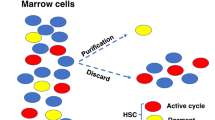
Abstract
Colony-stimulating factors and other cytokines signal via their cognate receptors to regulate hematopoiesis. In many developmental systems, inductive signalling determines cell fate and, by analogy with this, it has been postulated that cytokines, signalling via their cognate receptors, may play an instructive role in lineage specification in hematopoiesis. An alternative to this instructive hypothesis is the stochastic or permissive hypothesis. The latter proposes that commitment to a particular hematopoietic lineage is an event that occurs independently of extrinsic signals. It predicts that the role of cytokines is to provide nonspecific survival and proliferation signals. In this review, we look at the role of cytokine receptor signalling in hematopoiesis and consider the evidence for both hypotheses. Data from experiments that genetically manipulate receptor gene expression in vitro or in vivo are reviewed. Experiments in which cytokine receptors were installed in multipotential cells showed that, in some cases, stimulation with the cognate ligand could lead to alterations in lineage output. The creation of genetically manipulated mouse strains demonstrated that cytokine receptors are required for expansion and survival of single lineages but did not reveal a role in lineage commitment. We conclude that hematopoietic differentiation involves mainly stochastic events, but that cytokine receptors also have some instructive role.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Akashi K, Kondo M, von Freeden-Jeffry U, Murray R, Weissman IL . (1997). Bcl-2 rescues T lymphopoiesis in interleukin-7 receptor-deficient mice. Cell 89: 1033–1041.
Alexander WS, Roberts AW, Nicola NA, Li R, Metcalf D . (1996). Deficiencies in progenitor cells of multiple hematopoietic lineages and defective megakaryocytopoiesis in mice lacking the thrombopoietic receptor c-Mpl. Blood 87: 2162–2170.
Barner M, Mohrs M, Brombacher F, Kopf M . (1998). Differences between IL-4R alpha-deficient and IL-4-deficient mice reveal a role for IL-13 in the regulation of Th2 responses. Curr Biol 8: 669–672.
Behrmann I, Smyczek T, Heinrich PC, Schmitz-Van de Leur H, Komyod W, Giese B et al. (2004). Janus kinase (Jak) subcellular localization revisited: the exclusive membrane localization of endogenous Janus kinase 1 by cytokine receptor interaction uncovers the Jak receptor complex to be equivalent to a receptor tyrosine kinase. J Biol Chem 279: 35486–35493.
Betz UA, Bloch W, van den Broek M, Yoshida K, Taga T, Kishimoto T et al. (1998). Postnatally induced inactivation of gp130 in mice results in neurological, cardiac, hematopoietic, immunological, hepatic, and pulmonary defects. J Exp Med 188: 1955–1965.
Cantor AB, Orkin SH . (2001). Hematopoietic development: a balancing act. Curr Opin Genet Dev 11: 513–519.
Carver-Moore K, Broxmeyer HE, Luoh SM, Cooper S, Peng J, Burstein SA et al. (1996). Low levels of erythroid and myeloid progenitors in thrombopoietin-and c-mpl-deficient mice. Blood 88: 803–808.
Constantinescu SN, Keren T, Socolovsky M, Nam H, Henis YI, Lodish HF . (2001). Ligand-independent oligomerization of cell-surface erythropoietin receptor is mediated by the transmembrane domain. Proc Natl Acad Sci USA 98: 4379–4384.
D’Andrea AD . (1994). Hematopoietic growth factors and the regulation of differentiative decisions. Curr Opin Cell Biol 6: 804–808.
DeChiara TM, Vejsada R, Poueymirou WT, Acheson A, Suri C, Conover JC et al. (1995). Mice lacking the CNTF receptor, unlike mice lacking CNTF, exhibit profound motor neuron deficits at birth. Cell 83: 313–322.
DiSanto JP, Muller W, Guy-Grand D, Fischer A, Rajewsky K . (1995). Lymphoid development in mice with a targeted deletion of the interleukin 2 receptor gamma chain. Proc Natl Acad Sci USA 92: 377–381.
Evans CA, Pierce A, Winter SA, Spooncer E, Heyworth CM, Whetton AD . (1999). Activation of granulocyte–macrophage colony-stimulating factor and interleukin-3 receptor subunits in a multipotential hematopoietic progenitor cell line leads to differential effects on development. Blood 94: 1504–1514.
Fairbairn LJ, Cowling GJ, Reipert BM, Dexter TM . (1993). Suppression of apoptosis allows differentiation and development of a multipotent hemopoietic cell line in the absence of added growth factors. Cell 74: 823–832.
Fox N, Priestley G, Papayannopoulou T, Kaushansky K . (2002). Thrombopoietin expands hematopoietic stem cells after transplantation. J Clin Invest 110: 389–394.
Frohlich A, Marsland BJ, Sonderegger I, Kurrer M, Hodge MR, Harris NL et al. (2007). IL-21 receptor signaling is integral to the development of Th2 effector responses in vivo. Blood 109: 2023–2031.
Fukunaga R, Ishizaka-Ikeda E, Nagata S . (1993). Growth and differentiation signals mediated by different regions in the cytoplasmic domain of granulocyte colony-stimulating factor receptor. Cell 74: 1079–1087.
Gainsford T, Nandurkar HH, Metcalf D, Robb L, Begley CG, Alexander WS . (2000). The residual megakaryocyte and platelet production in c-Mpl-deficient mice is not dependent on the actions of interleukin-6, interleukin -11 or leukemia inhibitory factor. Blood 95: 528–534.
Goldsmith MA, Mikami A, You Y, Liu KD, Thomas L, Pharr P et al. (1998). Absence of cytokine receptor-dependent specificity in red blood cell differentiation in vivo. Proc Natl Acad Sci USA 95: 7006–7011.
Gurney AL, Carver-Moore K, de Sauvage FJ, Moore MW . (1994). Thrombocytopenia in c-mpl-deficient mice. Science 265: 1445–1447.
Hayashida K, Kitamura T, Gorman DM, Arai K, Yokota T, Miyajima A . (1990). Molecular cloning of a second subunit of the receptor for human granulocyte–macrophage colony-stimulating factor (GM-CSF): reconstitution of a high-affinity GM-CSF receptor. Proc Natl Acad Sci USA 87: 9655–9659.
Heim MH . (1999). The Jak–STAT pathway: cytokine signalling from the receptor to the nucleus. J Recept Signal Transduct Res 19: 75–120.
Hisakawa H, Sugiyama D, Nishijima I, Xu MJ, Wu H, Nakao K et al. (2001). Human granulocyte–macrophage colony-stimulating factor (hGM-CSF) stimulates primitive and definitive erythropoiesis in mouse embryos expressing hGM-CSF receptors but not erythropoietin receptors. Blood 98: 3618–3625.
Horan T, Wen J, Narhi L, Parker V, Garcia A, Arakawa T et al. (1996). Dimerization of the extracellular domain of granuloycte-colony stimulating factor receptor by ligand binding: a monovalent ligand induces 2:2 complexes. Biochemistry 35: 4886–4896.
Ichihara M, Hara T, Takagi M, Cho LC, Gorman DM, Miyajima A . (1995). Impaired interleukin-3 (IL-3) response of the A/J mouse is caused by a branch point deletion in the IL-3 receptor alpha subunit gene. EMBO J 14: 939–950.
Iwasaki-Arai J, Iwasaki H, Miyamoto T, Watanabe S, Akashi K . (2003). Enforced granulocyte/macrophage colony-stimulating factor signals do not support lymphopoiesis, but instruct lymphoid to myelomonocytic lineage conversion. J Exp Med 197: 1311–1322.
Jegalian AG, Acurio A, Dranoff G, Wu H . (2002). Erythropoietin receptor haploinsufficiency and in vivo interplay with granulocyte–macrophage colony-stimulating factor and interleukin 3. Blood 99: 2603–2605.
Just U, Stocking C, Spooncer E, Dexter TM, Ostertag W . (1991). Expression of the GM-CSF gene after retroviral transfer in hematopoietic stem cell lines induces synchronous granulocyte–macrophage differentiation. Cell 64: 1163–1173.
Kaushansky K . (2006). Lineage-specific hematopoietic growth factors. N Engl J Med 354: 2034–2045.
Kaushansky K, Fox N, Lin NL, Liles WC . (2002). Lineage-specific growth factors can compensate for stem and progenitor cell deficiencies at the postprogenitor cell level: an analysis of doubly TPO- and G-CSF receptor-deficient mice. Blood 99: 3573–3578.
Kertesz N, Wu J, Chen TH, Sucov HM, Wu H . (2004). The role of erythropoietin in regulating angiogenesis. Dev Biol 276: 101–110.
Kieran MW, Perkins AC, Orkin SH, Zon LI . (1996). Thrombopoietin rescues in vitro erythroid colony formation from mouse embryos lacking the erythropoietin receptor. Proc Natl Acad Sci USA 93: 9126–9131.
Kirby SL, Cook DN, Walton W, Smithies O . (1996). Proliferation of multipotent hematopoietic cells controlled by a truncated erythropoietin receptor transgene. Proc Natl Acad Sci USA 93: 9402–9407.
Kondo M, Scherer DC, Miyamoto T, King AG, Akashi K, Sugamura K et al. (2000). Cell-fate conversion of lymphoid-committed progenitors by instructive actions of cytokines. Nature 407: 383–386.
Lagasse E, Weissman IL . (1997). Enforced expression of Bcl-2 in monocytes rescues macrophages and partially reverses osteopetrosis in op/op mice. Cell 89: 1021–1031.
Li M, Sendtner M, Smith A . (1995). Essential function of LIF receptor in motor neurons. Nature 378: 724–727.
Lieschke GJ, Grail D, Hodgson G, Metcalf D, Stanley E, Cheers C et al. (1994). Mice lacking granulocyte colony-stimulating factor have chronic neutropenia, granulocyte and macrophage progenitor cell deficiency, and impaired neutrophil mobilization. Blood 84: 1737–1746.
Lin CS, Lim SK, D’Agati V, Costantini F . (1996). Differential effects of an erythropoietin receptor gene disruption on primitive and definitive erythropoiesis. Genes Dev 10: 154–164.
Liu F, Poursine-Laurent J, Wu HY, Link DC . (1997). Interleukin-6 and the granulocyte colony-stimulating factor receptor are major independent regulators of granulopoiesis in vivo but are not required for lineage commitment or terminal differentiation. Blood 90: 2583–2590.
Liu F, Wu HY, Wesselschmidt R, Kornaga T, Link DC . (1996). Impaired production and increased apoptosis of neutrophils in granulocyte colony-stimulating factor receptor-deficient mice. Immunity 5: 491–501.
Livnah O, Stura EA, Middleton SA, Johnson DL, Jolliffe LK, Wilson IA . (1999). Crystallographic evidence for preformed dimers of erythropoietin receptor before ligand activation. Science 283: 987–990.
Lodolce JP, Boone DL, Chai S, Swain RE, Dassopoulos T, Trettin S et al. (1998). IL-15 receptor maintains lymphoid homeostasis by supporting lymphocyte homing and proliferation. Immunity 9: 669–676.
Maki K, Sunaga S, Komagata Y, Kodaira Y, Mabuchi A, Karasuyama H et al. (1996). Interleukin 7 receptor-deficient mice lack gammadelta T cells. Proc Natl Acad Sci USA 93: 7172–7177.
Maraskovsky E, O’Reilly LA, Teepe M, Corcoran LM, Peschon JJ, Strasser A . (1997). Bcl-2 can rescue T lymphocyte development in interleukin-7 receptor-deficient mice but not in mutant rag-1−/− mice. Cell 89: 1011–1019.
McArthur GA, Rohrschneider LR, Johnson GR . (1994). Induced expression of c-fms in normal hematopoietic cells shows evidence for both conservation and lineage restriction of signal transduction in response to macrophage colony-stimulating factor. Blood 83: 972–981.
McKinstry WJ, Li CL, Rasko JE, Nicola NA, Johnson GR, Metcalf D . (1997). Cytokine receptor expression on hematopoietic stem and progenitor cells. Blood 89: 65–71.
Metcalf D . (1984). Clonal Culture of Hemopoietic Cells: Techniques and Applications. Elsevier: Amsterdam, The Netherlands.
Metcalf D . (1998). Regulatory mechanisms controlling hematopoiesis: principles and problems. Stem Cells 16(Suppl 1): 3–11.
Metcalf D, Burgess AW . (1982). Clonal analysis of progenitor cell commitment of granulocyte or macrophage production. J Cell Physiol 111: 275–283.
Nandurkar HH, Robb L, Tarlinton D, Barnett L, Kontgen F, Begley CG . (1997). Adult mice with a targeted mutation of the IL-11 receptor (IL11Ra) display normal hematopoiesis. Blood 90: 2148–2159.
Nicola NA . (1989). Hemopoietic cell growth factors and their receptors. Annu Rev Biochem 58: 45–77.
Nicola NA (ed). (1994). Guidebook to Cytokines and Their Receptors. Oxford University Press: Oxford/New York/Tokyo.
Nicola NA, Robb L, Metcalf D, Cary D, Drinkwater CC, Begley CG . (1996). Functional inactivation in mice of the gene for the interleukin-3 (IL-3) specific receptor β-chain: implications for Il-3 function and the mechanism of receptor transmodulation in hematopoietic cells. Blood 87: 2665–2674.
Nishinakamura R, Nakayama N, Hirabayashi Y, Inoue T, Aud D, McNeil T et al. (1995). Mice deficient for the IL-3/GM-CSF/IL-5 βc receptor exhibit lung pathology and impaired immune response, while βIL3 receptor-deficient mice are normal. Immunity 2: 211–222.
Orkin SH . (2000). Diversification of haematopoietic stem cells to specific lineages. Nat Rev Genet 1: 57–64.
Ozaki K, Spolski R, Feng CG, Qi CF, Cheng J, Sher A et al. (2002). A critical role for IL-21 in regulating immunoglobulin production. Science 298: 1630–1634.
Peschon JJ, Morrissey PJ, Grabstein KH, Ramsdell FJ, Maraskovsky E, Gliniak BC et al. (1994). Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J Exp Med 180: 1955–1960.
Pharr PN, Ogawa M, Hofbauer A, Longmore GD . (1994). Expression of an activated erythropoietin or a colony-stimulating factor 1 receptor by pluripotent progenitors enhances colony formation but does not induce differentiation. Proc Natl Acad Sci USA 91: 7482–7486.
Porteu F, Rouyez MC, Cocault L, Benit L, Charon M, Picard F et al. (1996). Functional regions of the mouse thrombopoietin receptor cytoplasmic domain: evidence for a critical region which is involved in differentiation and can be complemented by erythropoietin. Mol Cell Biol 16: 2473–2482.
Richards MK, Liu F, Iwasaki H, Akashi K, Link DC . (2003). Pivotal role of granulocyte colony-stimulating factor in the development of progenitors in the common myeloid pathway. Blood 102: 3562–3568.
Robb L, Drinkwater C, Metcalf D, Li R, Kontgen K, Nicola N et al. (1995). Hematopoietic and lung abnormalities in mice with a null mutation of the common β subunit of the receptors for granulocyte–macrophage colony-stimulating factor and interleukins 3 and 5. Proc Natl Acad Sci USA 92: 9565–9569.
Robb L, Li R, Hartley L, Nandurkar H, Koentgen F, Begley CG . (1998). Infertility in female mice lacking the receptor for interleukin 11 is due to a defective uterine response to implantation. Nat Med 4: 303–308.
Scott CL, Robb L, Mansfield R, Alexander WS, Begley CG . (2000). Granulocyte–macrophage colony-stimulating factor is not responsible for residual thrombopoiesis in mpl null mice. Exp Hematol 28: 1001–1007.
Semerad CL, Liu F, Gregory AD, Stumpf K, Link DC . (2002). G-CSF is an essential regulator of neutrophil trafficking from the bone marrow to the blood. Immunity 17: 413–423.
Semerad CL, Poursine-Laurent J, Liu F, Link DC . (1999). A role for G-CSF receptor signaling in the regulation of hematopoietic cell function but not lineage commitment or differentiation. Immunity 11: 153–161.
Seymour JF, Lieschke GJ, Grail D, Quilici C, Hodgson G, Dunn AR . (1997). Mice lacking both granulocyte colony-stimulating factor (CSF) and granulocyte–macrophage CSF have impaired reproductive capacity, perturbed neonatal granulopoiesis, lung disease, amyloidosis, and reduced long-term survival. Blood 90: 3037–3049.
Shivdasani RA, Orkin SH . (1996). The transcriptional control of hematopoiesis. Blood 87: 4025–4039.
Socolovsky M, Dusanter-Fourt I, Lodish HF . (1997). The prolactin receptor and severely truncated erythropoietin receptors support differentiation of erythroid progenitors. J Biol Chem 272: 14009–14012.
Solar GP, Kerr WG, Zeigler FC, Hess D, Donahue C, de Sauvage FJ et al. (1998). Role of c-mpl in early hematopoiesis. Blood 92: 4–10.
Stanley E, Lieschke GJ, Grail D, Metcalf D, Hodgson G, Gall JAM et al. (1994). Granulocyte/macrophage colony-stimulating factor-deficient mice show no major perturbation of hematopoiesis but develop a characteristic pulmonary pathology. Proc Natl Acad Sci USA 91: 5592–5596.
Stoffel R, Ziegler S, Ghilardi N, Ledermann B, de Sauvage FJ, Skoda RC . (1999). Permissive role of thrombopoietin and granulocyte colony-stimulating factor receptors in hematopoietic cell fate decisions in vivo. Proc Natl Acad Sci USA 96: 698–702.
Suzuki H, Duncan GS, Takimoto H, Mak TW . (1997). Abnormal development of intestinal intraepithelial lymphocytes and peripheral natural killer cells in mice lacking the IL-2 receptor beta chain. J Exp Med 185: 499–505.
Tamada T, Honjo E, Maeda Y, Okamoto T, Ishibashi M, Tokunaga M et al. (2006). Homodimeric cross-over structure of the human granulocyte colony-stimulating factor (GCSF) receptor signaling complex. Proc Natl Acad Sci USA 103: 3135–3140.
Tanaka M, Hirabayashi Y, Sekiguchi T, Inoue T, Katsuki M, Miyajima A . (2003). Targeted disruption of oncostatin M receptor results in altered hematopoiesis. Blood 102: 3154–3162.
Till JE, McCulloch EA, Siminovitch L . (1964). A stochastic model of stem cell proliferation, based on the growth of spleen colony-forming cells. Proc Natl Acad Sci USA 51: 29–36.
Tsai PT, Ohab JJ, Kertesz N, Groszer M, Matter C, Gao J et al. (2006). A critical role of erythropoietin receptor in neurogenesis and post-stroke recovery. J Neurosci 26: 1269–1274.
Ware CB, Horowitz MC, Renshaw BR, Hunt JS, Liggitt D, Koblar SA et al. (1995). Targeted disruption of the low-affinity leukemia inhibitory factor receptor gene causes placental, skeletal, neural and metabolic defects and results in perinatal death. Development 121: 1283–1299.
Watowich SS, Wu H, Socolovsky M, Klingmuller U, Constantinescu SN, Lodish HF . (1996). Cytokine receptor signal transduction and the control of hematopoietic cell development. Annu Rev Cell Dev Biol 12: 91–128.
Wells JA, de Vos AM . (1996). Hematopoietic receptor complexes. Annu Rev Biochem 65: 609–634.
Willerford DM, Chen J, Ferry JA, Davidson L, Ma A, Alt FW . (1995). Interleukin-2 receptor alpha chain regulates the size and content of the peripheral lymphoid compartment. Immunity 3: 521–530.
Wormald S, Hilton DJ . (2004). Inhibitors of cytokine signal transduction. J Biol Chem 279: 821–824.
Wu H, Lee SH, Gao J, Liu X, Iruela-Arispe ML . (1999). Inactivation of erythropoietin leads to defects in cardiac morphogenesis. Development 126: 3597–3605.
Wu H, Liu X, Jaenisch R, Lodish HF . (1995). Generation of committed erythroid BFU-E and CFU-E progenitors does not require erythropoietin or the erythropoietin receptor. Cell 83: 59–67.
Yoshida K, Taga T, Saito M, Suematsu S, Kumanogoh A, Tanaka T et al. (1996a). Targeted disruption of gp130, a common signal transducer for the interleukin 6 family of cytokines, leads to myocardial and hematological disorders. Proc Natl Acad Sci USA 93: 407–411.
Yoshida T, Ikuta K, Sugaya H, Maki K, Takagi M, Kanazawa H et al. (1996b). Defective B-1 cell development and impaired immunity against Angiostrongylus cantonensis in IL-5R alpha-deficient mice. Immunity 4: 483–494.
Acknowledgements
I thank all the scientists past and present of the Cancer and Hematology Division, at the Walter and Eliza Hall Institute for sharing with me their interest and vast knowledge of hematopoiesis. In particular, I thank Professor Donald Metcalf for his help and enthusiasm. Work in the author's laboratory is supported by grants from the National Institute of Health (CA-22556) and the National Health and Medical Research Council of Australia (Program Grants 257500, 461219, Project Grant 321704).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Robb, L. Cytokine receptors and hematopoietic differentiation. Oncogene 26, 6715–6723 (2007). https://doi.org/10.1038/sj.onc.1210756
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210756
Keywords
This article is cited by
-
E3 ubiquitin ligase on the biological properties of hematopoietic stem cell
Journal of Molecular Medicine (2023)
-
Single-chain tandem macrocyclic peptides as a scaffold for growth factor and cytokine mimetics
Communications Biology (2022)
-
Deciphering the Heterogeneity of Mitochondrial Functions During Hematopoietic Lineage Differentiation
Stem Cell Reviews and Reports (2022)
-
In vivo impact of JAK3 A573V mutation revealed using zebrafish
Cellular and Molecular Life Sciences (2022)
-
Vascular Regulation of Hematopoietic Stem Cell Homeostasis, Regeneration, and Aging
Current Stem Cell Reports (2021)