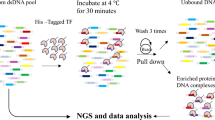
Abstract
p53 and p63 belong to a family of sequence-specific transcription factors regulating key cellular processes. Differential composition of the p53 and p63 DNA-binding sites may contribute to distinct functions of these protein homologues. We used SELEX (systematic evolution of ligands by exponential enrichment) methodology to identify nucleic acid ligands for p63. We found that p63 bound preferentially to DNA fragments conforming to the 20 bp sequence 5′-RRRC(A/G)(A/T)GYYYRRRC(A/T)(C/T)GYYY-3′. Relative to the p53 consensus, the p63 consensus DNA-binding site (DBS) was more degenerate, particularly at positions 10 and 11, and was enriched for A/G at position 5 and C/T at position 16 of the consensus. The differences in DNA-binding site preferences between p63 and p53 influenced their ability to activate transcription from select response elements (REs) in cells. A computer algorithm, p63MH, was developed to find candidate p63-binding motifs on input sequences. We identified genes responsive to p63 regulation that contain functional p63 REs. Our results suggest that the sequence composition of REs could be one contributing factor to target gene discrimination between p63 and p53.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Anwar A, Dehn D, Siegel D, Kepa JK, Tang LJ, Pietenpol JA et al. (2003). Interaction of human NAD(P)H:quinone oxidoreductase 1 (NQO1) with the tumor suppressor protein p53 in cells and cell-free systems. J Biol Chem 278: 10368–10373.
Barbieri CE, Barton CE, Pietenpol JA . (2003). Delta Np63 alpha expression is regulated by the phosphoinositide 3-kinase pathway. J Biol Chem 278: 51408–51414.
Barbieri CE, Perez CA, Johnson KN, Ely KA, Billheimer D, Pietenpol JA . (2005). IGFBP-3 is a direct target of transcriptional regulation by deltaNp63alpha in squamous epithelium. Cancer Res 65: 2314–2320.
Blackwell TK . (1995). Selection of protein binding sites from random nucleic acid sequences. Methods Enzymol 254: 604–618.
Campos EI, Chin MY, Kuo WH, Li G . (2004). Biological functions of the ING family tumor suppressors. Cell Mol Life Sci 61: 2597–2613.
Candi E, Terrinoni A, Rufini A, Chikh A, Lena AM, Suzuki Y et al. (2006). p63 is upstream of IKK alpha in epidermal development. J Cell Sci 119: 4617–4622.
Caserta TM, Kommagani R, Yuan Z, Robbins DJ, Mercer CA, Kadakia M . (2006). p63 overexpression induces the expression of sonic hedgehog. Mol Cancer Res 4: 759–768.
el-Deiry WS, Kern SE, Pietenpol JA, Kinzler KW, Vogelstein B . (1992). Definition of a consensus binding site for p53. Nat Genet 1: 45–49.
Flatt PM, Polyak K, Tang LJ, Scatena CD, Westfall MD, Rubinstein LA et al. (2000). p53-dependent expression of PIG3 during proliferation, genotoxic stress, and reversible growth arrest. Cancer Lett 156: 63–72.
Flatt PM, Price JO, Shaw A, Pietenpol JA . (1998). Differential cell cycle checkpoint response in normal human keratinocytes and fibroblasts. Cell Growth Differ 9: 535–543.
Harms K, Nozell S, Chen X . (2004). The common and distinct target genes of the p53 family transcription factors. Cell Mol Life Sci 61: 822–842.
He TC, Zhou S, da Costa LT, Yu J, Kinzler KW, Vogelstein B . (1998). A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci USA 95: 2509–2514.
Hearnes JM, Mays DJ, Schavolt KL, Tang L, Jiang X, Pietenpol JA . (2005). Chromatin immunoprecipitation-based screen to identify functional genomic binding sites for sequence-specific transactivators. Mol Cell Biol 25: 10148–10158.
Hoh J, Jin S, Parrado T, Edington J, Levine AJ, Ott J . (2002). The p53MH algorithm and its application in detecting p53-responsive genes. Proc Natl Acad Sci USA 99: 8467–8472.
Inga A, Storici F, Darden TA, Resnick MA . (2002). Differential transactivation by the p53 transcription factor is highly dependent on p53 level and promoter target sequence. Mol Cell Biol 22: 8612–8625.
Kent WJ, Sugnet CW, Furey TS, Roskin KM, Pringle TH, Zahler AM et al. (2002). The human genome browser at UCSC. Genome Res 12: 996–1006.
Klein C, Georges G, Kunkele KP, Huber R, Engh RA, Hansen S . (2001). High thermostability and lack of cooperative DNA binding distinguish the p63 core domain from the homologous tumor suppressor p53. J Biol Chem 276: 37390–37401.
Liu G, Chen X . (2002). The ferredoxin reductase gene is regulated by the p53 family and sensitizes cells to oxidative stress-induced apoptosis. Oncogene 21: 7195–7204.
McKay R, DiMaio D . (1981). Binding of an SV40T antigen-related protein to the DNA of SV40 regulatory mutants. Nature 289: 810–813.
Moll UM, Slade N . (2004). p63 and p73: roles in development and tumor formation. Mol Cancer Res 2: 371–386.
Ortt K, Sinha S . (2006). Derivation of the consensus DNA-binding sequence for p63 reveals unique requirements that are distinct from p53. FEBS Lett 580: 4544–4550.
Osada M, Park HL, Nagakawa Y, Yamashita K, Fomenkov A, Kim MS et al. (2005). Differential recognition of response elements determines target gene specificity for p53 and p63. Mol Cell Biol 25: 6077–6089.
Perez CA, Pietenpol JA . (2007). Transcriptional programs regulated by p63 in normal epithelium and tumors. Cell Cycle 6: 246–254.
Pietenpol JA, Lengauer C, Jordan J, Kinzler KW, Vogelstein B . (1996). Mammalian cells resistant to tumor suppressor genes. Proc Natl Acad Sci USA 93: 8390–8394.
Ponassi R, Terrinoni A, Chikh A, Rufini A, Lena AM, Sayan BS et al. (2006). p63 and p73, members of the p53 gene family, transactivate PKCdelta. Biochem Pharmacol 72: 1417–1422.
Sasaki Y, Ishida S, Morimoto I, Yamashita T, Kojima T, Kihara C et al. (2002). The p53 family member genes are involved in the Notch signal pathway. J Biol Chem 277: 719–724.
Shimada A, Kato S, Enjo K, Osada M, Ikawa Y, Kohno K et al. (1999). The transcriptional activities of p53 and its homologue p51/p63: similarities and differences. Cancer Res 59: 2781–2786.
Sniezek JC, Matheny KE, Westfall MD, Pietenpol JA . (2004). Dominant negative p63 isoform expression in head and neck squamous cell carcinoma. Laryngoscope 114: 2063–2072.
Thijs G, Marchal K, Lescot M, Rombauts S, De Moor B, Rouze P et al. (2002). A Gibbs sampling method to detect overrepresented motifs in the upstream regions of coexpressed genes. J Comput Biol 9: 447–464.
Tomso DJ, Inga A, Menendez D, Pittman GS, Campbell MR, Storici F et al. (2005). Functionally distinct polymorphic sequences in the human genome that are targets for p53 transactivation. Proc Natl Acad Sci USA 102: 6431–6436.
Wadham C, Gamble JR, Vadas MA, Khew-Goodall Y . (2003). The protein tyrosine phosphatase Pez is a major phosphatase of adherens junctions and dephosphorylates beta-catenin. Mol Biol Cell 14: 2520–2529.
Westfall MD, Mays DJ, Sniezek JC, Pietenpol JA . (2003). The Delta Np63 alpha phosphoprotein binds the p21 and 14-3-3 sigma promoters in vivo and has transcriptional repressor activity that is reduced by Hay-Wells syndrome-derived mutations. Mol Cell Biol 23: 2264–2276.
Wu G, Nomoto S, Hoque MO, Dracheva T, Osada M, Lee CC et al. (2003). DeltaNp63alpha and TAp63alpha regulate transcription of genes with distinct biological functions in cancer and development. Cancer Res 63: 2351–2357.
Yang A, Zhu Z, Kapranov P, McKeon F, Church GM, Gingeras TR et al. (2006). Relationships between p63 binding, DNA sequence, transcription activity, and biological function in human cells. Mol Cell 24: 593–602.
Ying H, Chang DL, Zheng H, McKeon F, Xiao ZX . (2005). DNA-binding and transactivation activities are essential for TAp63 protein degradation. Mol Cell Biol 25: 6154–6164.
Acknowledgements
This research was supported by the NIH Grants CA70856 and CA105436 (JA Pietenpol), ES00267 and CA68485 (core services), The Komen Foundation Grant DISS02201345, US Army Grant W81XWH-04-1-0304 (CA Perez) and Human Genome Institute Grant MH44292 (J Ott). We thank Dr Ishioka (Tokohu University, Japan) for TAp63γ, Dr Westfall for pAdEasy-1:myc-ΔNp63α adenovirus, Lucy Tang for R304W-ΔNp63α mutant and Dr Torres and Dr Barbieri for critical reading of the manuscript. This research is dedicated to the memory of Dr Philip Browning.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Perez, C., Ott, J., Mays, D. et al. p63 consensus DNA-binding site: identification, analysis and application into a p63MH algorithm. Oncogene 26, 7363–7370 (2007). https://doi.org/10.1038/sj.onc.1210561
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210561
Keywords
This article is cited by
-
Evolutionary history of the p53 family DNA-binding domain: insights from an Alvinella pompejana homolog
Cell Death & Disease (2022)
-
Mutual exclusivity of ESR1 and TP53 mutations in endocrine resistant metastatic breast cancer
npj Breast Cancer (2022)
-
p63, a key regulator of Ago2, links to the microRNA-144 cluster
Cell Death & Disease (2022)
-
G9a controls pluripotent-like identity and tumor-initiating function in human colorectal cancer
Oncogene (2021)
-
Motifome comparison between modern human, Neanderthal and Denisovan
BMC Genomics (2018)