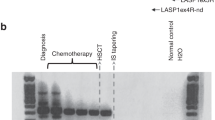
Summary:
Immunotherapy in the form of donor lymphocyte infusions in early-phase relapse might be advantageous as it induces a higher response, but this may be offset by increased toxicity, especially during the early period after transplantation. Among 45 consecutive patients receiving an allograft for CML, 13 patients were diagnosed to have molecular relapse (MRel), as defined by real-time quantitative reverse transcriptase-polymerase chain reaction, and another four patients were diagnosed to have cytogenetic relapse (CRel) within 6 months. Patients with MRel were randomly assigned to either a ‘no therapy’ group (group A, n=6), in which immunotherapy was reserved until CRel, or an ‘immunotherapy’ group (group B, n=7). In group A, all MRel progressed to CRel, and molecular remission (MR) was achieved in four (67%) after immunotherapy. The remaining two patients died of extensive GVHD and fungal pneumonia. In group B, only two MRel progressed to CRel and the remaining five (71%) achieved MR. Two patients died in the absence or loss of response. In patients relapsing directly into CRel (n=4), immunotherapy induced MR in two patients (50%). Earlier intervention played a role in preventing disease progression but this effect was not translated into better survival, which could have been overcome by imatinib mesylate, which induced MR and cytogenetic remission in nonresponders without toxicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Kurzrock R, Gutterman JU, Talpaz M . The molecular genetics of Philadelphia chromosome-positive leukemias. N Engl J Med 1988; 319: 990–998.
Melo JV . The diversity of BCR-ABL fusion proteins and their relationship to leukemia phenotype. Blood 1996; 88: 2375–2384.
Gratwohl A, Hermans J, Niederwieser D et al. Bone marrow transplantation for chronic myeloid leukemia: long-term results. Chronic Leukemia Working Party of the European Group for Bone Marrow Transplantation. Bone Marrow Transplant 1993; 12: 509–516.
Horowitz MM, Rowlings PA, Passweg JR . Allogeneic bone marrow transplantation for CML: a report from the International Bone Marrow Transplant Registry. Bone Marrow Transplant 1996; 17: S5–S6.
Kolb HJ, Schattenberg A, Goldman JM et al. Graft-versus-leukemia effect of donor lymphocyte transfusions in marrow grafted patients. European Group for Blood and Marrow Transplantation Working Party Chronic Leukemia. Blood 1995; 86: 2041–2050.
Branford S, Hughes TP, Rudzki Z . Monitoring chronic myeloid leukaemia therapy by real-time quantitative PCR in blood is a reliable alternative to bone marrow cytogenetics. Br J Haematol 1999; 107: 587–599.
Amabile M, Giannini B, Testoni N et al. Real-time quantification of different types of bcr-abl transcript in chronic myeloid leukemia. Haematologica 2001; 86: 252–259.
Emig M, Saussele S, Wittor H et al. Accurate and rapid analysis of residual disease in patients with CML using specific fluorescent hybridization probes for real time quantitative RT-PCR. Leukemia 1999; 13: 1825–1832.
Lee YK, Lee DW, Kim YL et al. Detection of the BCR-ABL gene by interphase fluorescence in situ hybridization (iFISH) in chronic myelogenous leukemia patients after hemopoietic stem cell transplantation: the feasibility of iFISH monitoring of therapeutic response in peripheral blood. Int J Hematol 2002; 76: 180–185.
Kim YJ, Kim DW, Lee S et al. Comprehensive comparison of FISH, RT-PCR, and Q-RT-PCR for monitoring the BCR-ABL gene after hematopoietic stem cell transplantation in CML. Eur J Haematol 2002; 68: 272–280.
Preudhomme C, Revillion F, Merlat A et al. Detection of BCR-ABL transcripts in chronic myeloid leukemia (CML) using a ‘real time’ quantitative RT-PCR assay. Leukemia 1999; 13: 957–964.
Eder M, Battmer K, Kafert S et al. Monitoring of BCR-ABL expression using real-time RT-PCR in CML after bone marrow or peripheral blood stem cell transplantation. Leukemia 1999; 13: 1383–1389.
van Rhee F, Savage D, Blackwell J et al. Adoptive immunotherapy for relapse of chronic myeloid leukemia after allogeneic bone marrow transplant: equal efficacy of lymphocytes from sibling and matched unrelated donors. Bone Marrow Transplant 1998; 21: 1055–1061.
Collins Jr RH, Shpilberg O, Drobyski WR et al. Donor leukocyte infusions in 140 patients with relapsed malignancy after allogeneic bone marrow transplantation. J Clin Oncol 1997; 15: 4334–4344.
Mackinnon S, Papadopoulos EB, Carabasi MH et al. Adoptive immunotherapy evaluating escalating doses of donor leukocytes for relapse of chronic myeloid leukemia after bone marrow transplantation: separation of graft-versus-leukemia responses from graft-versus-host disease. Blood 1995; 86: 1261–1268.
Formankova R, Honzatkova L, Moravcova J et al. Prediction and reversion of post-transplant relapse in patients with chronic myeloid leukemia using mixed chimerism and residual disease detection and adoptive immunotherapy. Leuk Res 2000; 24: 339–347.
Glucksberg H, Storb R, Fefer A et al. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation 1974; 18: 295–304.
Shulman HM, Sullivan KM, Weiden PL et al. Chronic graft-versus-host syndrome in man. A long-term clinicopathologic study of 20 seattle patients. Am J Med 1980, 204–217.
Hook EB . Exclusion of chromosomal mosaicism: tables of 90%, 95% and 99% confidence limits and comments on use. Am J Hum Genet 1977; 29: 94–97.
Cross NCP . Assessing residual leukaemia. Baillières Clin Haematol 1997; 10: 389–403.
van Rhee F, Lin F, Cullis JO et al. Relapse of chronic myeloid leukemia after allogeneic bone marrow transplant: the case for giving donor leukocyte transfusions before the onset of hematologic relapse. Blood 1994; 83: 3377–3383.
Porter DL, Roth MS, McGarigle C et al. Induction of graft-versus-host disease as immunotherapy for relapsed chronic myeloid leukemia. N Engl J Med 1994; 330: 100–106.
Wassmann B, Klein SA, Scheuring U et al. Hematologic and cytogenetic remission by STI571 (Glivec) in a patient relapsing with accelerated phase CML after second allogeneic stem cell transplantation. Bone Marrow Transplant 2001; 28: 721–724.
Olavarria E, Craddock C, Dazzi F et al. Imatinib mesylate (STI571) in the treatment of relapse of chronic myeloid leukemia after allogeneic stem cell transplantation. Blood 2002; 99: 3861–3862.
Kantarjian HM, O'Brien S, Cortes JE et al. Imatinib mesylate therapy for relapse after allogeneic stem cell transplantation for chronic myelogenous leukemia. Blood 2002; 100: 1590–1595.
McCann SR . Molecular response to imatinib mesylate following relapse after allogeneic SCT for CML. Blood 2003; 101: 1200–1201.
Kernan NA, Collins NH, Juliano L et al. T lymphocytes in T cell-depleted bone marrow transplants correlate with development of graft-v-host disease. Blood 1986; 68: 770–773.
Papadopoulos EB, Ladanyi M, Emanuel D et al. Infusions of donor leukocytes to treat Epstein–Barr virus-associated lymphoproliferative disorders after allogeneic bone marrow transplantation. N Engl J Med 1994; 330: 1185–1191.
Acknowledgements
We thank all staff members at the Catholic Hematopoietic Stem Cell Transplantation Center, and particularly the medical technicians for their excellent work in the molecular hematology laboratory. This work was supported by grant No. R21-2002-000-00010-0 from the Basic Research Program of the Korea Science & Engineering Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, YJ., Kim, DW., Lee, S. et al. Preemptive treatment of minimal residual disease post transplant in CML using real-time quantitative RT-PCR: a prospective, randomized trial. Bone Marrow Transplant 33, 535–542 (2004). https://doi.org/10.1038/sj.bmt.1704386
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704386