Abstract
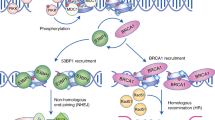
The DSS1 protein interacts with the breast cancer susceptibility protein BRCA2 that plays an integral role in the repair of DNA double-strand breaks (DSBs). DSS1 has also been shown to interact with components of the 26S proteasome in Saccharomyces cerevisiae and in human tumour cells. This raises the possibility of functional interplay between the DNA repair machinery and the proteasome. We show here that human DSS1 interacts with the RPN3 and RPN7 proteasome subunits and define regions of DSS1 important for the interactions with RPN3, RPN7 and BRCA2. We also show that BRCA2 interacts with RPN3 and RPN7 and that the BRCA2/RPN7 interaction is independent of DSS1. Finally, and most significantly, we demonstrate that the proteolytic activity of the proteasome is a determinant of the choice of DSB repair pathway; inhibition of proteasome proteolytic activity results in an increase in the utilization of potentially mutagenic single-strand annealing at the expense of a reduction in the level of error-free gene conversion. This confirms a functional link between DSB repair and proteasomal activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Brummelkamp TR, Bernards R, Agami R . (2002). A system for stable expression of short interfering RNAs in mammalian cells. Science 296: 550–553.
Crackower MA, Scherer SW, Rommens JM, Hui CC, Poorkaj P, Soder S et al. (1996). Characterization of the split hand/split foot malformation locus SHFM1 at 7q21.3–q22.1 and analysis of a candidate gene for its expression during limb development. Hum Mol Genet 5: 571–579.
Funakoshi M, Li X, Velichutina I, Hochstrasser M, Kobayashi H . (2004). Sem1, the yeast ortholog of a human BRCA2-binding protein, is a component of the proteasome regulatory particle that enhances proteasome stability. J Cell Sci 117: 6447–6454.
Gudmundsdottir K, Lord CJ, Witt E, Tutt AN, Ashworth A . (2004). DSS1 is required for RAD51 focus formation and genomic stability in mammalian cells. EMBO Rep 5: 989–993.
Isono E, Saito N, Kamata N, Saeki Y, Toh EA . (2005). Functional analysis of Rpn6p, a lid component of the 26 S proteasome, using temperature-sensitive rpn6 mutants of the yeast Saccharomyces cerevisiae. J Biol Chem 280: 6537–6547.
Josse L, Harley ME, Pires IM, Hughes DA . (2006). Fission yeast Dss1 associates with the proteasome and is required for efficient ubiquitin-dependent proteolysis. Biochem J 393: 303–309.
Kojic M, Yang H, Kostrub CF, Pavletich NP, Holloman WK . (2003). The BRCA2-interacting protein DSS1 is vital for DNA repair, recombination, and genome stability in Ustilago maydis. Mol Cell 12: 1043–1049.
Kojic M, Zhou Q, Lisby M, Holloman WK . (2005). Brh2–Dss1 interplay enables properly controlled recombination in Ustilago maydis. Mol Cell Biol 25: 2547–2557.
Krogan NJ, Lam MH, Fillingham J, Keogh MC, Gebbia M, Li J et al. (2004). Proteasome involvement in the repair of DNA double-strand breaks. Mol Cell 16: 1027–1034.
Li J, Zou C, Bai Y, Wazer DE, Band V, Gao Q . (2006). DSS1 is required for the stability of BRCA2. Oncogene 25: 1186–1194.
Marston NJ, Richards WJ, Hughes D, Bertwistle D, Marshall CJ, Ashworth A . (1999). Interaction between the product of the breast cancer susceptibility gene BRCA2 and DSS1, a protein functionally conserved from yeast to mammals. Mol Cell Biol 19: 4633–4642.
Schauber C, Chen L, Tongaonkar P, Vega I, Lambertson D, Potts W et al. (1998). Rad23 links DNA repair to the ubiquitin/proteasome pathway. Nature 391: 715–718.
Sin N, Kim KB, Elofsson M, Meng L, Auth H, Kwok BH et al. (1999). Total synthesis of the potent proteasome inhibitor epoxomicin: a useful tool for understanding proteasome biology. Bioorg Med Chem Lett 9: 2283–2288.
Sone T, Saeki Y, Toh-e A, Yokosawa H . (2004). Sem1p is a novel subunit of the 26S proteasome from Saccharomyces cerevisiae. J Biol Chem 279: 28807–28816.
Tutt A, Bertwistle D, Valentine J, Gabriel A, Swift S, Ross G et al. (2001). Mutation in Brca2 stimulates error-prone homology-directed repair of DNA double-strand breaks occurring between repeated sequences. EMBO J 20: 4704–4716.
Yang H, Jeffrey PD, Miller J, Kinnucan E, Sun Y, Thoma NH et al. (2002). BRCA2 function in DNA binding and recombination from a BRCA2-DSS1-ssDNA structure. Science 297: 1837–1848.
Acknowledgements
We thank Andrew Tutt for advice in the use and interpretation of the DSB reporter system. This work was funded by Breakthrough Breast Cancer and Cancer Research, UK.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gudmundsdottir, K., Lord, C. & Ashworth, A. The proteasome is involved in determining differential utilization of double-strand break repair pathways. Oncogene 26, 7601–7606 (2007). https://doi.org/10.1038/sj.onc.1210579
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210579
Keywords
This article is cited by
-
Increased chemosensitivity via BRCA2-independent DNA damage in DSS1- and PCID2-depleted breast carcinomas
Laboratory Investigation (2021)
-
PSMD1 and PSMD2 regulate HepG2 cell proliferation and apoptosis via modulating cellular lipid droplet metabolism
BMC Molecular Biology (2019)
-
Next-generation proteasome inhibitor MLN9708 sensitizes breast cancer cells to doxorubicin-induced apoptosis
Scientific Reports (2016)
-
DNA damage emergency: cellular garbage disposal to the rescue?
Oncogene (2014)
-
The proteasomal de-ubiquitinating enzyme POH1 promotes the double-strand DNA break response
The EMBO Journal (2012)