Abstract
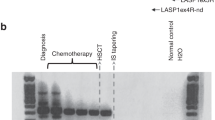
Peripheral blood stem cell support allows dose intensification of multiple cycle chemotherapy for metastatic tumors, including pediatric sarcomas. The VACIME protocol (vincristine, adriamycin, cyclophosphamide, ifosfamide, mesna and etoposide) utilizes peripheral blood stem cells (PBSC) collected following the treatment cycle as support for subsequent dose- and time-intensive chemotherapy. A critical assumption is that PBSC collected in this manner will be purged of residual tumor cells in vivo. We tested this assumption using sensitive reverse-transcriptase polymerase chain reaction (RT-PCR) to assess the presence of the characteristic translocations of the Ewing’s sarcoma family of tumors (ESFT) and alveolar rhabdomyosarcoma (ARMS), t(11;22), and t(2;13), respectively. We used RT-PCR to evaluate 122 samples of peripheral blood (PB), bone marrow (BM) and PBSC collected from 12 pediatric patients with metastatic ESFT and ARMS. The samples included pre-therapy BM and PB, as well as BM, PB, and PBSC collections at various times in the VACIME treatment course. Molecular evidence of tumor contamination was detected in 1/40 PBSC collections from 12 patients. In all patients, we documented clearance of disease by RT-PCR in peripheral blood and bone marrow by week 9 of the VACIME protocol. In vivo purging in combination with the intensive VACIME regime appears to be effective in removing tumor cells from PBSC, bone marrow, and peripheral blood as detected by RT-PCR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Thomson, B., Hawkins, D., Felgenhauer, J. et al. RT-PCR evaluation of peripheral blood, bone marrow and peripheral blood stem cells in children and adolescents undergoing VACIME chemotherapy for Ewing’s sarcoma and alveolar rhabdomyosarcoma. Bone Marrow Transplant 24, 527–533 (1999). https://doi.org/10.1038/sj.bmt.1701939
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1701939
Keywords
This article is cited by
-
Release of circulating tumor cells and cell-free nucleic acids is an infrequent event in synovial sarcoma: liquid biopsy analysis of 15 patients diagnosed with synovial sarcoma
Diagnostic Pathology (2018)
-
Circulating tumor cells in sarcomas: a brief review
Medical Oncology (2015)
-
Detection and clinical significance of bone marrow involvement in patients with rhabdomyosarcoma
Virchows Archiv (2010)
-
Consensus criteria for sensitive detection of minimal neuroblastoma cells in bone marrow, blood and stem cell preparations by immunocytology and QRT-PCR: recommendations by the International Neuroblastoma Risk Group Task Force
British Journal of Cancer (2009)
-
Ewing Sarcoma tumor cells express CD34: implications for autologous stem cell transplantation
Bone Marrow Transplantation (2007)