Summary:
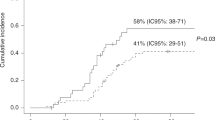
The majority of invasive aspergillosis (IA) in allogeneic stem cell transplantation (SCT) occurs during the post-engraftment period. We used Cox proportional hazards regression to evaluate post-engraftment IA risk in a cohort of 217 allogeneic SCT recipients from 1991 to 1998. The aim was to quantify the effects of dose-intensity and duration of corticosteroids and other risk factors. Median duration of follow-up was 330 days. There were 19 cases of IA (overall 8.8%) with 14 post-engraftment infections. In the final model, the risk of IA was greatest within 2 weeks of high-dose corticosteroids (HR 8.5, P=0.003), with risk extending to 4 weeks with doses of 0.25–1 mg/kg/day (HR 3.1, P=0.08). Ganciclovir was associated with greatest risk (HR 13.6). Grade 3 or 4 acute GVHD (HR 5.7) and secondary neutropenia (HR=1.3) were also additive risks. In the univariate analysis, corticosteroid doses of 0.25–1.0 mg/kg/day for any duration between 2 and 10 weeks demonstrated prolonged risk for IA. Moderate doses of corticosteroids can confer an increased risk for IA for extended periods which is almost as marked as that conferred by higher doses. Knowledge of these risks may facilitate the development of targeted surveillance and prophylaxis strategies for prevention of IA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Jantunen E, Ruutu P, Niskanen L et al. Incidence and risk factors for invasive fungal infections in allogeneic BMT recipients. Bone Marrow Transplant 1997; 19: 801–808.
Marr KA, Carter RA, Boeckh M et al. Invasive aspergillosis in allogeneic stem cell transplant recipients: changes in epidemiology and risk factors. Blood 2002; 100: 4358–4366.
Grow WB, Moreb JS, Roque D et al. Late onset of invasive aspergillus infection in bone marrow transplant patients at a university hospital. Bone Marrow Transplant 2002; 29: 15–19.
Baddley JW, Stroud TP, Salzman D et al. Invasive mold infections in allogeneic bone marrow transplant recipients. Clin Infect Dis 2001; 32: 1319–1324.
Singh N . Trends in the epidemiology of opportunistic fungal infections: predisposing factors and the impact of antimicrobial use practices. Clin Infect Dis 2001; 33: 1692–1696.
Junghanss C, Marr KA, Carter RA et al. Incidence and outcome of bacterial and fungal infections following nonmyeloablative compared with myeloablative allogeneic hematopoietic stem cell transplantation: a matched control study. Biol Blood Marrow Transplant 2002; 8: 512–520.
Jantunen E, Anttila VJ, Ruutu T . Aspergillus infections in allogeneic stem cell transplant recipients: have we made any progress? Bone Marrow Transplant 2002; 30: 925–929.
Winston DJ, Maziarz RT, Chandrasekar PH et al. Intravenous and oral itraconazole versus intravenous and oral fluconazole for long-term antifungal prophylaxis in allogeneic hematopoietic stem-cell transplant recipients. A multicenter, randomized trial. Ann Intern Med 2003; 138: 705–713.
O'Donnell MR, Schmidt GM, Tegtmeier BR et al. Prediction of systemic fungal infection in allogeneic marrow recipients: impact of amphotericin prophylaxis in high-risk patients. J Clin Oncol 1994; 12: 827–834.
Ribaud P, Chastang C, Latge JP et al. Survival and prognostic factors of invasive aspergillosis after allogeneic bone marrow transplantation. Clin Infect Dis 1999; 28: 322–330.
Boeckh M, Zaia JA, Jung D et al. A study of the pharmacokinetics, antiviral activity, and tolerability of oral ganciclovir for CMV prophylaxis in marrow transplantation. Biol Blood Marrow Transplant 1998; 4: 13–19.
Thomas ED, Storb R, Clift RA et al. Bone-marrow transplantation (second of two parts). N Engl J Med 1975; 292: 895–902.
Thomas E, Storb R, Clift RA et al. Bone-marrow transplantation (first of two parts). N Engl J Med 1975; 292: 832–843.
Nash R . Hematopoetic stem cell transplantation. In: Lee G, Foerston J, Lukens J (eds). Wintrobe's Clinical Hematology, 10th edn. Lippincott Williams and Wilkins: Philadelphia, 1999; pp. 882–884.
Ljungman P, Griffiths P, Paya C . Definitions of cytomegalovirus infection and disease in transplant recipients. Clin Infect Dis 2002; 34: 1094–1097.
Ascioglu S, Rex JH, de Pauw B et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis 2002; 34: 7–14.
Klein J, Moeschberger M . Time dependent covariates. In: Survival Analysis: Techniques for Censored and Truncated Data. Springer: New York, 2003; (Chapter 9.2).
Stata Corp. Stata Statistical Software. In: Release 6.0 ed. College Station, TX: Stata Corporation; 1997.
Stuck AE, Minder CE, Frey FJ . Risk of infectious complications in patients taking glucocorticosteroids. Rev Infect Dis 1989; 11: 954–963.
Boeckh M, Leisenring W, Riddell SR et al. Late cytomegalovirus disease and mortality in recipients of allogeneic hematopoietic stem cell transplants: importance of viral load and T-cell immunity. Blood 2003; 101: 407–414.
Bowden RA, Digel J, Reed EC et al. Immunosuppressive effects of ganciclovir on in vitro lymphocyte responses. J Infect Dis 1987; 156: 899–903.
Einsele H, Hebart H, Kauffmann-Schneider C et al. Risk factors for treatment failures in patients receiving PCR-based preemptive therapy for CMV infection. Bone Marrow Transplant 2000; 25: 757–763.
Slavin MA, Osborne B, Adams R et al. Efficacy and safety of fluconazole prophylaxis for fungal infections after marrow transplantation – a prospective, randomized, double-blind study. J Infect Dis 1995; 171: 1545–1552.
Acknowledgements
Many thanks to Peter and Ann-Marie Sherman who collected all data for this study, and to Thao Nguyen for data entry and validation. Supported by a grant from Gilead Sciences (MS). The supporting source had no role in the study design, data collection and analysis, or the interpretation and writing of the manuscript. The authors declare no competing interests with regard to the material published in this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thursky, K., Byrnes, G., Grigg, A. et al. Risk factors for post-engraftment invasive aspergillosis in allogeneic stem cell transplantation. Bone Marrow Transplant 34, 115–121 (2004). https://doi.org/10.1038/sj.bmt.1704543
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704543
Keywords
This article is cited by
-
Age difference of patients with and without invasive aspergillosis: a systematic review and meta-analysis
BMC Infectious Diseases (2024)
-
The Impact of Corticosteroids on the Outcome of Fungal Disease: a Systematic Review and Meta-analysis
Current Fungal Infection Reports (2023)
-
Foiling fungal disease post hematopoietic cell transplant: review of prophylactic strategies
Bone Marrow Transplantation (2018)
-
Immune Monitoring for CMV in Transplantation
Current Infectious Disease Reports (2018)
-
Hyperglycemia as a possible risk factor for mold infections—the potential preventative role of intensified glucose control in allogeneic hematopoietic stem cell transplantation
Bone Marrow Transplantation (2017)