Summary:
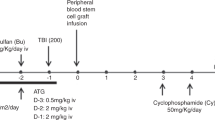
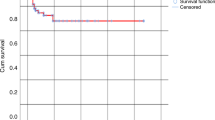
Graft rejection is a major cause of treatment failure after T-cell-depleted stem cell transplantation (TCD-SCT) and remains a therapeutic challenge. Donor leukocyte infusions (DLIs) have an efficient graft versus host effect, which has been successfully used to treat recipient relapses. We hypothesized that this effect could be exploited to counteract the host versus graft reactions responsible for graft rejection. We report two adult patients with haematological malignancies who underwent sex-mismatched TCD-SCT from HLA-identical sibling donors. Peripheral blood (PB) counts and bone marrow (BM) cellularity were studied on a serial basis. Sequential chimaerism and minimal residual disease analysis were performed by FISH on PB and BM samples as well as on leukocyte lineages (T and B lymphocytes and myeloid cells) purified from PB using immunomagnetic technology. Both patients were diagnosed with incipient graft rejection 2–3 months after engraftment, based on persistently decreasing PB counts and BM cellularity together with the observation of decreasing mixed chimaerism (increasing percentage of recipient cells), mostly in whole PB and T lymphocytes. Both patients were successfully treated with a single DLI (1 × 107 CD3+ cells/kg), thereafter achieving normal PB counts and BM cellularity as well as complete chimaerism. Interestingly, the only side effect observed was mild graft versus host disease that did not require treatment. In conclusion, provided that an early diagnosis is made, the graft versus host lymphohaemopoietic effect harboured by immunocompetent donor cells can be successfully used for the treatment of incipient graft rejection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Armitage J . Bone marrow transplantation. New Eng J Med 1994; 330: 827–838.
Marmont A, Horowitz MM, Gale RP et al. T-cell depletion of HLA-identical transplants in leukemia. Blood 1991; 78: 2120–2130.
Goldman JM, Gale RP, Horowitz MM et al. Bone marrow transplantation for chronic myelogenous leukemia in chronic phase. Increased risk for relapse with T-cell depletion. Ann Int Med 1988; 108: 806–814.
Patterson J, Prentice HG, Brenner MK et al. Graft rejection following HLA-matched T-lymphocyte depleted bone marrow transplantation. Br J Haematol 1986; 63: 221–230.
Chen J, Law P, Ball ED . Late graft failure. In: Ball ED, Lister J, Law P (eds.). Haematopoietic Stem Cell Therapy. Churchill Livingstone, London, 2000; pp 603–607.
Wolff SN . Second haematopoietic stem cell transplantation for the treatment of graft failure, graft rejection or relapse after allogeneic transplantation. Bone Marrow Transplant 2002; 9: 45–52.
Antin JH, Childs R, Filipovich AH et al. Establishment of complete and mixed donor chimaerism after allogeneic lymphohemopoietic transplantation: Recommendations from a workshop at the 2001 Tandem Meetings. Biol Blood Marrow Transplant 2001; 7: 473–485.
Buño I, Anta B, Moreno-López E et al. Lineage-specific chimaerism quantification after T-cell depleted peripheral blood stem cell transplantation. Leuk Lymphoma 2003; 44: 659–667.
Dubovski J, Daxberger H, Fritsch G et al. Kinetics of chimaerism during the early post-transplant period in pediatric patients with malignant and non-malignant haematologic disorders: implications for a timely detection of engraftment, graft failure and rejection. Leukemia 1999; 13: 2060–2069.
Urbano-Ispizua A, Rozman C, Pimentel P et al. The number of CD3+ cells is the most important factor for graft failure after allogeneic transplantation of CD34+ selected cells from peripheral blood from HLA-identical siblings. Blood 2001; 97: 383–387.
Kernan NA, Bordignon C, Heller G et al. Graft failure after T-cell-depleted human leukocyte antigen identical marrow transplants for leukemia: I. Analysis of risk factors and results of secondary transplants. Blood 1989; 74: 2227–2236.
Papadopoulos ED, Carabasi MH, Castro-Malaspina H et al. T-cell-depleted allogeneic bone marrow transplantation as postremission therapy for acute myelogenous leukemia: freedom from relapse in the absence of graft-versus-host disease. Blood 1998; 91: 1083–1090.
Aversa F, Tabilio A, Velardi A et al. Treatment of high-risk acute leukemia with T-cell-depleted stem cells from related donors with one fully mismatched HLA haplotype. New Eng J Med 1998; 339: 1186–1193.
Champlin RE, Horowitz MM, van Bekkum DW et al. Graft failure following bone marrow transplantation for severe aplastic anemia: risk factors and treatment results. Blood 1989; 73: 606–613.
McSweeney P, Storb R . Mixed chimaerism: preclinical studies and clinical applications. Biol Blood Marrow Transplant 1999; 5: 192–203.
Childs R, Clave E, Contentin N et al. Engraftment kinetics after nonmyeloablative allogeneic peripheral blood stem cell transplantation: full donor T-cell chimaerism precedes alloimmune responses. Blood 1999; 94: 3234–3241.
Higano CS, Brixey M, Bryant EM et al. Durable complete remission of acute nonlymphocytic leukemia associated with discontinuation of immunosupression following relapse after allogeneic bone marrow transplantation. Transplantation 1990; 50: 175–177.
Sykes M, Sachs D, Spitzer T . Mixed chimaerism followed by DLI for lymphohaematopoietic GVH reactions without GVHD: from animal models to patients. In: Schechter GP, Hoffman R, Schrier SL, Bajus JL (eds.) Hematology 1999. American Society of Hematology, Washington, 1999; pp 405–412.
Kolb HJ, Schattenberg A, Goldman JM et al. Graft-versus-leukemia effect of donor lymphocyte transfussions in marrow grafted patients. Blood 1995; 86: 2041–2050.
Horwitz ME, Barrett AJ, Brown MR et al. Treatment of chronic granulomatous disease with nonmyeloablative conditioning and a T-cell depleted hematopoietic allograft. N Engl J Med 2001; 344: 881–888.
Barrett AJ, Mavroudis D, Tisdale J et al. T cell-depleted bone marrow transplantation and delayed T cell add-back to control acute GVHD and conserve a graft-versus-leukemia effect. Bone Marrow Transplant 1998; 21: 543–551.
Aker M, Kapelushnik J, Pugatsch T et al. Donor lymphocyte infusions to displace residual host haematopoietic cells after allogeneic bone marrow transplantation for B-thalassemia major. J Pediatric Hematol Oncol 1998; 20: 145–148.
Baron F, Dresse MF, Beguin Y . Donor lymphocyte infusion to eradicate recurrent host haematopoiesis after allogeneic BMT for sickle cell disease. Transfusion 2000; 40: 1071–1073.
Saarinen-Pihkala UM, Taskinen M, Vettenranta K, Hovi L . Inminent allograft rejection prevented by donor leukocyte infusions: report of two paediatric cases. Bone Marrow Transplant 2003; 31: 833–836.
Schattenberg A, Schaap N, Van De Wiel-Van Kemenade E et al. In relapsed patients after lymphocyte depleted bone marrow transplantation the percentage of donor T lymphocytes correlates well with the outcome of donor leukocyte infusion. Leuk Lymphoma 1999; 32: 317–325.
Keil F, Haas OA, Fritsch G et al. Donor leukocyte infusion for leukemic relapse after allogeneic marrow transplantation: lack of residual donor haematopoiesis predicts aplasia. Blood 1997; 89: 3113–3117.
Mackinnon S, Papadopoulos EB, Caralasi MH et al. Adoptive immunotherapy evaluating escalating doses of donor leukocytes for relapse of chronic myeloid leukemia after bone marrow transplantation: separation of graft-versus-leukemia responses from graft-versus-host disease. Blood 1995; 86: 1261–1268.
Acknowledgements
This work was partially supported by Grants FIS01-3035 and FIS02-1177 from the Fondo de Investigación Sanitaria (Spain).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Díez-Martín, J., Gómez-Pineda, A., Serrano, D. et al. Successful treatment of incipient graft rejection with donor leukocyte infusions, further proof of a graft versus host lymphohaemopoietic effect. Bone Marrow Transplant 33, 1037–1041 (2004). https://doi.org/10.1038/sj.bmt.1704488
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704488
Keywords
This article is cited by
-
Early peripheral blood and T-cell chimerism dynamics after umbilical cord blood transplantation supported with haploidentical cells
Bone Marrow Transplantation (2014)