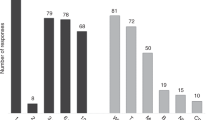
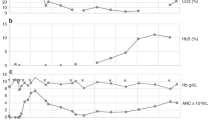
Summary:
The main goal of post-transplantation monitoring in hematopoietic stem cell transplantation (HSCT) is to predict negative events, such as disease relapse, graft rejection and graft-versus-host disease, in order to intervene with appropriate therapy. In this context, chimerism analysis is an important method in monitoring post HSCT outcome. Mixed chimerism (MC) is mainly evaluated to define engraftment and relapse. Detection of MC is a prerequisite in both myeloablative and nonmyeloablative HSCT, in order to assess the graft status and decide later therapeutic strategies such as donor lymphocyte infusion. In this review, we discuss various techniques including erythrocyte phenotyping, cytogenetic analysis, fluorescent in situ hybridization, restriction fragment length polymorphism, STR/VNTR analysis and real-time quantitative PCR, along with the various methods used to detect minimal residual disease (MRD) in different diseases such as chronic myeloid leukemia, acute myelomonocytic leukemia or acute lymphoblastic leukemia. The review mainly highlights the optimal methodological approach, which needs to be informative, sensitive and quantitatively accurate for MC detection. Future of post HSCT graft monitoring lies in the selection of the most accurate and sensitive technique to determine both MC and MRD. Such an approach would be helpful in not only determining relapse or rejection, but also in ascertaining various responses to different treatment modalities.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Rose HJ . A Handbook of Greek Mythology. Routledge: London and New York, 1989.
McCann SR, Lawler M . Mixed chimerism: detection and significance following BMT. Bone Marrow Transplant 1993; 11: 91–94.
Mapara MY, Kim YM, Marx J et al. Donor lymphocyte infusion-mediated graft-versus-leukemia effects in mixed chimeras established with a nonmyeloablative conditioning regimen: extinction of graft-versus-leukemia effects after conversion to full donor chimerism. Transplantation 2003; 76: 297–305.
Kvasnicka HM, Wickenhauser C, Thiele J et al. Mixed chimerism of bone marrow vessels (endothelial cells, myofibroblasts) following allogeneic transplantation for chronic myelogenous leukemia. Leuk Lymphoma 2003; 44: 321–328.
Elmaagacli A, Peceney R, Steckel N et al. Outcome of transplantation of highly purified peripheral blood CD34+ cells with T cells add back compared with unmanipulated bone marrow or peripheral blood stem cells from HLA identical sibling donors in first chronic phase CML. Blood 2003; 102: 446–453.
Bader P, Hancock J, Kreyenberg H et al. Minimal residual disease (MRD) status prior to allogeneic stem cell transplantation is a powerful predictor for post-transplant outcome in children with ALL. Leukemia 2002; 16: 1668–1672.
Bader P, Beck J, Frey A et al. Serial and quantitative analysis of mixed hematopoietic chimerism by PCR inpatients with acute leukemias allows the prediction of relapse after allogeneic BMT. Bone Marrow Transplant 1998; 2: 487–495.
Bader P, Holle W, Klingebiel T et al. Mixed hematopoietic chimerism after allogeneic bone marrow transplantation: the impact of quantitative PCR analysis for prediction of relapses and graft rejection in children. Bone Marrow Transplant 1997; 19: 697–702.
O'Donnell MR, Long GD, Parker PM et al. Busulfan/cyclophosphamide as conditioning regimen for allogeneic bone marrow transplantation for myelodysplasia. J Clin Oncol 1995; 13: 2973–2979.
Perreault C, Roy DC, Fortin C . Immunodominant minor histocompatibility antigens: the major ones. Immunol Today 1998; 19: 69–74.
Mutis T, Schrama E, van Luxemburg-Heijs SA et al. HLA class II restricted T-cell reactivity to a developmentally regulated antigen shared by leukemic cells and CD34+ early progenitor cells. Blood 1997; 90: 1083–1090.
Castro JE, Ball ED . Development of allogeneic hematopoietic stem cell transplantation (HSCT). Cancer Treat Res 2002; 110: 1–37.
Au WY, Chan EC, Lie AK et al. Poor engraftment after allogeneic bone marrow transplantation: role of chimerism analysis in treatment and outcome. Ann Hematol 2003; 82: 410–415.
Winiarski J, Gustafsson A, Wester D et al. Follow-up of chimerism, including T- and B-lymphocytes and granulocytes in children more than one year after allogeneic bone marrow transplantation. Pediatr Transplant 2000; 4: 132–139.
Park SJ, Min WS, Yang IH et al. Effects of mixed chimerism and immune modulation on GVHD, disease recurrence and survival after HLA-identical marrow transplantation for hematologic malignancies. Korean J Intern Med 2000; 15: 224–231.
Han D, Ricordi C, Xu X et al. Quantitative polymerase chain reaction assessment of chimerism in non-human primates after sex-mismatched islet and bone marrow transplantation. Transplantation 2000; 69: 1717–1721.
Socie G, Lawler M, Gluckman E et al. Studies on hemopoietic chimerism following allogeneic bone marrow transplantation in the molecular biology era. Leuk Res 1995; 19: 497–504.
Winiarski J, Gustafsson A, Wester D et al. Follow-up of chimerism, including T- and B-lymphocytes and granulocytes in children more than one year after allogeneic bone marrow transplantation. Pediatr Transplant 2000; 4: 132–139.
Park SJ, Min WS, Yang IH et al. Effects of mixed chimerism and immune modulation on GVHD, disease recurrence and survival after HLA-identical marrow transplantation for hematologic malignancies. Korean J Intern Med 2000; 15: 224–231.
Han D, Ricordi C, Xu X et al. Quantitative polymerase chain reaction assessment of chimerism in non-human primates after sex-mismatched islet and bone marrow transplantation. Transplantation 2000; 69: 1717–1721.
Thiele J, Wickenhauser C, Kvasnicka HM et al. Dynamics of lineage-restricted mixed chimerism following sex-mismatched allogeneic bone marrow transplantation. Histol Histopathol 2003; 18: 557–574.
Winiarski J, Gustafsson A, Wester D et al. Follow-up of chimerism, including T- and B-lymphocytes and granulocytes in children more than one year after allogeneic bone marrow transplantation. Pediatr Transplant 2000; 4: 132–139.
Blazar BR, Lees CJ, Martin PJ et al. Host T cells resist graft-versus-host disease mediated by donor leukocyte infusions. J Immunol 2000; 165: 4901–4909.
Mackinnon S, Barnett L, Heller G et al. Minimal residual disease is more common in patients who have mixed T-cell chimerism after bone marrow transplantation for chronic myelogenous leukemia. Blood 1994; 83: 3409–3416.
Choi SJ, Lee KH, Lee JH et al. Prognostic value of hematopoietic chimerism in patients with acute leukemia after allogeneic bone marrow transplantation: a prospective study. Bone Marrow Transplant 2000; 26: 327–332.
Serrano J, Roman J, Herrera C et al. Increasing mixed haematopoietic chimaerism after BMT with total depletion of CD4+ and partial depletion of CD8+ lymphocytes is associated with a higher incidence of relapse. Bone Marrow Transplant 1999; 23: 475–482.
Ramirez M, Diaz MA, Garcia-Sanchez F et al. Chimerism after allogeneic hematopoietic cell transplantation in childhood acute lymphoblastic leukemia. Bone Marrow Transplant 1996; 18: 1161–1165.
Suttorp M, Schmitz N, Dreger P et al. Monitoring of chimerism after allogeneic bone marrow transplantation with unmanipulated marrow by use of DNA polymorphisms. Leukemia 1993; 7: 679–687.
Molloy K, Goulden N, Lawler M et al. Patterns of hematopoietic chimerism following bone marrow transplantation for childhood acute lymphoblastic leukemia from volunteer unrelated donors. Blood 1996; 87: 3027–3031.
Alizadeh M, Bernard M, Danic B et al. Quantitative assessment of hematopoietic chimerism after bone marrow transplantation by real-time quantitative polymerase chain reaction. Blood 2002; 99: 4618–4625.
Elmaagacli AH, Becks HW, Beelen DW et al. Detection of minimal residual disease and persistence of host-type hematopoiesis: a study in 28 patients after sex-mismatched, non-T cell-depleted allogeneic bone marrow transplantation for Philadelphia-chromosome positive chronic myelogenous leukemia. Bone Marrow Transplant 1995; 16: 823–829.
Bertheas MF, Maraninchi D, Lafage M et al. Partial chimerism after T-cell-depleted allogeneic bone marrow transplantation in leukemic HLA-matched patients: a cytogenetic documentation. Blood 1988; 72: 89–93.
Seong CM, Giralt S, Kantarjian H et al. Early detection of relapse by hypermetaphase fluorescence in situ hybridization after allogeneic bone marrow transplantation for chronic myeloid leukemia. J Clin Oncol 2000; 18: 1831–1836.
Hibi S, Tsunamoto K, Todo S et al. Chimerism analysis on mononuclear cells in the CSF after allogeneic bone marrow transplantation. Bone Marrow Transplant 1997; 20: 503–506.
Najfeld V, Burnett W, Vlachos A et al. Interphase FISH analysis of sex-mismatched BMT utilizing dual color XY probes. Bone Marrow Transplant 1997; 19: 829–834.
Schaap N, Schattenberg A, Bar B et al. Red blood cell phenotyping is a sensitive technique for monitoring chronic myeloid leukaemia patients after T-cell-depleted bone marrow transplantation and after donor leucocyte infusion. Br J Haematol 2000; 108: 116–125.
Lo YM, Roux E, Jeannet M et al. Detection of chimaerism after bone marrow transplantation using the double amplification refractory mutation system. Br J Haematol 1993; 85: 223–226.
Buno I, Lopez-Fernandez C, Fernandez JL et al. Improving chimaerism quantification in bone marrow transplant recipients by image processing and analysis after restriction endonuclease in situ digestion (IPA-REISD). Leukemia 1996; 10: 1232–1236.
Gardiner N, Lawler M, O'Riordan J et al. Donor chimerism is a strong indicator of disease free survival following bone marrow transplantation for chronic myeloid leukemia. Leukemia 1997; 11 (Suppl 3): 512–515.
Hendriks EC, de Man AJ, van Berkel YC et al. Flow cytometric method for the routine follow-up of red cell populations after bone marrow transplantation. Br J Haematol 1997; 97: 141–145.
Petz LD, Calhoun L, Shulman IA et al. The sickle cell hemolytic transfusion reaction syndrome. Transfusion 1997; 37: 382–392.
Dewald GW, Schad CR, Christensen ER et al. Fluorescence in situ hybridization with X and Y chromosome probes for cytogenetic studies on bone marrow cells after opposite sex transplantation. Bone Marrow Transplant 1993; 12: 149–154.
Knuutila S, Majander P, Ruutu T . 8;21 and 15;17 translocations: abnormalities in a single cell lineage in acute myeloid leukemia. Acta Haematol 1994; 92: 88–90.
van Lom K, Hagemeijer A, Smit EM et al. In situ hybridization on May–Grunwald Giemsa-stained bone marrow and blood smears of patients with hematologic disorders allows detection of cell-lineage-specific cytogenetic abnormalities. Blood 1993; 82: 884–888.
Jolkowska J, Wachowiak J, Lange A et al. Molecular assessment of post-BMT chimerism using various biologic specimens and automated DNA sizing technology. J Hematother Stem Cell Res 2000; 9: 263–268.
Newton CR, Heptinstall LE, Summers C et al. Amplification refractory mutation system for prenatal diagnosis and carrier assessment in cystic fibrosis. Lancet 1989; 2: 1481–1483.
Gosalvez J, Lopez-Fernandez C, Buno I et al. Restriction endonuclease in situ digestion (REISD) and fluorescence in situ hybridization (FISH) as complementary methods to analyze chimerism and residual disease after bone marrow transplantation. Cancer Genet Cytogenet 1996; 89: 141–145.
Elmaagacli AH, Beelen DW, Kroll M et al. Detection of CBFbeta/MYH11 fusion transcripts in patients with inv(16) acute myeloid leukemia after allogeneic bone marrow or peripheral blood progenitor cell transplantation. Bone Marrow Transplant 1998; 21: 159–166.
Serrano J, Roman J, Sanchez J et al. Molecular analysis of lineage-specific chimerism and minimal residual disease by RT-PCR of p210(BCR-ABL) and p190(BCR-ABL) after allogeneic bone marrow transplantation for chronic myeloid leukemia: increasing mixed myeloid chimerism and p190(BCR-ABL) detection precede cytogenetic relapse. Blood 2000; 95: 2659–2665.
Brunstein CG, Hirsch BA, Miller JS et al. Non-leukemic autologous reconstitution after allogeneic bone marrow transplantation for Ph-positive chronic myelogenous leukemia: extended remission preceding eventual relapse. Bone Marrow Transplant 2000; 26: 1173–1177.
Miyamura K, Barrett AJ, Kodera Y et al. Minimal residual disease after bone marrow transplantation for chronic myelogenous leukemia and implications for graft-versus-leukemia effect: a review of recent results. Bone Marrow Transplant 1994; 14: 201–209.
Okuda T, van Deursen J, Hiebert SW et al. AML1, the target of multiple chromosomal translocations in human leukemia, is essential for normal fetal liver hematopoiesis. Cell 1996; 84: 321–330.
Shurtleff SA, Meyers S, Hiebert SW et al. Heterogeneity in CBF beta/MYH11 fusion messages encoded by the inv(16)(p13q22) and the t(16;16)(p13;q22) in acute myelogenous leukemia. Blood 1995; 85: 3695–3703.
Le Beau MM, Larson RA, Bitter MA et al. Association of an inversion of chromosome 16 with abnormal marrow eosinophil in acute myelomonocytic leukemia. A unique cytogenetic-clinicopathological association. N Engl J Med 1983; 309: 630–636.
Weinberg RS, Vlachos A, Najfeld V et al. Disparate lympho-erythroid donor to recipient chimaerism in a beta (0)-thalassaemia bone marrow transplant recipient with red cell indices indicative of apparent full engraftment. Br J Haematol 1997; 99: 61–63.
Andreani M, Manna M, Lucarelli G et al. Persistence of mixed chimerism in patients transplanted for the treatment of thalassemia. Blood 1996; 87: 3494–3499.
Kapelushnik J, Naparstek E, Nagler A et al. Second transplantation using allogeneic peripheral blood stem cells in a beta-thalassaemia major patient featuring stable mixed chimaerism. Br J Haematol 1996; 94: 285–287.
Beishuizen A, Verhoeven MA, van Wering ER et al. Analysis of Ig and T-cell receptor genes in 40 childhood acute lymphoblastic leukemias at diagnosis and subsequent relapse: implications for the detection of minimal residual disease by polymerase chain reaction analysis. Blood 1994; 3: 2238–2247.
Norris MD, Kwan E, Haber M et al. Detection of evolving immunoglobulin heavy-chain gene rearrangements in acute lymphoblastic leukemia: a PCR-based assay employing overlapping DJH primers. Leukemia 1995; 9: 1779–1782.
Steward CG, Goulden NJ, Katz F et al. A polymerase chain reaction study of the stability of Ig heavy-chain and T-cell receptor delta gene rearrangements between presentation and relapse of childhood B-lineage acute lymphoblastic leukemia. Blood 1994; 83: 1355–1362.
Forbes GM, Fogarty J, Meyer B et al. Intestinal mucosal mononuclear cell chimaerism after sex-mismatched allogeneic bone marrow transplantation. Bone Marrow Transplant 1995; 16: 589–593.
Garcia-Morales R, Esquenazi V, Zucker K et al. An assessment of the effects of cadaver donor bone marrow on kidney allograft recipient blood cell chimerism by a novel technique combining PCR and flow cytometry. Transplantation 1996; 62: 1149–1160.
van Leeuwen JE, van Tol MJ, Joosten AM et al. Persistence of host-type hematopoiesis after allogeneic bone marrow transplantation for leukemia is significantly related to the recipient's age and/or the conditioning regimen, but it is not associated with an increased risk of relapse. Blood 1994; 83: 3059–3067.
Briones J, Urbano-Ispizua A, Lawler M et al. High frequency of donor chimerism after allogeneic transplantation of CD34+-selected peripheral blood cells. Exp Hematol 1998; 26: 415–420.
Lo YM, Noakes L, Roux E et al. Application of a polymorphic Y microsatellite to the detection of post bone marrow transplantation chimaerism. Br J Haematol 1995; 89: 645–649.
Hancock JP, Burgess MF, Goulden NJ et al. Same-day determination of chimaeric status in the immediate period following allogeneic bone marrow transplantation. Br J Haematol 1997; 99: 403–409.
Nakao S, Nakatsumi T, Chuhjo T et al. Analysis of late graft failure after allogeneic bone marrow transplantation: detection of residual host cells using amplification of variable number of tandem repeats loci. Bone Marrow Transplant 1992; 9: 107–111.
Ortega M, Escudero T, Caballin MR et al. Follow-up of chimerism in children with hematological diseases after allogeneic hematopoietic progenitor cell transplants. Bone Marrow Transplant 1999; 24: 81–87.
Palka G, Stuppia L, Di Bartolomeo P et al. FISH detection of mixed chimerism in 33 patients submitted to bone marrow transplantation. Bone Marrow Transplant 1996; 17: 231–236.
Oyama Y, Traynor AE, Barr W et al. Allogeneic stem cell transplantation for autoimmune diseases: nonmyeloablative conditioning regimens. Bone Marrow Transplant 2003; 32 (Suppl 1): S81–S83.
Childs R, Clave E, Contentin N et al. Engraftment kinetics after non-myeloablative allogeneic peripheral blood stem cell transplantation: full donor T-cell chimerism precedes alloimmune responses. Blood 1999; 94: 3234–3241.
Antin JH, Childs R, Filipovich AH et al. Establishment of complete and mixed donor chimerism after allogeneic lymphohematopoietic transplantation: recommendations from a workshop at the 2001 Tandem meetings. Biol Blood Marrow Transplant 2001; 7: 473–485.
Bacigalupo A, McCann SR, Lawler M . Recurrence of Philadelphia chromosome-positive leukemia in donor cells after bone marrow transplantation for chronic granulocytic leukemia. Leuk Lymphoma 1993; 10: 419–425.
Collins RH, Shpilberg O, Drobyski WR et al. Donor leukocyte infusions in 140 patients with relapsed malignancy after allogeneic bone marrow transplantation. J Clin Oncol 1997; 15: 433–444.
Slavin S, Naparstek E, Nagler A et al. Allogeneic cell therapy for relapsed leukemia after bone marrow transplantation with donor peripheral blood lymphocytes. Exp Hematol 1995; 23: 1553–1562.
Drobyski WR, Roth MS, Thibodeau SN et al. Molecular remission occurring after donor leukocyte infusions for the treatment of relapsed chronic myelogenous leukemia after allogeneic bone marrow transplantation. Bone Marrow Transplant 1992; 10: 301–304.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khan, F., Agarwal, A. & Agrawal, S. Significance of chimerism in hematopoietic stem cell transplantation: new variations on an old theme. Bone Marrow Transplant 34, 1–12 (2004). https://doi.org/10.1038/sj.bmt.1704525
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704525
Keywords
This article is cited by
-
Chimerism analysis for clinicians: a review of the literature and worldwide practices
Bone Marrow Transplantation (2022)
-
Beyond chimerism analysis: methods for tracking a new generation of cell-based medicines
Bone Marrow Transplantation (2020)
-
Alternatives to amelogenin markers for sex determination in humans and their forensic relevance
Molecular Biology Reports (2020)
-
DNA methylation dynamic of bone marrow hematopoietic stem cells after allogeneic transplantation
Stem Cell Research & Therapy (2019)
-
Obstructive sleep apnea and craniofacial appearance in MPS type I-Hurler children after hematopoietic stem cell transplantation
Sleep and Breathing (2019)