Abstract
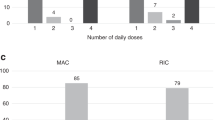
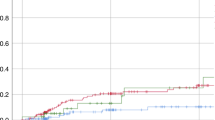
Published data suggest that the average concentration of busulfan at steady state (Bu Css) is critical for successful engraftment in children receiving busulfan as a conditioning agent for bone marrow transplantation (BMT). We previously found in children that a Bu Css <600 ng/ml correlated with autologous recovery/mixed chimerism; there was no correlation between Bu Css and regimen-related toxicity (RRT). In a cohort continuous with the previous trial, we prospectively evaluated targeted busulfan concentrations in 32 pediatric patients (age 0.6–18.5 years) with AML (n = 6), CML (n = 6) and non-malignant disorders (n = 20) receiving HLA-closely matched donor grafts. In this trial, individual busulfan pharmacokinetics were performed prior to admission. Busulfan doses were then adjusted to achieve a Bu Css target range of 600–900 ng/ml ± 10% depending on donor source and disease. A repeat study was done following dose 1 of the conditioning regimen. Thirty of thirty-two (94%) patients achieved target concentrations. Total busulfan doses ranged from 10.9 to 29 mg/kg. Thirty of thirty-two patients (94%) have durably engrafted. Grade 3/4 RRT occurred in seven patients (21%). Targeting Bu Css ranges of 600–900 ng/ml significantly improved our rate of successful engraftment from 74% to 94% (P = 0.043). These results indicate that targeted busulfan dosing optimizes allogeneic engraftment in children.
Bone Marrow Transplantation (2001) 28, 1013–1018.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Growchow LB, Krivit W, Whitley CB, Blazer BR . Busulfan disposition in children Blood 1990 75: 1723–1727
Hassan M, Oberg G, Beckassy AN et al. Pharmacokinetics of high dose busulphan in relation and age and chronopharmacology Cancer Chemother Pharmacol 1991 28: 130–134
Vassal G, Gouyette A, Hartmann O et al. Pharmacokinetics of high dose busulfan in children Cancer Chemother Pharmacol 1989 24: 386–390
Vassal G, Deroussent A, Hartmann O et al. Dose-dependent neurotoxicity of high-dose busulfan in children: a clinical and pharmacological study Cancer Res 1990 50: 6203–6207
Slattery JT, Sanders JE, Buckner CD et al. Graft-rejection and toxicity following bone marrow transplantation in relation to busulfan pharmacokinetics Bone Marrow Transplant 1995 16: 31–42
Slattery JT, Clift RA, Buckner CD et al. Marrow transplantation for chronic myeloid leukemia: the influence of plasma busulfan levels on the outcome of transplantation Blood 1997 89: 3055–3060
Bolinger AM, Zangwill AB, Slattery JT et al. An evaluation of engraftment, toxicity and busulfan toxicity in children receiving bone marrow transplantation for leukemia or genetic disease Bone Marrow Transplantation 2000 25: 925–930
Slattery JT, Risler LJ . Therapeutic monitoring of busulfan in hematopoietic stem cell transplantation Ther Drug Monitor 1998 20: 543–549
Bearman SI, Applelbaum FR, Buckner CD et al. Regimen-related toxicity in patients undergoing bone marrow transplantation J Clin Oncol 1988 6: 562–568
McCune JS, Gibbs JP, Slattery JT . Plasma concentration monitoring of busulfan: does it improve clinical outcome? Clin Pharmacokinetics 2000 39: 155–165
Regazzi MB, Locatelli F, Buggia I et al. Disposition of high-dose busulfan in pediatric patients undergoing bone marrow transplantation Clin Pharmacol Ther 1993 54: 45–52
Lee JL, Gooley T, Bensinger W et al. Veno-occlusive disease of the liver after busulfan, melphalan and thiotepa conditioning therapy: incidence, risk factors and outcome Biol Blood Marrow Transplant 1999 5: 306–315
Demirer T, Buckner CD, Applebaum FR et al. Busulfan, cyclophosphamide and fractionated total body irradiation for autologous or syngeneic marrow transplantation for acute and chronic myelogenous leukemia: phase I dose escalation of busulfan based on targeted plasma levels Bone Marrow Transplant 1996 17: 491–495
Chattergoon DS, Saunders EF, Klein J et al. Improved limited sampling method for individualized busulfan dosing in bone marrow transplantation in children Bone Marrow Transplant 1997 20: 347–354
Growchow LB . Busulfan disposition: the role of therapeutic monitoring in bone marrow transplantation induction regimens Semin Oncol 1993 20 (Suppl. 4): 18–25
Dix SP, Wingard JR, Mullins RE et al. Association of busulfan area under the curve with veno-occlusive disease following BMT Bone Marrow Transplant 1996 17: 225–230
Acknowledgements
This work was supported in part by NIH grant M01-RR01271, Pediatric Clinical Research Center; NIH grant CA18029.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bolinger, A., Zangwill, A., Slattery, J. et al. Target dose adjustment of busulfan in pediatric patients undergoing bone marrow transplantation. Bone Marrow Transplant 28, 1013–1018 (2001). https://doi.org/10.1038/sj.bmt.1703264
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703264
Keywords
This article is cited by
-
Clinical outcomes of individualized busulfan-dosing in hematopoietic stem cell transplantation in Chinese children undergoing with therapeutic drug monitoring
Bone Marrow Transplantation (2022)
-
Evaluation of two software using Bayesian methods for monitoring exposure and dosing once-daily intravenous busulfan in paediatric patients receiving haematopoietic stem cell transplantation
Cancer Chemotherapy and Pharmacology (2021)
-
Busulfan systemic exposure and its relationship with efficacy and safety in hematopoietic stem cell transplantation in children: a meta-analysis
BMC Pediatrics (2020)
-
Population pharmacokinetics of busulfan in Saudi pediatric patients undergoing hematopoietic stem cell transplantation
International Journal of Clinical Pharmacy (2020)
-
Use of busulfan in conditioning for allogeneic hematopoietic stem cell transplantation in adults: a survey by the Transplant Complications Working Party of the EBMT
Bone Marrow Transplantation (2019)