Abstract
Background and Objectives:
Among otherwise healthy adults, there is a subgroup of individuals who develop symptoms of hypoglycemia during episodes of food restriction. The aim of the present study was to investigate whether such individuals develop hypoglycemia or react abnormally in other metabolic aspects during a 24-hour fast.
Subjects and Methods:
Ninety medical students were asked if they wanted to participate. Sixteen were selected; none dropped out. A 24-hour fast was performed at a hospital ward. Blood samples and questionnaires were taken at eight specific times.
Result:
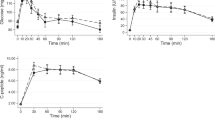
During the fast, the sensitive group reported significantly higher scores on ‘irritation’ and ‘shakiness’. However, no hypoglycemia occurred and the lowest detected blood glucose concentration was 3.7 mmol/l. There were no differences between the groups in plasma glucose, cortisol, growth hormone (GH), insulin, β-hydroxybutyrate (β-OH) and lactate levels. The blood pressures and heart rates were also similar.
Conclusions:
Adults, despite subjective signs of hypoglycemia, can fast without any metabolic or endocrine derangement.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Brun JF, Fedou C, Mercier J (2000). Postprandial reactive hypoglycemia. Diabetes Metab 26, 337–351.
Champe PC, Harvey RA (2005). Lippincott's Illustrated Reviews: Biochemistry. Lippincott, Williams & Wilkins: Philadelphia.
Chapman IM, Goble EA, Wittert GA, Morley JE, Horowitz M (1998). Effect of intravenous glucose and euglycemic insulin infusions on short-term appetite and food intake. Am J Physiol 274, R596–R603.
Dahlquist G, Gentz J, Hagenfeldt L, Larsson A, Low H, Persson B et al. (1979). Ketotic hypoglycemia of childhood – a clinical trial of several unifying etiological hypotheses. Acta Paediatr Scand 68, 649–656.
Elimam A, Horal M, Bergstrom M, Marcus C (1997). Diagnosis of hypoglycaemia: effects of blood sample handling and evaluation of a glucose photometer in the low glucose range. Acta Paediatr 86, 474–478.
Gielkens HA, Verkijk M, Lam WF, Lamers CB, Masclee AA (1998). Effects of hyperglycemia and hyperinsulinemia on satiety in humans. Metabolism 47, 321–324.
Grunt JA, McGarry ME, McCollum AT, Gould JB (1970). Studies of children with ketotic hypoglycemia. Yale J Biol Med 42, 420–438.
Hagstrom-Toft E, Bolinder J, Ungerstedt U, Arner P (1997). A circadian rhythm in lipid mobilization which is altered in IDDM. Diabetologia 40, 1070–1078.
Haymond MW, Howard C, Ben-Galim E, DeVivo DC (1983). Effects of ketosis on glucose flux in children and adults. Am J Physiol 245, E373–E378.
Horal M, Ungerstedt U, Persson B, Westgren M, Marcus C (1995). Metabolic adaptation in IUGR neonates determined with microdialysis – a pilot study. Early Hum Dev 42, 1–14.
Inoue K, Kakehashi Y, Oomori S, Koizumi A (2004). Biochemical hypoglycemia in female nurses during clinical shift work. Res Nurs Health 27, 87–96.
Kamel A, Norgren S, Persson B, Marcus C (1999). Insulin induced hypoglycaemia: comparison of glucose and glycerol concentrations in plasma and microdialysate from subcutaneous adipose tissue. Arch Dis Child 80, 42–45.
Karam JH (2001). Hypoglycemic disorders. In: Greenspan FS and Strewler GJ (eds). Basic & Clinical Endocrinology. Appleton & Lange: Stamford, CT, USA, pp 823.
Levin F, Edholm T, Schmidt PT, Gryback P, Jacobsson H, Degerblad M et al. (2006). Ghrelin stimulates gastric emptying and hunger in normal-weight humans. J Clin Endocrinol Metab 91, 3296–3302.
Marcus C (2001). How to measure and interpret glucose in neonates. Acta Paediatr 90, 963–964.
Marks V (1987). Glycaemic stability in healthy subjects: fluctuations in blood glucose Concentration During the Day. In: Andreani D, Marks V and Lefebvre PJ (eds). Hypoglycemia. Raven Press: NY, USA, pp 19–24.
Meyer EL, Waldenlind E, Marcus C (2003). Diminished nocturnal lipolysis in cluster headache: a sign of central sympathetic dysregulation? Neurology 61, 1250–1254.
Sasaki M, Mogi T, Wada Y, Hirosawa I, Koizumi A (1996). An endemic condition of biochemical hypoglycemia among male volunteers. Ind Health 34, 323–333.
Wiesli P, Brandle M, Zapf J, Seiler H, Zwimpfer C, Spinas GA et al. (2004). Assessment of hyperinsulinaemia at the termination of the prolonged fast. Clin Chim Acta 342, 227–231.
Wiesli P, Schwegler B, Schmid B, Spinas GA, Schmid C (2005). Mini-mental state examination is superior to plasma glucose concentrations in monitoring patients with suspected hypoglycaemic disorders during the 72-hour fast. Eur J Endocrinol 152, 605–610.
Wildenhoff KE (1970). A micro-method for the enzymatic determination of acetoacetate and 3-hydroxybutyrate in blood and urine. Scand J Clin Lab Invest 25, 171–179.
Williams G, Cai XJ, Elliott JC, Harrold JA (2004). Anabolic neuropeptides. Physiol Behav 81, 211–222.
Yager J, Young RT (1974). Non-hypoglycemia is an epidemic condition. N Engl J Med 291, 907–908.
Acknowledgements
This work was supported by the Freemasons in Stockholm Foundation for Children's Welfare and the Swedish Research Council #K2002-72X-009941-11B.
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributors: CM has contributed to the initiation of the study, designed the study protocol, participated in the writing of the manuscript and has been the supervisor of the study. EP has contributed to the study design and participated in the data collection. JA has contributed in all parts of the study
Rights and permissions
About this article
Cite this article
Alkén, J., Petriczko, E. & Marcus, C. Effect of fasting on young adults who have symptoms of hypoglycemia in the absence of frequent meals. Eur J Clin Nutr 62, 721–726 (2008). https://doi.org/10.1038/sj.ejcn.1602785
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejcn.1602785
Keywords
This article is cited by
-
Fatty acids homeostasis during fasting predicts protection from chemotherapy toxicity
Nature Communications (2022)