Med 1996; 2: 216–223.
Mitsuyasu RT, Anton P, Deeks SG et al. Prolonged survival and tissue trafficking following adoptive transfer of CD4ζ gene-modified autologous CD4+ and CD8+ T cells in HIV-infected subjects. Blood 2000; 96: 785–793.
Hamilton SE, Prlic M, Jameson SC . Environmental conservation: bystander CD4 T cells keep CD8 memories fresh. Nat Immunol 2004; 5: 873–874.
Rosenberg SA, Aebersold P, Cornetta K et al. Gene transfer into humans – immunotherapy of patients with advanced melanoma, using tumor-infiltrating lymphocytes modified by retroviral gene transduction. N Engl J Med 1990; 323: 570–578.
Yee C, Thompson JA, Byrd D et al. Adoptive T cell therapy using antigen-specific CD8+ T cell clones for the treatment of patients with metastatic melanoma: in vivo persistence, migration, and antitumor effect of transferred T cells. Proc Natl Acad Sci USA 2002; 99: 16168–16173.
Robbins PF, Dudley ME, Wunderlich J et al. Cutting edge: persistence of transferred lymphocyte clonotypes correlates with cancer regression in patients receiving cell transfer therapy. J Immunol 2004; 173: 7125–7130.
Topp MS, Riddell SR, Akatsuka Y et al. Restoration of CD28 expression in CD28- CD8+ memory effector T cells reconstitutes antigen-induced IL-2 production. J Exp Med 2003; 198: 947–955.
Degauque N, Schadendorf D, Brouard S et al. Blood T-cell Vbeta transcriptome in melanoma patients. Int J Cancer 2004; 110: 721–729.
Kircher MF, Allport JR, Graves EE et al. In vivo high resolution three-dimensional imaging of antigen-specific cytotoxic T-lymphocyte trafficking to tumors. Cancer Res 2003; 63: 6838–6846.
Arbab AS, Yocum GT, Kalish H et al. Efficient magnetic cell labeling with protamine sulfate complexed to ferumoxides for cellular MRI. Blood 2004; 104: 1217–1223.
Mackall CL, Fleisher TA, Brown MR et al. Distinctions between CD8(+) and CD4(+) T-cell regenerative pathways result in prolonged T-cell subset imbalance after intensive chemotherapy. Blood 1997; 89: 3700–3707.
Verfuerth S, Peggs K, Vyas P et al. Longitudinal monitoring of immune reconstitution by CDR3 size spectratyping after T-cell-depleted allogeneic bone marrow transplant and the effect of donor lymphocyte infusions on T-cell repertoire. Blood 2000; 95: 3990–3995.
Bomberger C, Singh-Jairam M, Rodey G et al. Lymphoid reconstitution after autologous PBSC transplantation with FACS-sorted CD34(+) hematopoietic progenitors. Blood 1998; 91: 2588–2600.
Benyunes MC, Massumoto C, York A et al. Interleukin-2 with or without lymphokine-activated killer cells as consolidative immunotherapy after autologous bone marrow transplantation for acute myelogenous leukemia. Bone Marrow Transplant 1993; 12: 159–163.
Hamon MD, Prentice HG, Gottlieb DJ et al. Immunotherapy with interleukin 2 after ABMT in AML. Bone Marrow Transplant 1993; 11: 399–401.
Soiffer RJ, Murray C, Cochran K et al. Clinical and immunologic effects of prolonged infusion of low-dose recombinant interleukin-2 after autologous and T-cell-depleted allogeneic bone marrow transplantation. Blood 1992; 79: 517–526.
Burns LJ, Weisdorf DJ, DeFor TE et al. IL-2-based immunotherapy after autologous transplantation for lymphoma and breast cancer induces immune activation and cytokine release: a phase I/II trial. Bone Marrow Transplant 2003; 32: 177–186.
Soiffer RJ, Murray C, Gonin R et al. Effect of low-dose interleukin-2 on disease relapse after T-cell-depleted allogeneic bone marrow transplantation. Blood 1994; 84: 964–971.
Slavin S, Naparstek E, Nagler A et al. Allogeneic cell therapy: the treatment of choice for all hematologic malignancies relapsing post BMT. Blood 1996; 87: 4011–4013.
Nadal E, Fowler A, Kanfer E et al. Adjuvant interleukin-2 therapy for patients refractory to donor lymphocyte infusions. Exp Hematol 2004; 32: 218–223.
Levine BL, Mosca J, Riley JL et al. Antiviral effect and ex vivo CD4+ T cell proliferation in HIV-positive patients as a result of CD28 costimulation. Science 1996; 272: 1939–1943.
Kalamasz D, Long SA, Taniguchi R et al. Optimization of human T-cell expansion ex vivo using magnetic beads conjugated with anti-CD3 and Anti-CD28 antibodies. J Immunother 2004; 27: 405–418.
King CL, Stupi RJ, Craighead N et al. CD28 stimulates IL-4 and IL-5 production and promotes Th2 subset differentiation by human CD4+ cells. Eur J Immunol 1995; 25: 587–595.
Broeren C, Gray GS, Carreno BM et al. Costimulation light: activation of of CD4+ T-cells with CD80 or CD86 rather than anti-CD28 Leads to a Th2 cytokine profile. J Immunol 2000; 165: 6908–6914.
Rapoport AP, Stadtmauer EA, Levine BL et al. Adoptive transfer of ex vivo costimulated autologous T-Cells in conjunction with pneumococcal conjugate vaccine immunizations accelerates post-transplant T-cell recovery after autotransplantation for myeloma: results of a randomized study. Blood 2004; 104: 128a abstract 439.
Porter D, Levine J, Kolb HJ . Adoptive immunotherapy in stem cell transplantation. In: Ferrara J, Cooke KR, Deeg HJ (eds) Graft-vs-Host Disease. Marcel Dekker: New York, 2004 pp 525–553.
Porter D, Levine B, Bunin N et al. Enhancing graft-vs-tumor activity of donor leukocyte infusions by ex-vivo expansion and activation with CD3/CD28 co-stimulation. Blood 2004; 104: 456a abstract #1640.
Rossi S, Blazar BR, Farrell CL et al. Keratinocyte growth factor preserves normal thymopoiesis and thymic microenvironment during experimental graft-versus-host disease. Blood 2002; 100: 682–691.
Spielberger R, Stiff P, Bensinger W et al. Palifermin for oral mucositis after intensive therapy for hematologic cancers. N Engl J Med 2004; 351: 2590–2598.
Schluns KS, Kieper WC, Jameson SC et al. Interleukin-7 mediates the homeostasis of naive and memory CD8 T cells in vivo. Nat Immunol 2000; 1: 426–432.
Jicha DL, Mule JJ, Rosenberg SA . Interleukin 7 generates antitumor cytotoxic T lymphocytes against murine sarcomas with efficacy in cellular adoptive immunotherapy. J Exp Med 1991; 174: 1511–1515.
Sinha ML, Fry TJ, Fowler DH et al. Interleukin 7 worsens graft-versus-host disease. Blood 2002; 100: 2642–2649.
Fry TJ, Moniuszko M, Creekmore S et al. IL-7 therapy dramatically alters peripheral T-cell homeostasis in normal and SIV-infected nonhuman primates. Blood 2003; 101: 2294–2299.
Riddell SR, Watanabe KS, Goodrich JM et al. Restoration of viral immunity in immunodeficient humans by the adoptive transfer of T cell clones. Science 1992; 257: 238–241.
Walter EA, Greenberg PD, Gilbert MJ et al. Reconstitution of cellular immunity against cytomegalovirus in recipients of allogeneic bone marrow by transfer of T-cell clones from the donor. N Engl J Med 1995; 333: 1038–1044.
Einsele H, Roosnek E, Rufer N et al. Infusion of cytomegalovirus (CMV)-specific T cells for the treatment of CMV infection not responding to antiviral chemotherapy. Blood 2002; 99: 3916–3922.
Heslop HE, Rooney CM . Adoptive cellular immunotherapy for EBV lymphoproliferative disease. Immunol Rev 1997; 157: 217–222.
O'Reilly RJ, Small TN, Papadopoulos E et al. Biology and adoptive cell therapy of Epstein–Barr virus-associated lymphoproliferative disorders in recipients of marrow allografts. Immunol Rev 1997; 157: 195–216.
Slavin S, Nagler A, Naparstek E et al. Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic diseases. Blood 1998; 91: 756–763.
Dazzi F, Szydlo RM, Craddock C et al. Comparison of single-dose and escalating-dose regimens of donor lymphocyte infusion for relapse after allografting for chronic myeloid leukemia. Blood 2000; 95: 67–71.
Bishop MR, Fowler DH, Marchigiani D et al. Allogeneic lymphocytes induce tumor regression of advanced metastatic breast cancer. J Clin Oncol 2004; 22: 3886–3892.
Rapoport AP, Levine BL, Badros A et al. Molecular remission of CML after autotransplantation followed by adoptive transfer of costimulated autologous T cells. Bone Marrow Transplant 2004; 33: 53–60.
Fowler DH, Bishop MR, Gress RE . Immunoablative reduced- intensity stem cell transplantation: potential role of donor Th2 and Tc2 cells. Semin Oncol 2004; 31: 56–67.
Jung U, Foley JE, Erdmann AA et al. CD3/CD28-costimulated T1 and T2 subsets: differential in vivo allosensitization generates distinct GVT and GVHD effects. Blood 2003; 102: 3439–3446.
Bonini C, Ferrari G, Verzeletti S et al. HSV-TK gene transfer into donor lymphocytes for control of allogeneic graft-versus-leukemia. Science 1997; 276: 1719–1724.
Berger C, Blau CA, Huang ML et al. Pharmacologically regulated Fas-mediated death of adoptively transferred T cells in a nonhuman primate model. Blood 2004; 103: 1261–1269.
Bachar-Lustig E, Reich-Zeliger S, Reisner Y . Anti-third-party veto CTLs overcome rejection of hematopoietic allografts: synergism with rapamycin and BM cell dose. Blood 2003; 102: 1943–1950.
McFarland HI, Hansal SA, Morris DI et al. Signaling, through MHC in transgenic mice generates a population of memory phenotype cytolytic cells that lack TCR. Blood 2003; 101: 4520–4528.
Fowler DH, Gress RE . Th2 and Tc2 cells in the regulation of GVHD, GVL, and graft rejection: considerations for the allogeneic transplantation therapy of leukemia and lymphoma. Leuk Lymphoma 2000; 38: 221–234.
Fink PJ, Rammensee HG, Benedetto JD et al. Studies on the mechanism of suppression of primary cytotoxic responses by cloned cytotoxic T lymphocytes. J Immunol 1984; 133: 1769–1774.
Eshhar Z, Waks T, Bendavid A et al. Functional expression of chimeric receptor genes in human T cells. J Immunol Methods 2001; 248: 67–76.
Sadelain M, Riviere I, Brentjens R . Targeting tumours with genetically enhanced T lymphocytes. Nat Rev Cancer 2003; 3: 35–45.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Porter, D., June, C. T-cell reconstitution and expansion after hematopoietic stem cell transplantation: ‘T’ it up!. Bone Marrow Transplant 35, 935–942 (2005). https://doi.org/10.1038/sj.bmt.1704953
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704953
Keywords
This article is cited by
-
Venetoclax imparts distinct cell death sensitivity and adaptivity patterns in T cells
Cell Death & Disease (2021)
-
Interleukin-21 promotes thymopoiesis recovery following hematopoietic stem cell transplantation
Journal of Hematology & Oncology (2017)
-
Generation of multi-leukemia antigen-specific T cells to enhance the graft-versus-leukemia effect after allogeneic stem cell transplant
Leukemia (2013)
-
A Novel Human Glycoprotein ACA is an Upstream Regulator of Human Hematopoiesis
Bulletin of Experimental Biology and Medicine (2013)
-
Integration of autologous dendritic cell-based immunotherapy in the standard of care treatment for patients with newly diagnosed glioblastoma: results of the HGG-2006 phase I/II trial
Cancer Immunology, Immunotherapy (2012)