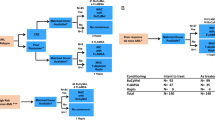
Summary:
Hematological malignancies in Jehovah's Witnesses are often difficult to cure since these patients deny transfusions. By a retrospective analysis, we report the possibility of treating some tumors, mostly hematological, with either autologous or allogeneic bone marrow transplantation (BMT) without blood support. Eight patients were evaluated, including lymphoma (two patients), acute lymphoblastic (one patient) and myeloblastic (one patient) leukemia, chronic lymphocytic leukemia (one patient), refractory anemia with blasts in transformation (one patient), chronic myeloid leukemia (one patient) and metastatic breast cancer (one patient). All patients experienced a severe cytopenia with no major side effects or life-threatening complications. We had four deaths: three from relapse and progression of the disease (at 5, 8 and 15 months after the stem cell infusion), and one from acute intestinal GVHD (at 2 months after the stem cell infusion). Four patients are in complete clinical remission (at 8, 10, 16 and 26 months after the stem cell infusion), and this was related to the disease outcome. We conclude that autologous and allogeneic BMT are feasible without the support of transfusions. We believe that this should be performed as soon as possible in the course of the disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Cullis JO, Ducombe AS, Dudley JM et al. Acute leukemia in Jehovah's Witness. Br J Haematol 1998; 100: 664–668.
Broccia G . Long-term continuous complete remission of acute myeloid leukemia in a Jehovah's Witness treated without blood support. Haematologica 1994; 79: 180–181.
Takeuchi S, Utsonomiya A, Makino T et al. Successful treatment for acute lymphoblastic leukemia without blood transfusion in a Jehovah's Witness. Am J Hematol 1999; 60: 168–169.
Fujita H, Maruta A, Koharazawa H et al. Successful treatment of a Jehovah's Witness with acute promyelocytic leukemia. Int J Hematol 1997; 65: 415–416.
Kerridge I, Lowe M, Seldon M et al. Clinical and ethical issues in the treatment of a Jehovah's Witness with acute myeloblastic leukemia. Arch Intern Med 1998; 157: 1753–1757.
Ballen KK, Ford PA, Waitkus H et al. Successful autologous bone marrow transplant without the use of blood products support. Bone Marrow Transplant 2000; 26: 1140–1141.
Mazza P, Palazzo G, Amurri B et al. Acute leukemia in Jehovah's Witness: a challenge for hematologists. Haematologica 2001; 85: 1221–1222.
Russo D, Pricolo G, Micheli MG et al. Fludarabine, arabinosyl cytosine and idarubicin (FLAI) for remission induction in poor-risk acute myeloid leukemia. Leuk Lymphoma 2001; 40: 335–343.
Magrath IT . Management of high-grade lymphomas. Oncology (Hunting) 1998; 12: 40–48.
Multani P, White CA, Grillo-Lopez A . Non-Hodgkin's lymphomas: review of conventional treatments. Curr Pharm Biotechnol 2001; 2: 279–291.
Isaac N, Panzarella T, Mayers C et al. Concurrent cyclophosphamide, methotrexate, 5-fluorouracil chemotherapy and radiotherapy for breast carcinoma: a well tolerated adjuvant regimen. Cancer 2002; 95: 496–703.
Annino L, Vegna ML, Camera A et al. Treatment of adult acute lymphoblastic leukemia (ALL): long-term follow-up of the GIMEMA ALL 0288 randomized study. Blood 2002; 99: 863–871.
Mills W, Chopra R, McMillan A et al. BEAM chemotherapy and autologous bone marrow transplantation for patients with relapsed or refractory non-Hodgkin's lymphoma. J Clin Oncol 1995; 13: 588–595.
Estrin JT, Ford PA, Henry DH et al. Erythropoietin permits high dose chemotherapy with peripheral blood stem-cell transplant for a Jehovah's Witness. Am J Hematol 1997; 55:51–52.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mazza, P., Prudenzano, A., Amurri, B. et al. Myeloablative therapy and bone marrow transplantation in Jehovah's Witnesses with malignancies: single center experience. Bone Marrow Transplant 32, 433–436 (2003). https://doi.org/10.1038/sj.bmt.1704179
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704179
Keywords
This article is cited by
-
Bloodless tandem autologous transplant in Jehovah’s Witness patients
Bone Marrow Transplantation (2018)
-
Autologous and allogeneic stem cell transplant in Jehovah’s Witnesses: a single-center experience on 22 patients
Bone Marrow Transplantation (2016)
-
Ethical issues in autologous stem cell transplantation (ASCT) in advanced breast cancer: A systematic literature review
BMC Medical Ethics (2011)
-
How we approach patient evaluation for hematopoietic stem cell transplantation
Bone Marrow Transplantation (2010)
-
SCT in Jehovah's Witnesses: the bloodless transplant
Bone Marrow Transplantation (2008)