Abstract
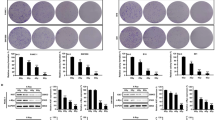
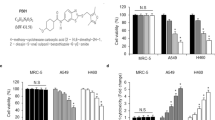
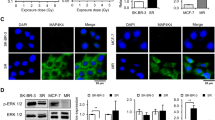
Resistance of pancreatic cancer to current treatments including radiotherapy remains a major challenge in oncology and may be caused by defects in apoptosis programs. Since ‘inhibitor of apoptosis proteins’ (IAPs) block apoptosis at the core of the apoptotic machinery by inhibiting caspases, therapeutic modulation of IAPs could tackle a key resistance mechanism. Here, we report that targeting X-linked inhibitor of apoptosis (XIAP) by RNA-interference-mediated knockdown or overexpression of second mitochondria-derived activator of caspase significantly enhanced apoptosis and markedly reduced clonogenic growth of pancreatic carcinoma cells upon γ-irradiation. Analysis of signaling pathways revealed that antagonizing XIAP increased activation of caspase-2, -3, -8 and -9 and loss of mitochondrial membrane potential upon γ-irradiation. Interestingly, inhibition of caspases also reduced the cooperative effect of XIAP targeting and γ-irradiation to trigger mitochondrial perturbations, suggesting that XIAP controls a feedback mitochondrial amplification loop by regulating caspase activity. Importantly, our data demonstrate for the first time that small molecule XIAP inhibitors sensitized pancreatic carcinoma cells for γ-irradiation-induced apoptosis, whereas they had no effect on γ-irradiation-mediated apoptosis of non-malignant fibroblasts indicating some tumor specificity. In conclusion, targeting XIAP, for example by small molecules, is a promising novel approach to enhance radiosensitivity of pancreatic cancer that warrants further investigation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Cao C, Mu Y, Hallahan DE, Lu B . (2004). XIAP and survivin as therapeutic targets for radiation sensitization in preclinical models of lung cancer. Oncogene 23: 7047–7052.
Cummins JM, Kohli M, Rago C, Kinzler KW, Vogelstein B, Bunz F . (2004). X-linked inhibitor of apoptosis protein (XIAP) is a nonredundant modulator of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-mediated apoptosis in human cancer cells. Cancer Res 64: 3006–3008.
Fulda S, Debatin KM . (2006a). Therapeutic modulation of apoptosis in cancer therapy. In: Debatin, KM, Fulda, S (eds) Apoptosis and Cancer Therapy. Whiley-VCH: Weinheim, pp 849–861.
Fulda S, Debatin KM . (2006b). Extrinsic versus intrinsic apoptosis pathways in anti-cancer chemotherapy. Oncogene 25: 4798–4811.
Fulda S, Debatin KM . (2006c). Targeting inhibitor of apoptosis proteins (IAPs) for diagnosis and treatment of human diseases. Rec Patents Anti-Cancer Drug Disc 1: 81–90.
Fulda S, Sieverts H, Friesen C, Herr I, Debatin KM . (1997). The CD95 (APO-1/Fas) system mediates drug-induced apoptosis in neuroblastoma cells. Cancer Res 57: 3823–3829.
Fulda S, Wick W, Weller M, Debatin KM . (2002). Smac agonists sensitize for Apo2L/TRAIL- or anticancer drug-induced apoptosis and induce regression of malignant glioma in vivo. Nat Med 8: 808–815.
Giagkousiklidis S, Vogler M, Westhoff MA, Kasperczyk H, Debatin KM, Fulda S . (2005). Sensitization for γ-irradiation-induced apoptosis by Smac. Cancer Res 65: 10502–10513.
Guo F, Nimmanapalli R, Paranawithana S, Wittman S, Griffin D, Bali P . (2002). Ectopic overexpression of second mitochondria-derived activator of caspases (Smac/DIABLO) or cotreatment with N-terminus of Smac/DIABLO peptide potentiates epothilone B derivative-(BMS 247550) and Apo-2L/TRAIL-induced apoptosis. Blood 99: 3419–3426.
Holcik M, Yeh C, Korneluk RG, Chow T . (2000). Translational upregulation of X-linked inhibitor of apoptosis (XIAP) increases resistance to radiation induced cell death. Oncogene 19: 4174–4177.
Jendrossek V, Belka C . (2006). Radiation therapy and apoptosis. In: Debatin, KM, Fulda, S (eds) Apoptosis and Cancer Therapy. Whiley-VCH: Weinheim, pp 1049–1086.
Lu B, Mu Y, Cao C, Zeng F, Schneider S, Tan J et al. (2004). Survivin as a therapeutic target for radiation sensitization in lung cancer. Cancer Res 64: 2840–2845.
McManus DC, Lefebvre CA, Cherton-Horvat G, St-Jean M, Kandimalla ER, Agrawal S et al. (2004). Loss of XIAP protein expression by RNAi and antisense approaches sensitizes cancer cells to functionally diverse chemotherapeutics. Oncogene 23: 8105–8117.
Oost TK, Sun C, Armstrong RC, Al-Assaad AS, Betz SF, Deckwerth TL et al. (2004). Discovery of potent antagonists of the antiapoptotic protein XIAP for the treatment of cancer. J Med Chem 47: 4417–4426.
Rodel C, Haas J, Groth A, Grabenbauer GG, Sauer R, Rodel F . (2003). Spontaneous and radiation-induced apoptosis in colorectal carcinoma cells with different intrinsic radiosensitivities: survivin as a radioresistance factor. Int J Radiat Oncol Biol Phys 55: 1341–1347.
Saelens X, Festjens N, Walle LV, van Gurp M, van Loo G, Vandenabeele P . (2004). Toxic proteins released from mitochondria in cell death. Oncogene 23: 2861–2874.
Salvesen GS, Duckett CS . (2002). IAP proteins: blocking the road to death's door. Nat Rev Mol Cell Biol 3: 401–410.
Schmidt-Ullrich RK, Dent P, Grant S, Mikkelsen RB, Valerie K . (2000). Signal transduction and cellular radiation responses. Radiat Res 153: 245–257.
Schneider G, Siveke JT, Eckel F, Schmid RM . (2005). Pancreatic cancer: basic and clinical aspects. Gastroenterology 128: 1606–1625.
Slee EA, Adrain C, Martin SJ . (1999). Serial killers: ordering caspase activation events in apoptosis. Cell Death Differ 6: 1067–1074.
Slee EA, Keogh SA, Martin SJ . (2000). Cleavage of BID during cytotoxic drug and UV radiation-induced apoptosis occurs downstream of the point of Bcl-2 action and is catalysed by caspase-3: a potential feedback loop for amplification of apoptosis-associated mitochondrial cytochrome c release. Cell Death Differ 7: 556–565.
Sohn D, Schulze-Osthoff K, Janicke RU . (2005). Caspase-8 can be activated by interchain proteolysis without receptor-triggered dimerization during drug-induced apoptosis. J Biol Chem 280: 5267–5273.
Turk B, Turk D, Salvesen GS . (2002). Regulating cysteine protease activity: essential role of protease inhibitors as guardians and regulators. Curr Pharm Des 8: 1623–1637.
van Engeland M, Nieland LJ, Ramaekers FC, Schutte B, Reutelingsperger CP . (1998). Annexin V-affinity assay: a review on an apoptosis detection system based on phosphatidylserine exposure. Cytometry 31: 1–9.
Vogler M, Durr K, Jovanovic M, Debatin KM, Fulda S . (2007). Regulation of TRAIL-induced apoptosis by XIAP in pancreatic carcinoma cells. Oncogene 26: 248–257.
Vogler M, Giagkousiklidis S, Genze F, Gschwend JE, Debatin KM, Fulda S . (2005). Inhibition of clonogenic tumor growth: a novel function of Smac contributing to its antitumor activity. Oncogene 24: 7190–7202.
Westphal S, Kalthoff H . (2003). Apoptosis: targets in pancreatic cancer. Mol Cancer 2: 6.
Wilkinson JC, Cepero E, Boise LH, Duckett CS . (2004). Upstream regulatory role for XIAP in receptor-mediated apoptosis. Mol Cell Biol 24: 7003–7014.
Acknowledgements
We thank Idun Pharmaceuticals now Pfizer Inc. for providing XIAP antagonists. The help of Marjana Jovanovic, Katharina Dürr and Meike Vogler to generate pancreatic carcinoma cells with XIAP knockdown or Smac overexpression is greatly appreciated. This work has been partially supported by grants from the Deutsche Forschungsgemeinschaft, the Deutsche Krebshilfe and the Else-Kröner- Fresenius Stiftung (to SF and KMD).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Rights and permissions
About this article
Cite this article
Giagkousiklidis, S., Vellanki, S., Debatin, KM. et al. Sensitization of pancreatic carcinoma cells for γ-irradiation-induced apoptosis by XIAP inhibition. Oncogene 26, 7006–7016 (2007). https://doi.org/10.1038/sj.onc.1210502
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210502
Keywords
This article is cited by
-
Smac mimetic suppresses tunicamycin-induced apoptosis via resolution of ER stress
Cell Death & Disease (2019)
-
An ABCG2 non-substrate anticancer agent FL118 targets drug-resistant cancer stem-like cells and overcomes treatment resistance of human pancreatic cancer
Journal of Experimental & Clinical Cancer Research (2018)
-
Targeting IAP proteins in combination with radiotherapy
Radiation Oncology (2015)
-
Clinical significance of autophagic protein LC3 levels and its correlation with XIAP expression in hepatocellular carcinoma
Medical Oncology (2014)
-
Smac mimetic compound LCL161 sensitizes esophageal carcinoma cells to radiotherapy by inhibiting the expression of inhibitor of apoptosis protein
Tumor Biology (2014)