Abstract
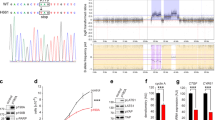
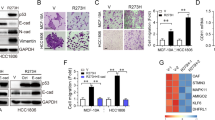
The tumor suppressor KLF6 is a member of the Krüppel-like family of transcription factors, which has been implicated in the pathogenesis of several human carcinomas. Uncovering the transcriptional targets relevant for its tumorigenic properties, including cellular proliferation and invasion, will be essential to understanding possible mechanisms by which KLF6 and its antagonistic splice form, KLF6-SV1, regulate this development. To begin defining possible metastatic-related pathways, we analysed the effect of KLF6 dysregulation on a recognized suppressor of cellular invasion, E-cadherin. Targeted KLF6 reduction in an ovarian cancer cell line, SKOV-3, resulted in a 50% reduction of E-cadherin expression (P<0.01) and conversely, KLF6-SV1 silencing upregulated E-cadherin approximately fivefold (P<0.0001). These changes resulted from KLF6 directly transactivating the E-cadherin promoter as demonstrated by luciferase promoter assay and chromatin immunoprecipitation (ChIP). KLF6-mediated changes in E-cadherin levels were accompanied by downstream changes in both the subcellular localization of β-catenin and c-myc expression levels. Moreover, and consistent with these experimental findings, patient-derived epithelial ovarian tumors with low KLF6 and high KLF6-SV1 expression ratios had significantly decreased E-cadherin expression (P<0.0001). These combined findings highlight the E-cadherin pathway as a novel and functionally important mediator by which changes in KLF6 and KLF6-SV1 can directly alter ovarian tumor invasion and metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ananthanarayanan M, Li S, Balasubramaniyan N, Suchy FJ, Walsh MJ . (2004). J Biol Chem 279: 54348–54357.
Benzeno S, Narla G, Allina J, Cheng GZ, Reeves HL, Banck MS et al. (2004). Cancer Res 64: 3885–3891.
Berchuck A, Charney M . (1997). Biochem Pharmacol 54: 541–544.
Bieker JJ . (2001). J Biol Chem 276: 34355–34358.
Black AR, Black JD, Azizkhan-Clifford J . (2001). J Cell Physiol 188: 143–160.
Chen C, Hyytinen ER, Sun X, Helin HJ, Koivisto PA, Frierson HF et al. (2003). Am J Pathol 162: 1349–1354.
Chen HK, Liu XQ, Lin J, Chen TY, Feng QS, Zeng YX et al. (2002). Ai Zheng 21: 1047–1050.
Cho YG, Kim CJ, Park CH, Yang YM, Kim SY, Nam SW et al. (2005). Oncogene 24: 4588–4590.
Conacci-Sorrell M, Simcha I, Ben-Yedidia T, Blechman J, Savagner P, Ben-Ze'ev A . (2003). J Cell Biology 163: 847–857.
DiFeo A, Narla G, Hirsnfeld J, Camacho-Vanegas O, Narla J, Rose S et al. (2006). Clin Cancer Res (in press).
Frixen UH, Behrens J, Sachs M, Eberle G, Voss B, Warda A et al. (1991). J Cell Biol 113: 173–185.
Gabbert HE, Mueller W, Schneiders A, Meier S, Moll R, Birchmeier W et al. (1996). Int J Cancer 69: 184–189.
Glinsky GV, Glinskii AB, Stephenson AJ, Hoffman RM, Gerald WI . (2004). J Clin Invest 113: 913–923.
Hosono S, Gross I, English MA, Hajra KM, Fearon ER, Licht JD et al. (2000). J Biol Chem 275: 10943–10953.
Ito G, Uchiyama M, Kondo M, Mori S, Usami N, Maeda O et al. (2004). Cancer Res 64: 3838–3843.
Jeng YM, Hsu HC . (2003). Int J Cancer 105: 625–629.
Kaczynski J, Cook T, Urrutia R . (2003). Genome Biol 4: 206.
Kettunen E, Anttila S, Seppanen JK, Karjalainen A, Edgren H, Lindstrom I et al. (2004). Cancer Genet Cytogenet 149: 98–106.
Kim Y, Ratziu V, Choi SG, Lalazar A, Theiss G, Dang Q et al. (1998). J Biol Chem 273: 33750–33758.
Kremer-Tal S, Reeves HL, Narla G, Tnung SN, Schwartz M, DiFeo A et al. (2004). Hepatology 40: 1047–1052.
Macleod K, Mullen P, Sewell J, Rabiasz G, Lawrie S, Miller E et al. (2005). Cancer Res 65: 6789–6800.
Naora H, Montell DJ . (2005). Nat Rev Cancer 5: 355–366.
Narla G, DiFeo A, Reeves HL, Schaid DJ, Hirshfeld J, Hod E et al. (2005a). Cancer Res 65: 1213–1222.
Narla G, DiFeo A, Yao S, Banno A, Hod E, Reeves HL et al. (2005b). Cancer Res 65: 5761–5768.
Narla G, Heath KE, Reeves HL, Li D, Giono LE, Kimmelman AC et al. (2001). Science 294: 2563–2566.
Reeves HL, Narla G, Ogunbiyi O, Haq AI, Katz A, Benzeno S et al. (2004). Gastroenterology 126: 1090–1103.
Rubinstein M, Idelman G, Plymate SR, Narla G, Friedman SL, Werner H et al. (2004). Endocrinology 145: 3769–3777.
Singh D, Febbo PG, Ross K, Jackson DG, Manola J, Ladd C et al. (2002). Cancer Cell 2: 203–209.
Slavin DA, Koritschoner NP, Prieto CC, Lopez-Diaz FJ, Chatton B, Bocco JL et al. (2004). Oncogene 23: 8196–8205.
Stanbrough M, Bubley GJ, Ross K, Golub TR, Rubin MA, Penning TM et al. (2006). Cancer Res 66: 2815–2825.
Stockinger A, Eger A, Wolf J, Beug H, Foisner R . (2001). J Cell Biol 154: 1185–1196.
Tal-Kremer S, Reeves HL, Narla G, Thung SN, Schwartz M, DiFeo A et al. (2004). Hepatology 40: 1047–1052.
Vleminckx K, Vakaet Jr L, Mareel M, Fiers W, van Roy F et al. (1991). Cell 66: 107–119.
Acknowledgements
This research was financially supported by a Grant, JAM: DAMD17-03-10129.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
DiFeo, A., Narla, G., Camacho-Vanegas, O. et al. E-cadherin is a novel transcriptional target of the KLF6 tumor suppressor. Oncogene 25, 6026–6031 (2006). https://doi.org/10.1038/sj.onc.1209611
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209611
Keywords
This article is cited by
-
miR-106a-5p carried by tumor-derived extracellular vesicles promotes the invasion and metastasis of ovarian cancer by targeting KLF6
Clinical & Experimental Metastasis (2022)
-
KLF7: a new candidate biomarker and therapeutic target for high-grade serous ovarian cancer
Journal of Experimental & Clinical Cancer Research (2020)
-
Hotspot mutant p53-R273H inhibits KLF6 expression to promote cell migration and tumor metastasis
Cell Death & Disease (2020)
-
Blastocyst-like structures generated solely from stem cells
Nature (2018)
-
A novel KLF6-Rho GTPase axis regulates hepatocellular carcinoma cell migration and dissemination
Oncogene (2016)