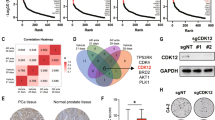
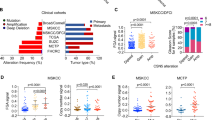
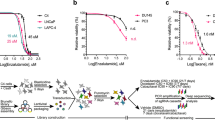
Abstract
A major obstacle for clinicians in the treatment of advanced prostate cancer is the inevitable progression to chemoresistance, especially to docetaxel. It is essential to understand the molecular events that lead to docetaxel resistance in order to identify means to prevent or interfere with chemoresistance. In initial attempts to detect these events, we analysed genomic differences between non-resistant and docetaxel-resistant prostate tumor cells and, of the genes modulated by docetaxel treatment, we observed Stat1 and clusterin gene expression heightened in the resistant phenotype. In this study, we provide biochemical and biological evidence that these two gene products are related. Stat1 and clusterin protein expression was induced upon docetaxel treatment of DU145 cells and highly overexpressed in the docetaxel-resistant DU145 cells (DU145-DR). The increase in total Stat1 corresponded to an increase in phosphorylated Stat1. Interestingly, there was no detectable difference between DU145 and DU145-DR cells expression of total Stat3 and phosphorylated Stat3. Treatment of DU145-DR cells with small interfering RNA targeted for Stat1 not only resulted in the knockdown of Stat1 expression, but it also caused the inhibition of clusterin expression. Thus, Stat1 appears to play a key role in the regulation of clusterin. Remarkably, inhibition of Stat1 or clusterin expression resulted in the re-sensitization of DU145-DR cells to docetaxel. These results offer the first evidence that Stat1, and its subsequent regulation of clusterin, are essential for docetaxel resistance in prostate cancer. Targeting this pathway could be a potential therapeutic means for intervention of docetaxel resistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Assikis VJ, Do KA, Wen S, Wang X, Cho-Vega JH, Brisbay S et al. (2004). Clin Cancer Res 10: 6770–6778.
Barton BE, Murphy TF, Shu P, Huang HF, Meyenhofer M, Barton A . (2004). Mol Cancer Ther 3: 1183–1191.
Bettuzzi S, Davalli P, Astancolle S, Carani C, Madeo B, Tampieri A et al. (2000). Cancer Res 60: 28–34.
Cochet O, Frelin C, Peyron JF, Imbert V . (2006). Cell Signal 18: 449–455.
Culine S, Droz JP . (2000). Ann Oncol 11: 1523–1530.
Darnell Jr JE . (1997). Science 277: 1630–1635.
Debatin KM, Poncet D, Kroemer G . (2002). Oncogene 21: 8786–8803.
De Marzo AM, DeWeese TL, Platz EA, Meeker AK, Nakayama M, Epstein JI et al. (2004). J Cell Biochem 91: 459–477.
DeMiguel F, Lee SO, Lou W, Xiao X, Pflug BR, Nelson JB et al. (2002). Prostate 52: 123–129.
Dhir R, Ni Z, Lou W, DeMiguel F, Grandis JR, Gao AC . (2002). Prostate 51: 241–246.
El-Hashemite N, Zhang H, Walker V, Hoffmeister KM, Kwiatkowski DJ . (2004). Cancer Res 64: 3436–3443.
Geney R, Ungureanu M, Ojima I . (2002). Clin Chem Lab Med 40: 918–925.
Hengartner MO . (2000). Nature 407: 770–776.
Herr I, Debatin KM . (2001). Blood 98: 2603–2614.
Ho SM, Leav I, Ghatak S, Merk F, Jagannathan VS, Mallery K . (1998). Am J Pathol 153: 131–139.
Horvath CM . (2000). Trends Biochem Sci 25: 496–502.
Humphreys DT, Carver JA, Easterbrook-Smith SB, Wilson MR . (1999). J Biol Chem 274: 6875–6881.
Johnstone RW, Ruefli AA, Lowe SW . (2002). Cell 108: 153–164.
July LV, Beraldi E, So A, Fazli L, Evans K, English JC et al. (2004). Mol Cancer Ther 3: 223–232.
Lacroix H, Ligeza C . (1998). Expert Opin Investig Drugs 7: 273–281.
Lee SO, Lou W, Johnson CS, Trump DL, Gao AC . (2004). Prostate 60: 178–186.
Legrand A, Vadrot N, Lardeux B, Bringuier AF, Guillot R, Feldmann G . (2004). Liver Int 24: 149–160.
Liu K, Caldwell SA, Abrams SI . (2005). Cancer Res 65: 1045–1054.
Lowe SW, Lin AW . (2000). Carcinogenesis 21: 485–495.
Kominsky SL, Hobeika AC, Lake FA, Torres BA, Johnson HM . (2000). Cancer Res 60: 3904–3908.
Makarovskiy AN, Siryaporn E, Hixson DC, Akerley W . (2002). Cell Mol Life Sci 59: 1198–1211.
Milbank AJ, Dreicer R, Klein EA . (2002). Urology 60: 738–741.
Miller ML, Ojima I . (2001). Chem Rec 1: 195–211.
Miyake H, Chi KN, Gleave ME . (2000a). Clin Cancer Res 6: 1655–1663.
Miyake H, Hara I, Kamidono S, Gleave ME, Eto H . (2003). Oncol Rep 10: 469–473.
Miyake H, Nelson C, Rennie PS, Gleave ME . (2000b). Cancer Res 60: 170–176.
Miyake H, Pollak M, Gleave ME . (2000c). Cancer Res 60: 3058–3064.
Nelson WG, De Marzo AM, Isaacs WB . (2003). N Engl J Med 349: 366–381.
Nickerson T, Miyake H, Gleave ME, Pollak M . (1999). Cancer Res 59: 3392–3395.
O'Sullivan J, Whyte L, Drake J, Tenniswood M . (2003). Cell Death Differ 10: 914–927.
Ouyang XS, Wang X, Lee DT, Tsao SW, Wong YC . (2001). Carcinogenesis 22: 965–973.
Peto J . (2001). Nature 411: 390–395.
Petrylak DP . (2005). Urology 65: 3–7.
Petrylak DP, Tangen CM, Hussain MH, Lara Jr PN, Jones JA, Taplin ME et al. (2004). N Engl J Med 351: 1513–1520.
Pucci S, Bonanno E, Pichiorri F, Angeloni C, Spagnoli LG . (2004). Oncogene 23: 2298–2304.
Stephanou A, Latchman DS . (2003). Int J Exp Pathol 84: 239–244.
Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN et al. (2004). N Engl J Med 351: 1502–1512.
Thomas M, Finnegan CE, Rogers KM, Purcell JW, Trimble A, Johnston PG et al. (2004). Cancer Res 64: 8357–8364.
Trougakos IP, Gonos ES . (2002). Int J Biochem Cell Biol 34: 1430–1448.
Trougakos IP, So A, Jansen B, Gleave ME, Gonos ES . (2004). Cancer Res 64: 1834–1842.
Wang T, Niu G, Kortylewski M, Burdelya L, Shain K, Zhang S et al. (2004). Nat Med 10: 48–54.
Zellweger T, Kiyama S, Chi K, Miyake H, Adomat H, Skov K et al. (2003). BJU Int 92: 463–469.
Zellweger T, Miyake H, Cooper S, Chi K, Conklin BS, Monia BP et al. (2001). J Pharmacol Exp Ther 298: 934–940.
Acknowledgements
This work was supported by NIH Grants AI056213 and CA98080, and by Aventis Pharmaceuticals. We thank Linda Mora for her technical support on immunohistochemistry. We would also like to thank the Molecular Imaging Core and the Flow Cytometry Core at H Lee Moffitt Cancer Center.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Patterson, S., Wei, S., Chen, X. et al. Novel role of Stat1 in the development of docetaxel resistance in prostate tumor cells. Oncogene 25, 6113–6122 (2006). https://doi.org/10.1038/sj.onc.1209632
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209632
Keywords
This article is cited by
-
The role and function of CLU in cancer biology and therapy
Clinical and Experimental Medicine (2022)
-
STAT signaling as a target for intervention: from cancer inflammation and angiogenesis to non-coding RNAs modulation
Molecular Biology Reports (2022)
-
Cellular rewiring in lethal prostate cancer: the architect of drug resistance
Nature Reviews Urology (2020)
-
Cytochrome P450 3A4, 3A5, and 2C8 expression in breast, prostate, lung, endometrial, and ovarian tumors: relevance for resistance to taxanes
Cancer Chemotherapy and Pharmacology (2019)
-
Silencing of miR-193a-5p increases the chemosensitivity of prostate cancer cells to docetaxel
Journal of Experimental & Clinical Cancer Research (2017)