Abstract
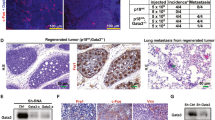
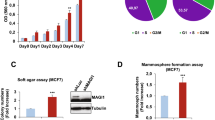
Overexpression of the adaptor/scaffolding protein Gab2 has been detected in primary human breast cancer cells and cell lines, although its functional significance in breast carcinogenesis is not fully understood. Here, we show a requirement for Gab2 in promoting mammary tumor metastasis. Although Gab2 expression levels were elevated in mammary tumors induced by the Neu (ErbB-2) oncogene, homozygous deletion of Gab2 in mice had only a modest effect on the initiation of Neu-induced mammary tumors. Notably, ablation of Gab2 severely suppressed lung metastasis. Gab2-deficient cancer cells displayed normal Akt activities, and their proliferative rate in vitro was similar to control cells. However, Gab2−/− cancer cells exhibited decreased migration and impaired Erk activation, and the defects were rescued by re-introduction of Gab2 into Gab2−/− cells. These findings suggest that although Gab2 overexpression may confer growth advantage to tumor cells, the functional requirement for Gab2 in mammary tumor initiation/growth may be dispensable, and that Gab2 may have a prominent role in promoting mammary tumor metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Abassi YA, Jackson JA, Zhu J, O'Connell J, Wang X, Xu X . (2004). Label-free, real-time monitoring of IgE-mediated mast cell activation on microelectronic cell sensor arrays. J Immunol Methods 292: 195–205.
Amundadottir LT, Leder P . (1998). Signal transduction pathways activated and required for mammary carcinogenesis in response to specific oncogenes. Oncogene 16: 737–746.
Bentires-Alj M, Gil SG, Chan R, Wang ZC, Wang Y, Imanaka N et al. (2006). A role for the scaffolding adapter GAB2 in breast cancer. Nat Med 12: 114–121.
Bourguignon LY, Gunja-Smith Z, Iida N, Zhu HB, Young LJ, Muller WJ et al. (1998). CD44v(3,8–10) is involved in cytoskeleton-mediated tumor cell migration and matrix metalloproteinase (MMP-9) association in metastatic breast cancer cells. J Cell Physiol 176: 206–215.
Brummer T, Schramek D, Hayes VM, Bennett HL, Caldon CE, Musgrove EA et al. (2005). Increased proliferation and altered growth factor dependency of human mammary epithelial cells overexpressing the Gab2 docking protein. J Biol Chem 281: 626–637.
Daly RJ, Gu H, Parmar J, Malaney S, Lyons RJ, Kairouz R et al. (2002). The docking protein Gab2 is overexpressed and estrogen regulated in human breast cancer. Oncogene 21: 5175–5181.
Dankort D, Maslikowski B, Warner N, Kanno N, Kim H, Wang Z et al. (2001). Grb2 and Shc adapter proteins play distinct roles in Neu (ErbB-2)-induced mammary tumorigenesis: implications for human breast cancer. Mol Cell Biol 21: 1540–1551.
Gu H, Neel BG . (2003). The ‘Gab’ in signal transduction. Trends Cell Biol 13: 122–130.
Huang E, Cheng SH, Dressman H, Pittman J, Tsou MH, Horng CF et al. (2003). Gene expression predictors of breast cancer outcomes. Lancet 361: 1590–1596.
Liu Y, Rohrschneider LR . (2002). The gift of Gab. FEBS Lett 515: 1–7.
Meng S, Chen Z, Munoz-Antonia T, Wu J . (2005). Participation of both Gab1 and Gab2 in the activation of the ERK/MAPK pathway by epidermal growth factor. Biochem J 391: 143–151.
Muller WJ, Sinn E, Pattengale PK, Wallace R, Leder P . (1988). Single-step induction of mammary adenocarcinoma in transgenic mice bearing the activated c-neu oncogene. Cell 54: 105–115.
Olayioye MA, Neve RM, Lane HA, Hynes NE . (2000). The ErbB signaling network: receptor heterodimerization in development and cancer. EMBO J 19: 3159–3167.
Reddy KB, Nabha SM, Atanaskova N . (2003). Role of MAP kinase in tumor progression and invasion. Cancer Metastasis Rev 22: 395–403.
Siegel PM, Ryan ED, Cardiff RD, Muller WJ . (1999). Elevated expression of activated forms of Neu/ErbB-2 and ErbB-3 are involved in the induction of mammary tumors in transgenic mice: implications for human breast cancer. EMBO J 18: 2149–2164.
Sivaraman VS, Wang H, Nuovo GJ, Malbon CC . (1997). Hyperexpression of mitogen-activated protein kinase in human breast cancer. J Clin Invest 99: 1478–1483.
Slamon D, Pegram M . (2001). Rationale for trastuzumab (Herceptin) in adjuvant breast cancer trials. Semin Oncol 28: 13–19.
Webster MA, Hutchinson JN, Rauh MJ, Muthuswamy SK, Anton M, Tortorice CG et al. (1998). Requirement for both Shc and phosphatidylinositol 3′ kinase signaling pathways in polyomavirus middle T-mediated mammary tumorigenesis. Mol Cell Biol 18: 2344–2359.
White DE, Kurpios NA, Zuo D, Hassell JA, Blaess S, Mueller U et al. (2004). Targeted disruption of beta1-integrin in a transgenic mouse model of human breast cancer reveals an essential role in mammary tumor induction. Cancer Cell 6: 159–170.
Xing JZ, Zhu L, Jackson JA, Gabos S, Sun XJ, Wang XB et al. (2005). Dynamic monitoring of cytotoxicity on microelectronic sensors. Chem Res Toxicol 18: 154–161.
Yamada K, Nishida K, Hibi M, Hirano T, Matsuda Y . (2001). Comparative FISH mapping of Gab1 and Gab2 genes in human, mouse and rat. Cytogenet Cell Genet 94: 39–42.
Yu WM, Hawley TS, Hawley RG, Qu CK . (2002). Role of the docking protein Gab2 in beta(1)-integrin signaling pathway-mediated hematopoietic cell adhesion and migration. Blood 99: 2351–2359.
Zhang EE, Chapeau E, Hagihara K, Feng GS . (2004). Neuronal Shp2 tyrosine phosphatase controls energy balance and metabolism. Proc Natl Acad Sci USA 101: 16064–16069.
Zhao C, Yu DH, Shen R, Feng GS . (1999). Gab2, a new pleckstrin homology domain-containing adapter protein, acts to uncouple signaling from ERK kinase to Elk-1. J Biol Chem 274: 19649–19654.
Acknowledgements
We thank Dr R Cardiff at UC Davis for pathological examination of tumor metastasis in lung, Dr DH Yu for work done in the early stages of the Gab2 gene targeting project, X Xiao (Aceabio sciences Inc) for helpful discussion and generosity in providing RT-CES biosensor and also J Zhu for technical assistance, Dr D Schlaepfer at Scripps for anti-FAK antibody. This work was supported by NIH grants (HL66208, CA78606, CA102583). YK is a recipient of a postdoctoral fellowship (9FB-0170) from the California Breast Cancer Research Program. WJM is supported by the CRC Chair in Molecular Oncology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ke, Y., Wu, D., Princen, F. et al. Role of Gab2 in mammary tumorigenesis and metastasis. Oncogene 26, 4951–4960 (2007). https://doi.org/10.1038/sj.onc.1210315
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210315
Keywords
This article is cited by
-
Liver-specific overexpression of Gab2 accelerates hepatocellular carcinoma progression by activating immunosuppression of myeloid-derived suppressor cells
Oncogene (2022)
-
Gab2 deficiency suppresses high-fat diet-induced obesity by reducing adipose tissue inflammation and increasing brown adipose function in mice
Cell Death & Disease (2021)
-
USP35, regulated by estrogen and AKT, promotes breast tumorigenesis by stabilizing and enhancing transcriptional activity of estrogen receptor α
Cell Death & Disease (2021)
-
Elevated Gab2 induces tumor growth and angiogenesis in colorectal cancer through upregulating VEGF levels
Journal of Experimental & Clinical Cancer Research (2017)
-
Gab2 facilitates epithelial-to-mesenchymal transition via the MEK/ERK/MMP signaling in colorectal cancer
Journal of Experimental & Clinical Cancer Research (2016)