Abstract
Objective:
To examine the effects of single and long-term administration of wheat albumin (WA) on blood glucose levels and blood glucose control, respectively.
Design:
Randomly arranged crossover trial for single administration in healthy subjects and double-blinded randomized controlled trial for long-term administration (3 months) in diabetic patients. In vitro α-amylase inhibitory activity of WA was also determined.
Setting:
Central Research Laboratories of Nisshin Flour Milling Co. Ltd. (Saitama, Japan) for single administration and Aiwa Clinic (Saitama, Japan) for long-term administration.
Subjects:
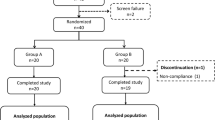
A total of 12 healthy adult male volunteers for the single administration and 24 type II outpatient diabetics with mild hyperglycemia for the long-term administration.
Interventions:
Subjects took soups containing 0, 0.25, 0.5, and 1.0 g WA before test meals for single administration, and patients took soups with or without 0.5 g WA before every meal for the long-term (3 months) administration.
Results:
In vitro α-amylase inhibitory activity of WA was 150 times that of wheat flour. In the single administration experiment, WA suppressed peak postprandial blood glucose levels in a dose-dependent manner: 31, 47, and 50% reduction after 0.25, 0.5, and 1.0 g administrations, respectively. In the long-term administration, 0.5 g of WA did not affect fasting blood glucose levels, whereas it reduced hemoglobin A1c levels. No significant adverse effects such as hypoglycemia or gastrointestinal disturbances were observed in the two experiments.
Conclusion:
In the treatment of type II diabetic patients, WA might be a useful functional food, which, with diet and exercise, could help to improve blood glucose control without any critical adverse effects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Amano M, Ogawa H, Kojima K, Kamidaira T, Suetsugu S, Yoshihama M, Satoh T, Samejima T & Matsumoto I (1998): Identification of the major allergens in wheat flour responsible for baker's asthma. Biochem. J. 330, 1229–1234.
Baba S (1994): Alpha-glucosidase inhibitor. In Novel Development in Pharmacological Therapy of Diabetes. Baba S pp 53–54. Japan Churchill Livingstone. (in Japanese).
Bunn HF, Haney DN, Kamin S, Gabbay KH & Gallop PM (1976): The biosynthesis of human hemoglobin A1c. J. Clin. Invest. 57, 1652–1659.
Carrascosa M, Pascual F & Aresti S (1997): Acarbose-induced acute severe hepatotixicity. Lancet 349, 698–699.
Chiasson JL, Josse RG, Gomis R, Hanefeld M, Karasik A, Laakso M & STOP-NIDDM Trial Research Group (2002): Acarbose for prevention of type 2 diabetes mellitus: the STOP-NIDDM randomized trial. Lancet 359, 2072–2077.
Chiasson JL, Josse RG, Gomis R, Hanefeld M, Karasik A, Laakso M & STOP-NIDDM Trial Research Group (2003): Acarbose treatment and the risk of cardiovascular disease and hypertension in patients with impaired glucose tolerance: the STOP-NIDDM trial. J. Am. Med. Assoc. 290, 486–494.
Choudhury A, Maeda K, Murayama R & DiMagno EP (1996): Character of a wheat amylase inhibitor preparation and effects on fasting human pancreaticobilary secretions and hormones. Gastroenterology 111, 1313–1320.
DePonte R, Parlamenti R, Petrucci T, Silano V & Tomasi M (1976): Albumin α-amylase inhibitor families from wheat flour. Cereal Chem. 53, 805–820.
The Diabetes Control and Complications Trial Research Group (1993): The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. New Engl. J. Med. 329, 977–986.
Diaz-Gutierrez FL, Ladero JM & Diaz-Rubio M (1998): Acarbose-induced acute hepatitis. Am. J. Gastroenterol. 93, 481.
Frier BM (2002): Hypoglycemic valleys: an under-recognised problem in type 2 diabetes? Int. J. Clin. Pract. Suppl. 129, 12–19.
Fujimoto Y, Ohhira M, Miyokawa N, Kitamori S & Kohgo T (1998): Acarbose-induced hepatic injury. Lancet 351, 340.
Goto Y, Baba S, Nakagawa S, Kuzuya T, Hirata Y, Akunuma Y, Sakamoto N, Shigeta Y & Mimura G (1992): The availability for non-insulin dependent diabetes mellitus of α-glucosidase inhibitor AO-128. Phase III comparison test between double blind groups. Igaku No Ayumi 160, 943–971. (in Japanese).
Japanese Ministry of Health and Public Welfare (1999): National survey of diabetes mellitus. 1997 (in Japanese).
Japanese Ministry of Health, Labor, and Public Welfare (2003): Brief report of the national survey of diabetes mellitus. 2002 (in Japanese).
Johnston PS, Lebovitz HE, Coniff RF, Simonson DC, Raskin P & Munera CL (1998): Advantage of α-glucosidase inhibition as monotherapy in elderly type 2 diabetic patients. J. Clin. Endocrinol. Metab. 83, 1515–1522.
Josse RG, Chiasson JL, Ryan EA, Lau DCW, Ross SA, Yale JF, Leiter LA, Maheux P, Tessier D, Wolever TMS, Gerstein H, Rodger NW, Dornan JM, Murphy LJ, Rabasa-Lhoret R & Meneilly GS (2003): Acarbose in the treatment of elderly patients with type 2 diabetes. Diabetes Res. Clin. Pract. 59, 37–42.
Kashlan N & Richardson M (1981): The complete amino acid sequence of a major wheat protein inhibitor of α-amylase. Phytochemistry. 20, 1781–1784.
Kawakami S, Arima T, Harada K, Miyazono H, Oketani M, Miyazaki H & Arima T (2001): Hepatic necrosis with cholestasis induced by long-term voglibose administration. Intern. Med. 40, 484–488.
Kneen E & Sandstedt RM (1943): An amylase inhibitor from certain cereals. J. Am. Chem. Soc. 65, 1247.
Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM & Diabetes Prevention Program Research Group (2002): Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. New Engl. J. Med. 346, 393–403.
Koenig RJ, Peterson CM, Jones RL, Saudek C, Lehrman M & Cerami A (1976): Correlation of glucose regulation and hemoglobin A1c in diabetes mellitus. New Engl. J. Med. 295, 417–420.
Koike D, Yamadera K & DiMagno EP (1995): Effect of a wheat amylase inhibitor on canine carbohydrate digestion, gastrointestinal function, and pancreatic growth. Gastroenterology 108, 1221–1229.
Maeda K, Hase T & Matsubara H (1983): Complete amino acid sequence of an alpha-amylase inhibitor in wheat kernel. Biochim. Biophys. Acta 743, 52–57.
Oda Y, Matsunaga T, Fukuyama K, Miyazaki T & Morimoto T (1997): Tertiary and quaternary structures of 0.19 α-amylase inhibitor from wheat kernel determined by X-ray analysis at 2.06-A resolution. Biochemistry 36, 13503–13511.
Ohkubo Y, Kishikawa H, Araki E, Miyata T, Isami S, Motoyoshi S, Kojima Y, Furuyoshi N & Shichiri M (1995): Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non-insulin-dependent diabetes mellitus: a randomized prospective 6-year study. Diabetes Res. Clin. Pract. 28, 103–117.
Ohno T (1995): Investigation of gastrointestinal adverse effects by alpha-glucosidase inhibitor, especially the development of ileus. J. New Remedies Clin. 44, 1114–1116. (in Japanese).
Rosenstock J (2000): Treatment strategies and new therapeutic advances for type 2 diabetes. Diabet. Educ. 26 (Suppl), 14–18.
Saloranta C, Guitard C, Pecher E, De Pablos-Velasco P, Lahti K, Brunel P & Groop L (2002): Nateglinide improves early insulin secretion and controls postprandial glucose excursions in a prediabetic population. Diabetes Care 25, 2141–2146.
Scherbaum WA & Zimmet P (1995): The prevention of the NIDDM epidemic. IDF Bull. 40, 6–16.
Sodini G, Silano V, de Agazio M, Pocchiari F, Tentori L & Vivaldi G (1970): Purification and properties of a Triticum aestivum specific albumin. Phytochemistry 9, 1167–1172.
Stratton IM, Kohner EM, Aldington SJ, Turner RC, Holman RR, Manley SE & Matthews DR (2001): UKPDS 50: risk factors for incidence and progression of retinopathy in type II diabetes over 6 years from diagnosis. Diabetologia 44, 156–163.
UK Prospective Diabetes Study (UKPDS) Group (1998): Intensive blood-glucose control with sulpfonylureas or insulin compaired with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 352, 837–853.
Yanai H (2004): Statcel—The Usefull Add-in Software Forms on Excel 2nd Edition, Tokyo, Japan. OMS (in Japanese).
Author information
Authors and Affiliations
Corresponding author
Additional information
Guarantor: S Inoue.
Contributors: This study was designed by TK and SI. SI, YN, JS and YT contributed to the subject recruiting and clinical data collection. TK, TM and IK carried out in vitro experiments and sample preparation. YS contributed to the manuscript preparation. SI was the main person responsible for all stages of the study.
Rights and permissions
About this article
Cite this article
Kodama, T., Miyazaki, T., Kitamura, I. et al. Effects of single and long-term administration of wheat albumin on blood glucose control: randomized controlled clinical trials. Eur J Clin Nutr 59, 384–392 (2005). https://doi.org/10.1038/sj.ejcn.1602085
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ejcn.1602085
Keywords
This article is cited by
-
Phenolic bioactive-linked antioxidant, anti-hyperglycemic, and antihypertensive properties of sweet potato cultivars with different flesh color
Horticulture, Environment, and Biotechnology (2023)
-
Plant-Derived Bioactive Peptides: A Treatment to Cure Diabetes
International Journal of Peptide Research and Therapeutics (2020)