Abstract
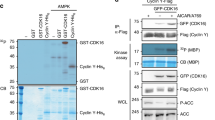
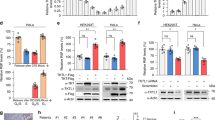
We report here that glyceraldehyde-3-phosphate dehydrogenase (GAPDH) interacts in vitro and in vivo with the protein SET. This interaction is performed through the acidic domain of SET located at the carboxy terminal region. On analysing the functional relevance of SET-GAPDH interaction, we observed that GAPDH reverses in a dose-dependent manner, the inhibition of cyclin B-cdk1 activity produced by SET. Similarly to SET, GAPDH associates with cyclin B, suggesting that the regulation of cyclin B-cdk1 activity might be mediated not only by the interaction of GAPDH with SET but also with cyclin B. To analyse the putative role of GAPDH on cell cycle progression, HCT116 cells were transfected with a GAPDH expression vector. Results indicate that overexpression of GAPDH does not affect the timing of DNA replication but induces an increase in the number of mitosis, an advancement of the peak of cyclin B-cdk1 activity and an acceleration of cell cycle progression. All these results suggest that GAPDH might be involved in cell cycle regulation by modulating cyclin B-cdk1 activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Adachi Y, Pavlakis GN, Copeland TD . (1994). J Biol Chem 269: 2258–2262.
Adler HT, Nallaseth FS, Walter G, Tkachuk DC . (1997). J Biol Chem 272: 28407–28414.
Beresford PJ, Zhang D, Oh DY, Fan Z, Greer EL, Russo ML et al. (2001). J Biol Chem 276: 43285–43293.
Canela N, Rodriguez-Vilarrupla A, Estanyol JM, Diaz C, Pujol MJ, Agell N et al (2003). J Biol Chem 278: 1158–1164.
Carrascal M, Carujo S, Bachs O, Abian J . (2002). Proteomics 2: 455–468.
Cervoni N, Detich N, Seo SB, Chakravarti D, Szyf M . (2002). J Biol Chem 277: 25026–25031.
Chai Z, Sarcevic B, Mawson A, Toh BH . (2001). J Biol Chem 276: 33665–33674.
Chen J, Jackson PK, Kirschner MW, Dutta A . (1995). Nature 374: 386–388.
Chuang DM, Hough C, Senatorov VV . (2005). Annu Rev Pharmacol Toxicol 45: 269–290.
Coqueret O . (2003). Trends Cell Biol 13: 65–70.
Estanyol JM, Jaumot M, Casanovas O, Rodriguez-Vilarrupla A, Agell N, Bachs O . (1999). J Biol Chem 274: 33161–33165.
Fan Z, Beresford PJ, Oh DY, Zhang D, Lieberman J . (2003). Cell 112: 659–672.
Fan Z, Beresford PJ, Zhang D, Lieberman J . (2002). Mol Cell Biol 22: 2810–2820.
Glaser PE, Gross RW . (1995). Biochemistry 34: 12193–12203.
Hara MR, Agrawal N, Kim SF, Cascio MB, Fujimuro M, Ozeki Y et al. (2005). Nat Cell Biol 7: 665–674.
Kawase H, Okuwaki M, Miyaji M, Ohba R, Handa H, Ishimi Y et al. (1996). Genes Cells 1: 1045–1056.
Kellogg DR, Kikuchi A, Fujii-Nakata T, Turck CW, Murray AW . (1995). J Cell Biol 130: 661–673.
Krynetski EY, Krynetskaia NF, Bianchi ME, Evans WE . (2003). Cancer Res 63: 100–106.
Krynetski EY, Krynetskaia NF, Gallo AE, Murti KG, Evans WE . (2001). Mol Pharmacol 59: 367–374.
Kutney SN, Hong R, Macfarlan T, Chakravarti D . (2004). J Biol Chem 279: 30850–30855.
Li M, Makkinje A, Damuni Z . (1996). J Biol Chem 271: 11059–11062.
Lieberman J, Fan Z . (2003). Curr Opin Immunol 15: 553–559.
Luo Y, Hurwitz J, Massague J . (1995). Nature 375: 159–161.
Matsumoto K, Nagata K, Ui M, Hanaoka F . (1993). J Biol Chem 268: 10582–10587.
Morgan DO . (1997). Annu Rev Cell Dev Biol 13: 261–291.
Nagata K, Kawase H, Handa H, Yano K, Yamasaki M, Ishimi Y et al. (1995). Proc Natl Acad Sci USA 92: 4279–4283.
Nakagawa T, Hirano Y, Inomata A, Yokota S, Miyachi K, Kaneda M et al. (2003). J Biol Chem 278: 20395–20404.
Nakanishi M, Adami GR, Robetorye RS, Noda A, Venable SF, Dimitrov D et al. (1995). Proc Natl Acad Sci USA 92: 4352–4356.
Okuwaki M, Nagata K . (1998). J Biol Chem 273: 34511–34518.
Ozbun LL, You L, Kiang S, Angdisen J, Martinez A, Jakowlew SB . (2001). Genomics 73: 179–193.
Pandey AV, Mellon SH, Miller WL . (2003). J Biol Chem 278: 2837–2844.
Qu D, Li Q, Lim HY, Cheung NS, Li R, Wang JH et al. (2002). J Biol Chem 277: 7324–7332.
Robbins AR, Ward RD, Oliver C . (1995). J Cell Biol 130: 1093–1104.
Sanchez-Piris M, Posas F, Alemany V, Winge I, Hidalgo E, Bachs O et al. (2002). J Biol Chem 277: 17722–17727.
Seo SB, McNamara P, Heo S, Turner A, Lane WS, Chakravarti D . (2001). Cell 104: 119–130.
Shen HH, Huang AM, Hoheisel J, Tsai SF . (2001). Genomics 71: 21–33.
Shevchenko A, Chernushevich I, Wilm M, Mann M . (2000). Methods Mol Biol 146: 1–16.
Shikama N, Chan HM, Krstic-Demonacos M, Smith L, Lee CW, Cairns W et al. (2000). Mol Cell Biol 20: 8933–8943.
Singh R, Green MR . (1993). Science 259: 365–368.
Sirover MA . (1999). Biochim Biophys Acta 1432: 159–184.
Sundararaj KP, Wood RE, Ponnusamy S, Salas AM, Szulc Z, Bielawska A et al. (2004). J Biol Chem 279: 6152–6162.
Tisdale EJ . (2001). J Biol Chem 276: 2480–2486.
Vogel T, Dittrich O, Mehraein Y, Dechend F, Schnieders F, Schmidtke J . (1998). Cytogenet Cell Genet 81: 265–270.
von Lindern M, van Baal S, Wiegant J, Raap A, Hagemeijer A, Grosveld G . (1992). Mol Cell Biol 12: 3346–3355.
Wen C, Levitan D, Li X, Greenwald I . (2000). Proc Natl Acad Sci USA 97: 14524–14529.
Yi M, Schultz DE, Lemon SM . (2000). J Virol 74: 6459–6468.
Zheng L, Roeder RG, Luo Y . (2003). Cell 114: 255–266.
Acknowledgements
We are grateful to Dr Sonia Brun for technical support and to Dr Núria Canela and Jorge Vera for their collaboration. Mass spectrometry analysis were performed at the ‘Unitat de proteòmica dels Serveis cientific-tècnics de la Universitat de Barcelona, Facultat de Medicina’. This work was supported by Grants SAF2002-00452, SAF 2003-08329, GEN2003-20243-C08 from the Ministerio de Educación y Ciencia of Spain. Financial support was also obtained from the networks of cooperative research on Cancer (C03/10) from the Instituto Carlos III.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carujo, S., Estanyol, J., Ejarque, A. et al. Glyceraldehyde 3-phosphate dehydrogenase is a SET-binding protein and regulates cyclin B-cdk1 activity. Oncogene 25, 4033–4042 (2006). https://doi.org/10.1038/sj.onc.1209433
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209433
Keywords
This article is cited by
-
Targeting CDK1 in cancer: mechanisms and implications
npj Precision Oncology (2023)
-
Targeting of SET/I2PP2A oncoprotein inhibits Gli1 transcription revealing a new modulator of Hedgehog signaling
Scientific Reports (2021)
-
Probiotic Aspergillus oryzae produces anti-tumor mediator and exerts anti-tumor effects in pancreatic cancer through the p38 MAPK signaling pathway
Scientific Reports (2021)
-
Glyceraldehyde-3-phosphate dehydrogenase is overexpressed in colorectal cancer onset
Translational Medicine Communications (2017)
-
Molecular association of glucose-6-phosphate isomerase and pyruvate kinase M2 with glyceraldehyde-3-phosphate dehydrogenase in cancer cells
BMC Cancer (2016)