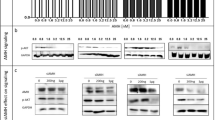
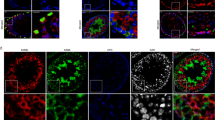
Abstract
Anti-Müllerian hormone (AMH) is a member of the transforming growth factor-β family responsible for regression of Müllerian ducts during male sexual differentiation and for regulation of gonadal steroidogenesis. AMH is also a gonadal tumor suppressor which mediates its effects through a specific type II receptor and the bone morphogenetic protein (BMP)-specific Smad proteins, suggesting that AMH and BMPs could also share type I receptors, namely activin-like kinases (ALKs)2, 3 or 6. However, attempts to identify a unique AMH type I receptor among them were unsuccessful. Here, using kinase-deficient type I receptors and small interfering RNA technology, we demonstrate that, in an AMH Sertoli target cell line, ALK3 mediates AMH effects on both Smad1 activation and P450 side-chain cleavage enzyme. In addition, transfecting a combination of normal and kinase-deficient receptors, we show that ALK2 can compensate for the absence of ALK3 and probably acts in synergy with ALK3 at high concentrations of AMH to activate Smad1, whereas ALK6 has a competitive inhibitory effect. These results are a first step in understanding how AMH transduces its effects in immature Sertoli cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Allard S, Adin P, Gouédard L, di Clemente N, Josso N, Orgebin-Crist MC, Picard JY and Xavier F . (2000). Development, 127, 3349–3360.
Aoki H, Fujii M, Imamura T, Yagi K, Takehara K, Kato M and Miyazono K . (2001). J. Cell Sci., 114, 1483–1489.
Arango NA, Lovell-Badge R and Behringer RR . (1999). Cell, 99, 409–419.
Clarke TR, Hoshiya Y, Yi SE, Liu XH, Lyons KM and Donahoe PK . (2001). Mol. Endocrinol., 15, 946–959.
di Clemente N, Wilson CA, Faure E, Boussin L, Carmillo P, Tizard R, Picard JY, Vigier B, Josso N and Cate RL . (1994). Mol. Endocrinol., 8, 1006–1020.
Dutertre M, Gouédard L, Xavier F, Long WQ, di Clemente N, Picard JY and Rey R . (2001). Endocrinology, 142, 4040–4046.
Dutertre M, Rey R, Porteu A, Josso N and Picard JY . (1997). Mol. Cell. Endocrinol., 136, 57–65.
Faure E, Gouédard L, Imbeaud S, Cate RL, Picard JY, Josso N and di Clemente N . (1996). J. Biol. Chem., 271, 30571–30575.
Gouédard L, Chen YG, Thevenet L, Racine C, Borie S, Lamarre I, Josso N, Massagué J and di Clemente N . (2000). J. Biol. Chem., 275, 27973–27978.
Hay E, Lemonnier J, Fromigue O, Guenou H and Marie PJ . (2004). J. Biol. Chem., 279, 1650–1658.
Hoshiya Y, Gupta V, Segev DL, Hoshiya M, Carey JL, Sasur LM, Tran TT, Ha TU and Maheswaran S . (2003). Mol. Cell. Endocrinol., 211, 43–49.
Imbeaud S, Faure E, Lamarre I, Mattei MG, di Clemente N, Tizard R, Carré-Eusèbe D, Belville C, Tragethon L, Tonkin C, Nelson J, McAuliffe M, Bidart JM, Lababidi A, Josso N, Cate RL and Picard JY . (1995). Nat. Genet., 11, 382–388.
Jamin SP, Arango NA, Mishina Y, Hanks MC and Behringer RR . (2002). Nat. Genet., 32, 408–410.
Jamin SP, Arango NA, Mishina Y, Hanks MC and Behringer RR . (2003). Mol. Cell. Endocrinol., 211, 15–19.
Josso N and di Clemente N . (2003). Trends Endocrinol. Metab., 14, 91–97.
Lagna G, Hata A, Hemmat-Brivanlou A and Massagué J . (1996). Nature, 383, 832–836.
Lo RS, Chen YG, Shi YG, Pavletich NP and Massagué J . (1998). EMBO J., 17, 996–1005.
Long WQ, Ranchin V, Pautier P, Belville C, Denizot P, Cailla H, Lhommé C, Picard JY, Bidart JM and Rey R . (2000). J. Clin. Endocrinol. Metab., 85, 540–544.
Massagué J and Wotton D . (2000). EMBO J., 19, 1745–1754.
Messika-Zeitoun L, Gouédard L, Belville C, Dutertre M, Lins L, Imbeaud S, Hughes IA, Picard JY, Josso N and di Clemente N . (2001). J. Clin. Endocrinol. Metab., 86, 4390–4397.
Mishina Y, Rey R, Finegold MJ, Matzuk MM, Josso N, Cate RL and Behringer RR . (1996). Genes Dev., 10, 2577–2587.
Shi YG and Massagué J . (2003). Cell, 113, 685–700.
Teixeira J, Maheswaran S and Donahoe PK . (2001). Endocr. Rev., 22, 657–674.
ten Dijke P, Korchynskyi O, Valdimarsdottir G and Goumans MJ . (2003). Mol. Cell. Endocrinol., 211, 105–113.
Vigier B, Picard JY, Tran D, Legeai L and Josso N . (1984). Endocrinology, 114, 1315–1320.
Visser JA, Olaso R, Verhoef-Post M, Kramer P, Themmen APN and Ingraham HA . (2001). Mol. Endocrinol., 15, 936–945.
Wieser R, Attisano L, Wrana JL and Massagué J . (1993). Mol. Cell. Biol., 13, 7239–7247.
Acknowledgements
We thank Drs J Massagué, A Atfi, G Rawadi and S Maheswaran for kindly providing DNA constructs. We are grateful to Nathalie Ledée and Sylvie Dubanchet for advice on real-time PCR. We are grateful to the Association pour la Recherche sur le Cancer for its generous support to Nathalie di Clemente (grant no. 4253).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Belville, C., Jamin, S., Picard, JY. et al. Role of type I receptors for anti-Müllerian hormone in the SMAT-1 Sertoli cell line. Oncogene 24, 4984–4992 (2005). https://doi.org/10.1038/sj.onc.1208686
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208686
Keywords
This article is cited by
-
New insights into anti-Müllerian hormone role in the hypothalamic–pituitary–gonadal axis and neuroendocrine development
Cellular and Molecular Life Sciences (2021)
-
Immunohistochemical characterization of the anti-Müllerian hormone receptor type 2 (AMHR-2) in human testes
Endocrine (2020)
-
Differential molecular regulation of processing and membrane expression of Type-I BMP receptors: implications for signaling
Cellular and Molecular Life Sciences (2017)
-
Altered testicular development as a consequence of increase number of sertoli cell in male lambs exposed prenatally to excess testosterone
Endocrine (2013)
-
The potential role of microRNAs in regulating gonadal sex differentiation in the chicken embryo
Chromosome Research (2012)