Abstract
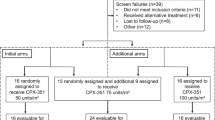
this prospective trial evaluated the efficacy and toxicity of granisetron for antiemetic control in patients receiving high-dose cyclophosphamide (cy)-containing regimens with/without tbi for bone marrow (bm) or peripheral blood stem cell (pbsc) transplantation or pbsc mobilization. granisetron 1 mg i.v. plus dexamethasone 10 mg i.v. were administered daily 30 min before chemotherapy or radiation for a median of 5 days. response was defined as the number of emetic episodes per 24 h: complete response, 0 and no emetic rescue; major response, 1–2; minor response, 3–5; failure, >5. One hundred patients were enrolled. Ninety-eight received CY-containing regimens and 26 of these additionally received TBI (12 Gy divided over 4 days). Response was complete on 216 (47%) of a total 456 patient days, major on 222 (49%), minor on 14 (3%), and failure on 4 (1%). Mean number of emetic episodes per patient per day and breakthrough medication required per patient per day was 0.24 (range 0–8) and 0.40 (range 0–8), respectively. Adverse effects were minimal, with headache (20%) reported most frequently. Based on these results, granisetron plus dexamethasone is an effective and well-tolerated antiemetic regimen in BMT/PBSCT recipients conditioned with high-dose chemotherapy with/without TBI. Bone Marrow Transplantation (2000) 25, 1279–1283.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Coates A, Abraham S, Haye SB et al. On the receiving end – patient perception of the side effects of cancer chemotherapy Eur J Cancer Clin Oncol 1983 19: 203–208
Clavel M, Soukop M, Greenstreet YL . Improved control of emesis and quality of life with ondansetron in breast cancer Oncology 1993 50: 180–185
Perez EA, Gandara DR . Advances in the control of chemotherapy-induced emesis Ann Oncol 1992 3: S47-S50
Mitchell EP, Schein PS . Gastrointestinal toxicity of chemotherapy agents. In: Perry MC (ed) The Chemotherapy Sourcebook Williams & Wilkins: Baltimore 1992 pp–620–634
Dodds LJ . The control of cancer chemotherapy-induced nausea and vomiting J Clin Hosp Pharm 1995 10: 143–166
Ettinger CS . Preventing chemotherapy-induced nausea and vomiting: an update and a review of emesis Semin Oncol 1995 22: (Suppl.10) 6–18
Plosker GL, Goa KL . Granisetron: a review of its pharmacological properties and therapeutic use as an antiemetic Drugs 1991 42: 805–824
Okamoto S, Takahashi S, Tanosaki R et al. Granisetron in the prevention of vomiting induced by conditioning for stem cell transplantation: a prospective randomized study Bone Marrow Transplant 1996 17: 679–683
Hunter AE, Prentice HG, Pothcary K et al. Granisetron, a selective 5-HT3 receptor antagonist, for the prevention of radiation induced emesis during total body irradiation Bone Marrow Transplant 1991 7: 439–441
Prentice HG, Cunningham S, Ghandhi L et al. Granisetron in the prevention of irradiation-induced emesis Bone Marrow Transplant 1995 15: 445–487
Roberts JT, Priestman TJ . A review of ondansetron in the management of radiotherapy-induced emesis Oncology 1993 50: 173–179
Kamamabrou D . Intravenous granisetron-establishing the optimal dose Eur J Cancer 1992 28A: (Suppl. 1) S6–S11
Heron JF, Goedhals L, Jordaan JP et al. Oral granisetron alone and in combination with dexamethasone: a double-blind randomized comparison against high-dose metoclopramide plus dexamethasone in prevention of cisplatin-induced emesis Ann Oncol 1994 5: 579–584
Abbott B, Ippoliti C, Bruton J et al. Antiemetic efficacy of granisetron plus dexamethasone in bone marrow transplant patients receiving chemotherapy and total body irradiation Bone Marrow Transplant 1999 23: 265–269
Hewitt M, Cornish J, Pamphilon D et al. Effective emetic control during conditioning of children for bone marrow transplantation using ondansetron, a 5-HT3 antagonist Bone Marrow Transplant 1991 7: 431–433
Barbounis V, Koumakis G, Vassilomanolakis M et al. A phase II study of ondansetron as antiemetic prophylaxis in patients receiving high-dose polychemotherapy and stem cell transplantation Support Care Cancer 1995 3: 301–306
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abbott, B., Ippoliti, C., Hecth, D. et al. Granisetron (Kytril) plus dexamethasone for antiemetic control in bone marrow transplant patients receiving highly emetogenic chemotherapy with or without total body irradiation. Bone Marrow Transplant 25, 1279–1283 (2000). https://doi.org/10.1038/sj.bmt.1702424
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1702424
Keywords
This article is cited by
-
A randomized, placebo-controlled pilot trial of aprepitant combined with standard antiemetic therapy for the prevention of chemotherapy-induced nausea and vomiting in patients undergoing cyclophosphamide-based conditioning regimens prior to hematopoietic stem cell transplant (HSCT)
Bone Marrow Transplantation (2018)
-
Palonosetron versus first-generation 5-hydroxytryptamine type 3 receptor antagonists for emesis prophylaxis in patients undergoing allogeneic hematopoietic stem cell transplantation
Annals of Hematology (2014)
-
Granisetron in the control of radiotherapy-induced nausea and vomiting: a comparison with other antiemetic therapies
Supportive Care in Cancer (2005)
-
Granisetron vs ondansetron for prevention of nausea and vomiting in hematopoietic stem cell transplant patients: results of a prospective, double-blind, randomized trial
Bone Marrow Transplantation (2004)
-
Granisetron Plus Dexamethasone Versus Granisetron Alone in the Prevention of Vomiting Induced by Conditioning for Stem Cell Transplantation: A prospective Randomized Study
International Journal of Hematology (2003)